NEET Exam > NEET Notes > Chemistry Class 11 > Mind Map: Equilibrium
Mind Map: Equilibrium
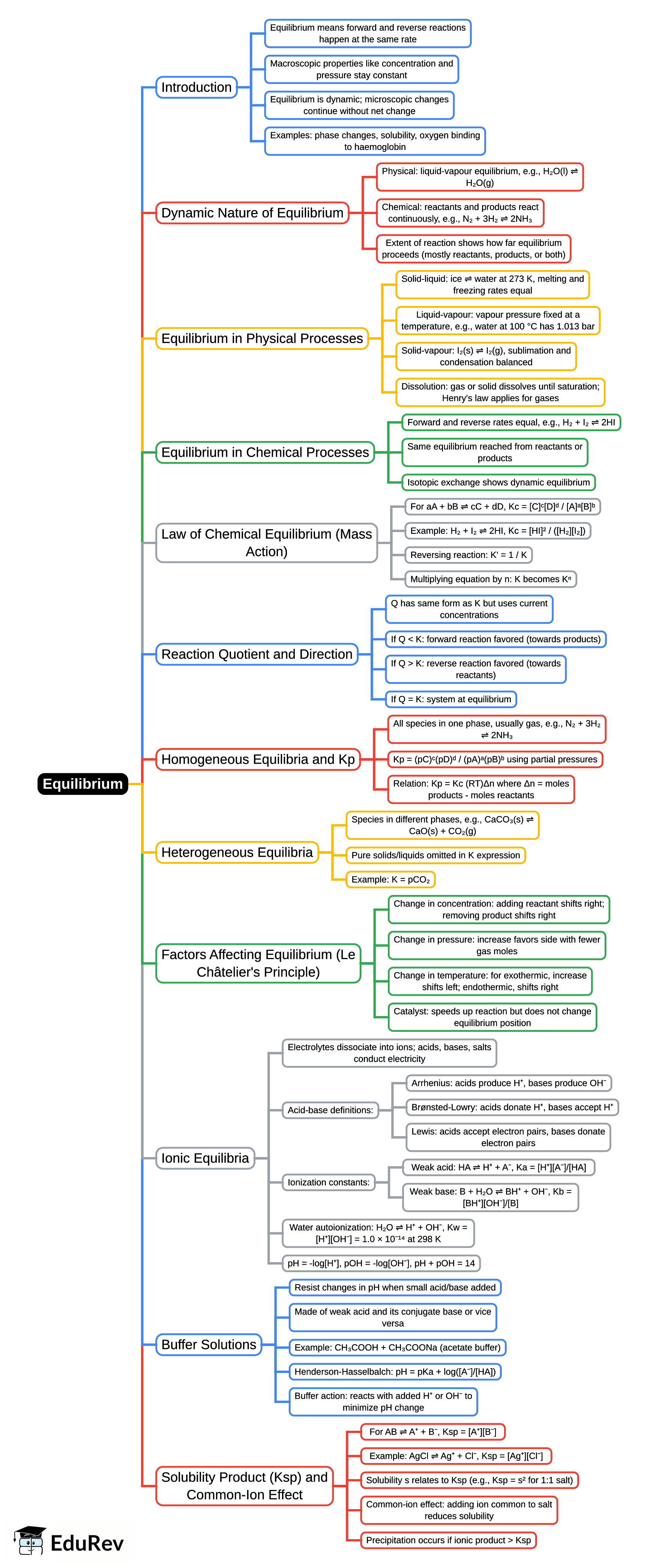
The document Mind Map: Equilibrium is a part of the NEET Course Chemistry Class 11.
All you need of NEET at this link: NEET
FAQs on Mind Map: Equilibrium
| 1. What is the difference between chemical equilibrium and dynamic equilibrium in reversible reactions? |  |
Ans. Chemical equilibrium occurs when forward and reverse reaction rates become equal, stopping visible changes. Dynamic equilibrium means reactions continue at molecular level despite no observable macroscopic changes. In dynamic equilibrium, both forward and backward reactions proceed simultaneously at identical rates, maintaining constant concentrations of reactants and products throughout the system.
| 2. How do I calculate the equilibrium constant (Kc) and what does a large or small value actually mean? |  |
Ans. The equilibrium constant Kc equals the ratio of product concentrations to reactant concentrations, each raised to their stoichiometric coefficients at equilibrium. A large Kc value indicates products are favoured, meaning the reaction proceeds nearly to completion. A small Kc suggests reactants are favoured, with minimal product formation at equilibrium state.
| 3. Why does increasing pressure shift equilibrium towards fewer moles of gas according to Le Chatelier's principle? |  |
Ans. Systems respond to pressure changes by shifting equilibrium toward the side with fewer total moles of gas molecules. This reduces overall volume and internal pressure, counteracting the applied external pressure. For example, in N₂O₄ ⇌ 2NO₂, increasing pressure shifts left toward N₂O₄ since it involves fewer gas molecules, minimising system resistance.
| 4. What's the connection between Gibbs free energy and whether a reaction will reach equilibrium naturally? |  |
Ans. Gibbs free energy (ΔG) determines reaction spontaneity and equilibrium position. When ΔG is negative, reactions proceed spontaneously forward; when positive, they favour reverse reactions. At equilibrium, ΔG equals zero, indicating no net change occurs. The relationship ΔG° = -RT ln(K) directly links standard free energy to the equilibrium constant value.
| 5. How can temperature changes affect both the equilibrium position and the equilibrium constant itself? |  |
Ans. Temperature shifts equilibrium position by favouring endothermic or exothermic directions depending on whether heat increases or decreases. Unlike concentration or pressure changes, temperature actually alters the equilibrium constant value itself. For endothermic reactions, higher temperatures increase Kc; for exothermic reactions, higher temperatures decrease Kc, fundamentally changing the equilibrium state.
Related Searches
Summary, Free, Semester Notes, study material, past year papers, practice quizzes, Important questions, ppt, video lectures, pdf , Sample Paper, Viva Questions, Extra Questions, Objective type Questions, Previous Year Questions with Solutions, MCQs, Mind Map: Equilibrium, mock tests for examination, Exam, Mind Map: Equilibrium, Mind Map: Equilibrium, shortcuts and tricks;






