NEET Exam > NEET Notes > Chemistry Class 11 > Mind Map: Redox Reaction
Mind Map: Redox Reaction
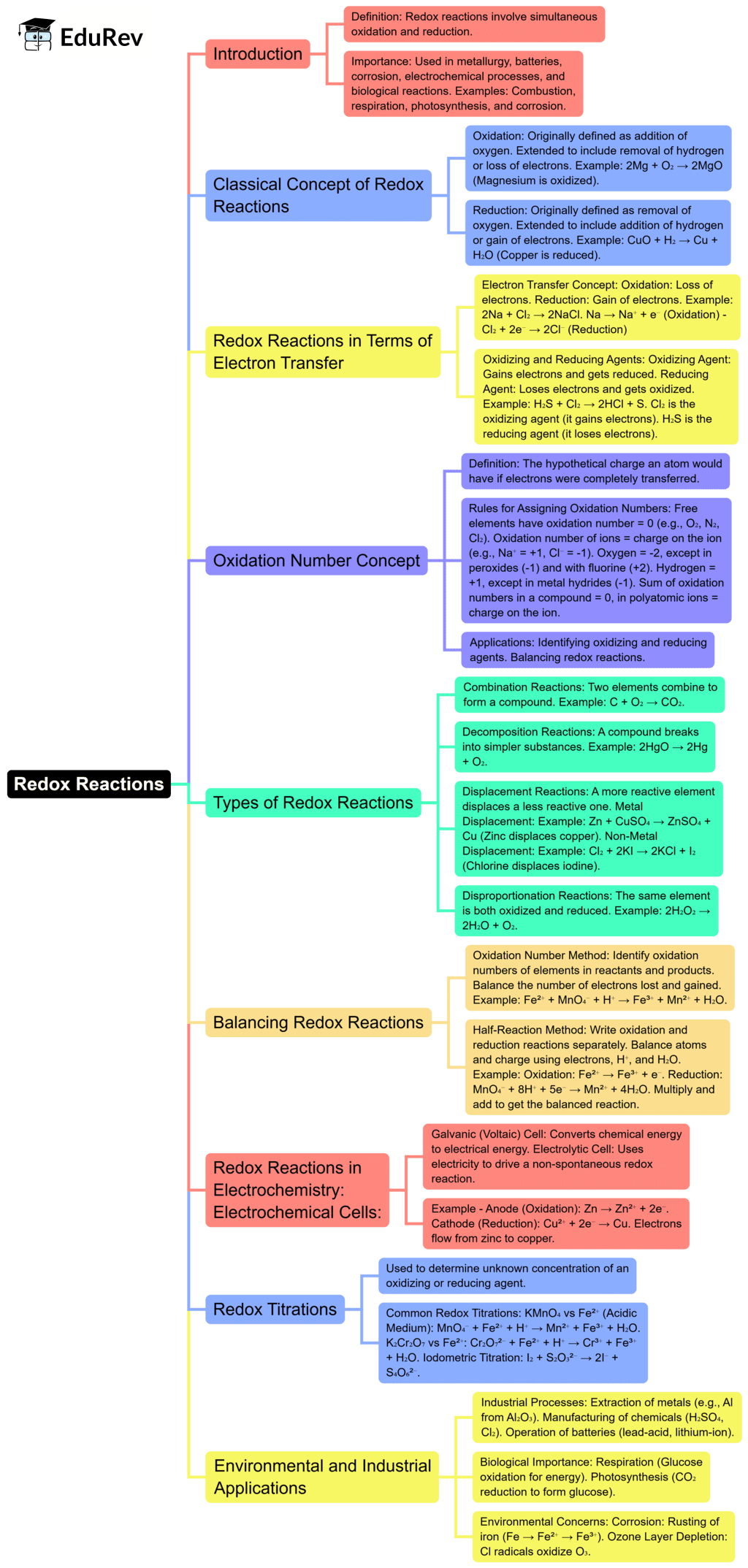
The document Mind Map: Redox Reaction is a part of the NEET Course Chemistry Class 11.
All you need of NEET at this link: NEET
FAQs on Mind Map: Redox Reaction
| 1. How do I identify oxidation numbers in a redox reaction quickly? |  |
Ans. Oxidation numbers are assigned using specific rules: elements in their elemental state have zero, monatomic ions equal their charge, oxygen is typically -2, and hydrogen is usually +1. Assign numbers to all atoms, then compare before and after the reaction-whichever element's number increases undergoes oxidation, while the one decreasing undergoes reduction. Practice with common compounds to build speed for NEET Chemistry.
| 2. What's the difference between oxidation and reduction in redox reactions? |  |
Ans. Oxidation is the loss of electrons (or gain of oxygen/loss of hydrogen), while reduction is the gain of electrons (or loss of oxygen/gain of hydrogen). These always occur together in redox reactions-one substance oxidises while another reduces simultaneously. Understanding this electron transfer concept is fundamental to balancing equations and identifying oxidising and reducing agents in redox chemistry problems.
| 3. Why do some elements act as both oxidising and reducing agents? |  |
Ans. Elements with variable oxidation states (like sulphur, nitrogen, and halogens) can both lose and gain electrons depending on their reaction partners. Sulphur in +4 state can be oxidised to +6 or reduced to lower states. This disproportionation behaviour appears frequently in NEET questions. Refer to mind maps and flashcards to recognise which elements commonly display this dual reactivity pattern.
| 4. How do I balance redox equations using the oxidation number method? |  |
Ans. First, assign oxidation numbers to identify which atoms are oxidised and reduced. Calculate the total electron loss and gain, then adjust coefficients so electrons lost equal electrons gained. This algebraic balancing method works best for ionic and covalent redox reactions. Visual aids like mind maps help clarify the electron redistribution across complex equations in Class 11 Chemistry.
| 5. What exactly is a redox pair, and why does it matter for exams? |  |
Ans. A redox pair comprises an oxidising agent (substance that gains electrons and gets reduced) and a reducing agent (substance that loses electrons and gets oxidised). Recognising these pairs helps predict reaction direction and products. NEET frequently tests understanding of conjugate redox pairs-like Fe²⁺/Fe³⁺ or MnO₄⁻/Mn²⁺-making this concept essential for scoring marks in chemical reaction questions.
Related Searches
ppt, shortcuts and tricks, practice quizzes, Free, pdf , Previous Year Questions with Solutions, Exam, video lectures, Important questions, Summary, Mind Map: Redox Reaction, Mind Map: Redox Reaction, Extra Questions, Semester Notes, study material, past year papers, Mind Map: Redox Reaction, Viva Questions, mock tests for examination, Sample Paper, MCQs, Objective type Questions;






