NEET Exam > NEET Notes > Physics Class 11 > Mind Map: Thermal Properties of Matter
Mind Map: Thermal Properties of Matter
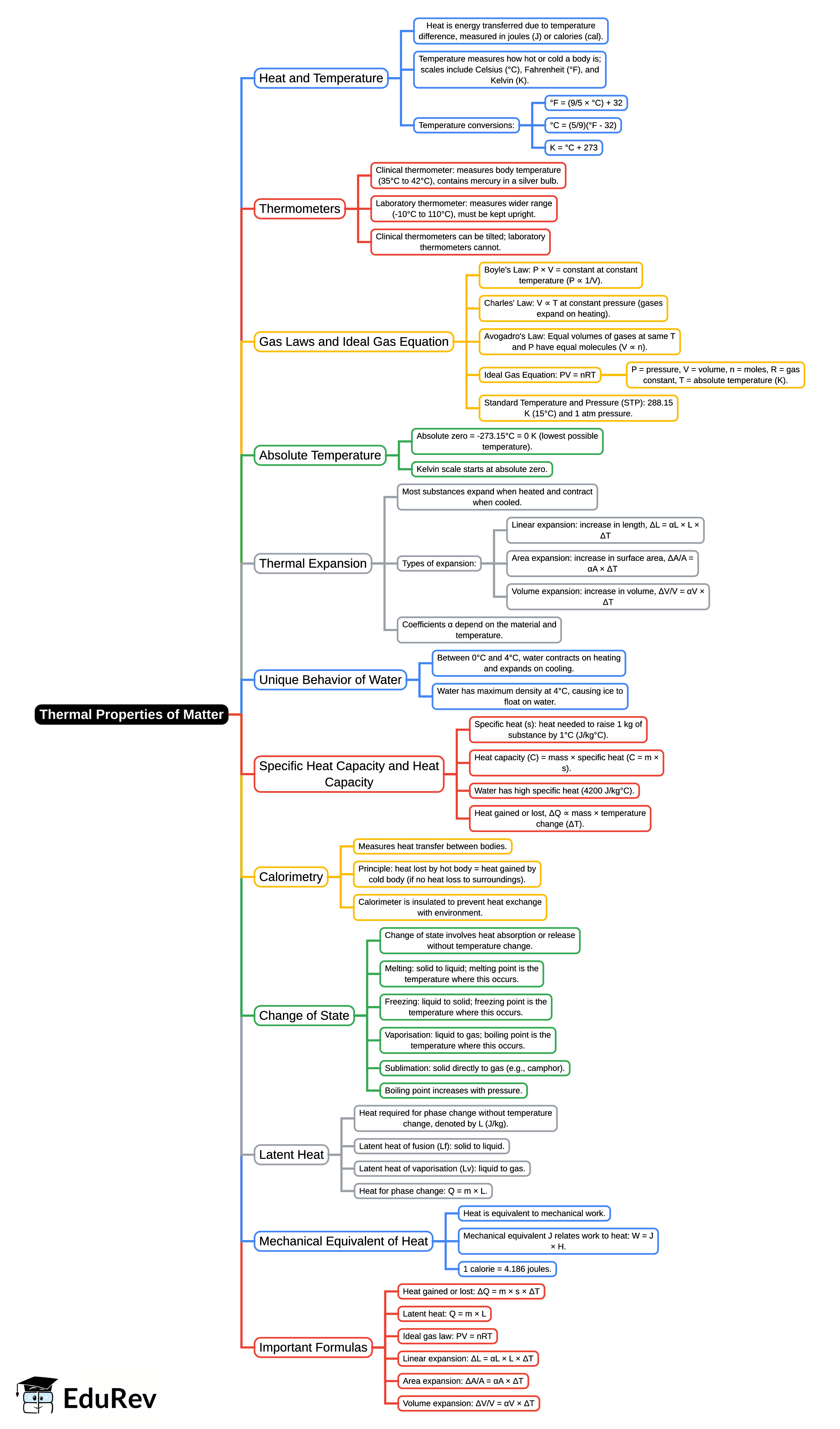
The document Mind Map: Thermal Properties of Matter is a part of the NEET Course Physics Class 11.
All you need of NEET at this link: NEET
FAQs on Mind Map: Thermal Properties of Matter
| 1. What's the difference between heat and temperature in thermal properties of matter? |  |
Ans. Heat is the transfer of thermal energy between objects at different temperatures, while temperature measures the average kinetic energy of molecules in a substance. Temperature is a property of matter itself; heat is energy in motion. Understanding this distinction is crucial for NEET physics, as many thermal properties questions test whether students confuse these two fundamental concepts.
| 2. How does thermal expansion work and why do materials expand when heated? |  |
Ans. When heated, molecules gain kinetic energy and vibrate more vigorously, increasing the average distance between them, causing the material to expand. This occurs in solids, liquids, and gases differently-solids show linear and volumetric expansion, while liquids and gases expand primarily by volume. Thermal expansion coefficients quantify this expansion rate and vary by material type, making this essential for solving NEET problems on bridges, railway tracks, and liquid containers.
| 3. What is specific heat capacity and how do I calculate it in exam questions? |  |
Ans. Specific heat capacity is the amount of heat energy required to raise the temperature of one kilogramme of a substance by one degree Celsius. The formula Q = mcΔT connects heat absorbed (Q), mass (m), specific heat capacity (c), and temperature change (ΔT). Different materials have different specific heat capacities; water has exceptionally high capacity, which is why it's used as a coolant in thermal applications studied in NEET.
| 4. Why do some materials conduct heat faster than others-what determines thermal conductivity? |  |
Ans. Thermal conductivity depends on a material's molecular structure and how freely electrons or vibrational energy can move through it. Metals conduct heat rapidly due to free electrons; poor conductors like wood and rubber have tightly bound electrons. Understanding thermal conductivity helps explain real-world phenomena like why metals feel cold to touch and why insulators are used in thermal applications-common NEET conceptual questions.
| 5. What's latent heat and when does it matter in thermal properties questions? |  |
Ans. Latent heat is the thermal energy absorbed or released during a phase change (solid to liquid or liquid to gas) without changing temperature. During melting or boiling, energy goes into breaking molecular bonds rather than increasing kinetic energy. Latent heat of fusion and vaporisation are distinct values; recognising when a substance undergoes phase change without temperature change is critical for solving NEET thermal properties problems accurately.
Related Searches
Objective type Questions, practice quizzes, Summary, shortcuts and tricks, Previous Year Questions with Solutions, mock tests for examination, study material, Semester Notes, MCQs, Extra Questions, video lectures, Viva Questions, Sample Paper, Mind Map: Thermal Properties of Matter, past year papers, ppt, Mind Map: Thermal Properties of Matter, Exam, Free, Important questions, pdf , Mind Map: Thermal Properties of Matter;






