NEET Exam > NEET Notes > Physics Class 11 > Mind Map: Thermodynamics
Mind Map: Thermodynamics
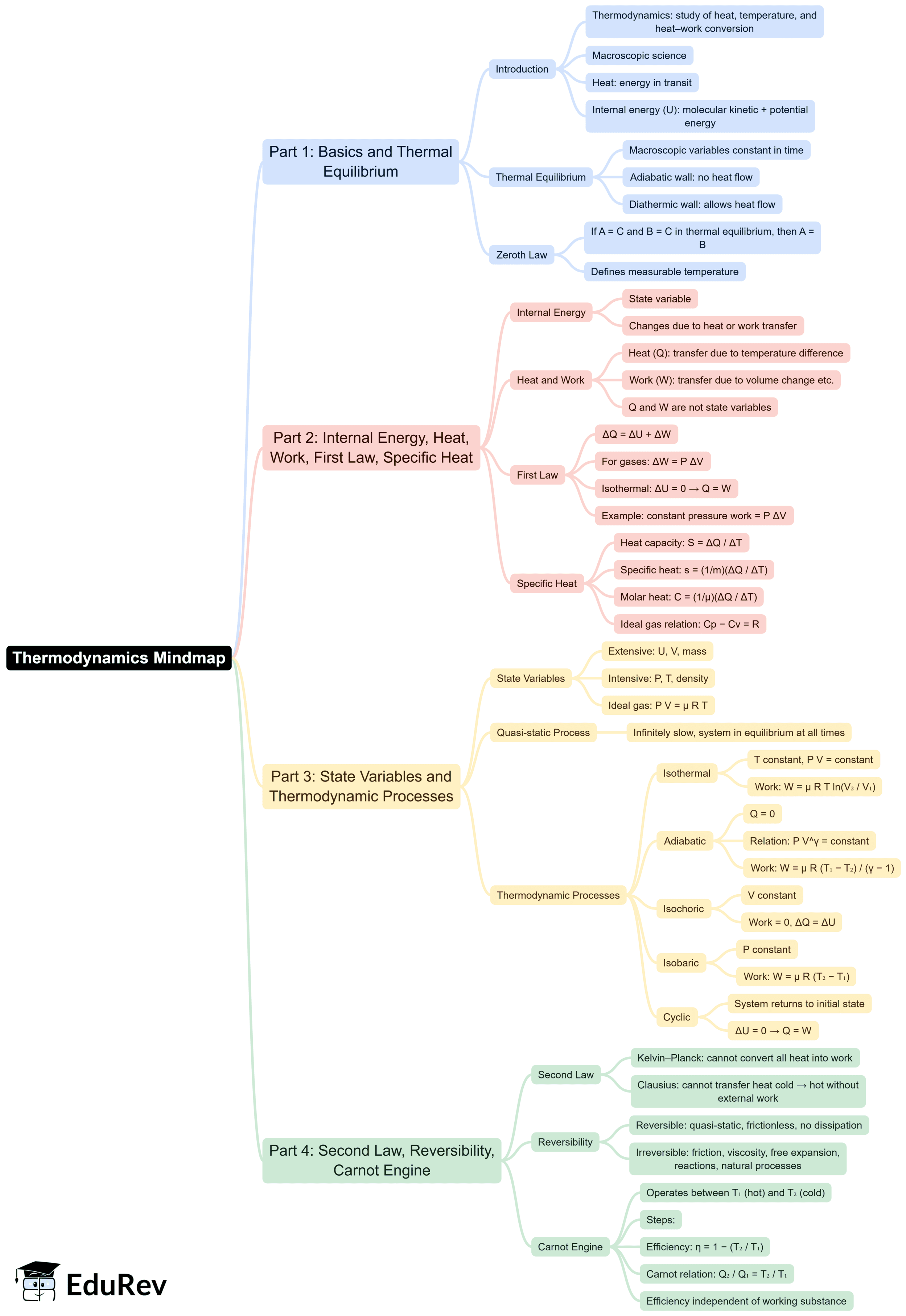
The document Mind Map: Thermodynamics is a part of the NEET Course Physics Class 11.
All you need of NEET at this link: NEET
FAQs on Mind Map: Thermodynamics
| 1. What are the first law of thermodynamics and second law of thermodynamics, and how do they differ? |  |
Ans. The first law states that energy cannot be created or destroyed, only converted between forms-it's about energy conservation in a system. The second law states that entropy in an isolated system always increases, meaning heat naturally flows from hot to cold objects. While the first law governs energy balance, the second law determines the direction and feasibility of thermodynamic processes.
| 2. How do I identify whether a thermodynamic process is isothermal, adiabatic, isobaric, or isochoric? |  |
Ans. Each process has distinct characteristics: isothermal maintains constant temperature (ΔT = 0), adiabatic involves no heat exchange (Q = 0), isobaric keeps pressure constant (ΔP = 0), and isochoric maintains constant volume (ΔV = 0). Students can use mind maps to visualize these processes side-by-side, noting how pressure, volume, and temperature change differently in each scenario during thermodynamic cycles.
| 3. Why does internal energy change in some thermodynamic processes but not others? |  |
Ans. Internal energy depends only on temperature for an ideal gas. In isothermal processes, temperature remains constant, so internal energy doesn't change regardless of pressure or volume shifts. In adiabatic, isobaric, or isochoric processes, temperature typically changes, causing internal energy to increase or decrease. Understanding this relationship clarifies why work and heat distribution vary across different thermodynamic pathways.
| 4. What's the difference between heat capacity at constant pressure and heat capacity at constant volume? |  |
Ans. Heat capacity at constant pressure (Cp) measures energy needed to raise temperature when pressure stays fixed, while heat capacity at constant volume (Cv) measures energy needed when volume stays fixed. Cp is always greater than Cv because at constant pressure, some energy goes into doing work against external pressure. The relationship Cp - Cv = R (gas constant) helps predict energy requirements in different heating scenarios.
| 5. How do I approach thermodynamic cycle problems, and what role does the P-V diagram play? |  |
Ans. A P-V (pressure-volume) diagram visually represents thermodynamic cycles, showing how pressure and volume change through each process stage. The area enclosed within the cycle represents net work done by or on the system. By identifying each segment as isothermal, adiabatic, isobaric, or isochoric, students can calculate work, heat, and internal energy changes systematically using appropriate equations and formulas for Class 11 NEET preparation.
Related Searches
Free, Viva Questions, shortcuts and tricks, Extra Questions, mock tests for examination, pdf , Sample Paper, practice quizzes, Previous Year Questions with Solutions, ppt, Semester Notes, Summary, study material, Mind Map: Thermodynamics, Mind Map: Thermodynamics, Important questions, past year papers, MCQs, Exam, video lectures, Mind Map: Thermodynamics, Objective type Questions;






