Electronic Configuration of Elements & Periodic Table
Introduction: What are Electron Configurations?
Electron configuration describes the distribution of electrons in the atomic orbitals of an atom.
It follows a standard notation in which subshells are written in order of increasing energy, with superscripts indicating the number of electrons present. For example, the electron configuration of sodium (Z = 11) is 1s² 2s² 2p⁶ 3s¹.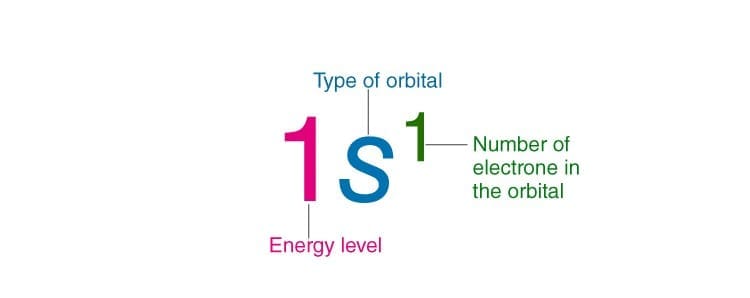
For elements with many electrons the full notation becomes long. An abbreviated (condensed) form uses the symbol of the nearest preceding noble gas, placed in square brackets, to represent the core (completely filled) subshells. For example, sodium can be written in the noble-gas shorthand as [Ne] 3s1, since neon has the configuration 1s2 2s2 2p6. This notation helps simplify configurations of heavier elements and is commonly used while writing configurations of transition metals. Electron configurations are useful for:
Electron configurations are useful for:
- determining the number of valence electrons and hence the likely bonding behaviour and valency of an element;
- explaining similarities of elements in the same group (elements with similar outer-shell configurations show similar chemical properties);
- predicting the magnetic nature of atoms and ions (paramagnetic or diamagnetic) based on the presence of unpaired electrons.
The use of this notation and ideas about discrete electron energy levels followed developments that began with the Bohr model and were refined by quantum mechanics after 1913.
Writing Electron Configurations
Shells
Electrons are arranged in shells identified by the principal quantum number n. The maximum possible number of electrons that can be accommodated in a shell is given by the formula 2n². where n = 1, 2, 3, ...
Shell and 'n' value | Max. Electrons in the Electron Configuration |
K shell, n = 1 | 2*12 = 2 |
L shell, n = 2 | 2*22 = 8 |
M shell, n = 3 | 2*32 = 18 |
N shell, n = 4 | 2*42 = 32 |
Subshells
- Each shell (value of n) contains subshells determined by the azimuthal (angular momentum) quantum number l, where l = 0, 1, 2, ... , (n - 1).
- Subshells are named s, p, d and f for l = 0, 1, 2, 3 respectively.
- The maximum number of electrons in a subshell is 2(2l + 1). Thus s, p, d and f subshells can hold 2, 6, 10 and 14 electrons respectively.
All possible subshells for n up to 4 are:
Principle Quantum Number Value | Value of Azimuthal Quantum Number | Resulting Subshell in the Electron Configuration |
n = 1 | l = 0 | 1s |
n = 2 | l = 0 | 2s |
l = 1 | 2p | |
n = 3 | l = 0 | 3s |
l = 1 | 3p | |
l = 2 | 3d | |
n = 4 | l = 0 | 4s |
l = 1 | 4p | |
l = 2 | 4d | |
l = 3 | 4f |
Note that subshells such as 1p, 2d or 3f do not exist because the value of l must be less than n.
Notation
- An electron configuration uses subshell labels combining the shell number, subshell letter and a superscript for electrons in that subshell (for example 1s2).
- These labels contain the shell number (given by the principal quantum number), the subshell name (given by the azimuthal quantum number) and the total number of electrons in the subshell in superscript.
- For magnesium (atomic number 12) the configuration is 1s2 2s2 2p6 3s2.
- Noble-gas shorthand can replace a string of filled inner subshells; e.g., magnesium is [Ne] 3s2.
Filling of Atomic Orbitals
Aufbau Principle
- The Aufbau principle (from German for "building up") states that electrons occupy orbitals of lower energy before occupying those of higher energy.
- The energy of an orbital is calculated by the sum of the principal and the azimuthal quantum numbers.
- A common mnemonic for the energy ordering is obtained from the (n + l) rule: orbitals with lower (n + l) are filled first; if two orbitals have the same (n + l) value, the orbital with lower n is filled first.
- A typical filling order used in many textbooks is: 1s → 2s → 2p → 3s → 3p → 4s → 3d → 4p → 5s → 4d → 5p → 6s → 4f → 5d → 6p → 7s → 5f → 6d → 7p ...
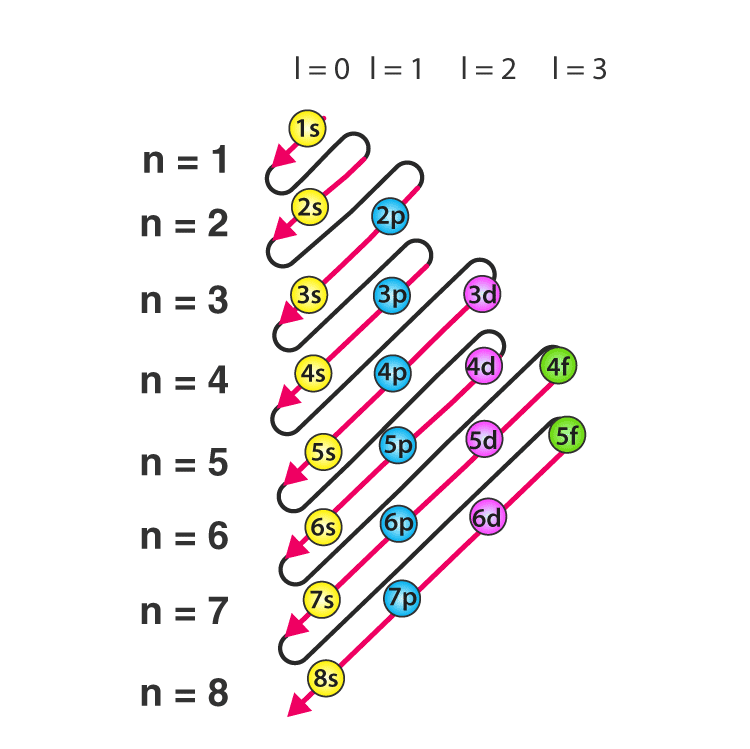
There are known exceptions to this ordering such as chromium and copper, for certain transition and heavier elements; these arise because the relative energies of the ns and (n - 1)d or (n - 2)f orbitals are very close and electronic exchange and electron-electron interactions can stabilise half-filled or fully filled subshells.
Pauli Exclusion Principle
- The Pauli exclusion principle requires that no two electrons in an atom have the same set of all four quantum numbers.
- As a practical rule this means if the principal, azimuthal, and magnetic numbers are the same for two electrons, they must have opposite spins.
Hund's Rule
- Hund's rule governs the filling of several degenerate orbitals (orbitals of the same energy, for example the three 2p orbitals): every orbital in the subshell is singly occupied before any orbital receives a second electron.
- Electrons placed singly in degenerate orbitals have parallel spins so as to maximise the total spin (this arrangement lowers the energy by exchange stabilisation).
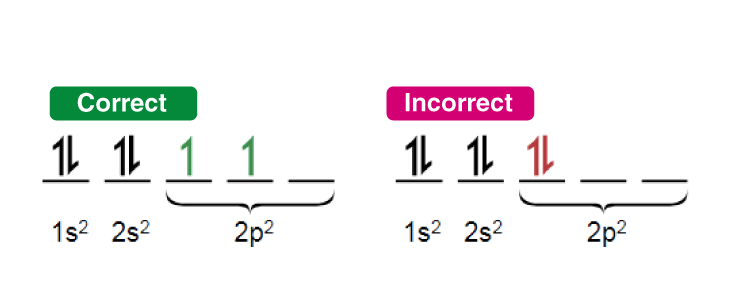
Common exceptions to Aufbau ordering
These exceptions arise due to very small energy differences between ns and (n-1)d orbitals.
- Chromium: expected configuration by simple filling would be [Ar] 3d4 4s2, but actual ground state is [Ar] 3d5 4s1 because a half-filled 3d subshell gives extra stability.
- Copper: expected [Ar] 3d9 4s2, but actual is [Ar] 3d10 4s1 due to stability of a filled d subshell. Similar exceptions occur for Mo, Ag, Au and some heavier elements.
- For ions of transition metals electrons are removed first from the outermost s orbital (ns) and then from the (n - 1)d orbitals, e.g. Fe: [Ar] 3d6 4s2; Fe2+ is [Ar] 3d6 (4s electrons removed).
Examples of Electron Configurations
Hydrogen
The atomic number of hydrogen is 1, so it has one electron which occupies the 1s orbital. The configuration is 1s1.
 Electron Configuration of Hydrogen
Electron Configuration of HydrogenOxygen
Oxygen has atomic number 8 and therefore 8 electrons. These fill as:
- K shell (n = 1): 2 electrons → 1s2
- L shell (n = 2): 6 electrons → 2s2 2p4
Hence oxygen is 1s2 2s2 2p4.
 Electron Configuration of Oxygen
Electron Configuration of OxygenChlorine
Chlorine has atomic number 17. Electrons distribute as:
- K shell: 2 electrons
- L shell: 8 electrons
- M shell: 7 electrons
The full configuration is 1s2 2s2 2p6 3s2 3p5, which can be written in condensed form as [Ne] 3s2 3p5.
 Electron Configuration of Chlorine
Electron Configuration of ChlorineTransition-metal examples (illustrative)
- Chromium: [Ar] 3d5 4s1 (exception - half-filled 3d stabilised).
- Copper: [Ar] 3d10 4s1 (exception - filled 3d stabilised).
- Zinc: [Ar] 3d10 4s2 (3d completely filled).
Electronic Configuration in Periods and Groups
The electronic configuration of an element determines its position in the periodic table and explains many periodic trends. The period number equals the principal quantum number n of the valence shell and elements in the same group have the same number of valence electrons giving them similar chemical properties.
Electronic configuration across periods
- First period (n = 1): filling of 1s. Elements: hydrogen (1s1) and helium (1s2).
- Second period (n = 2): starts with Li and Be filling 2s and then B → Ne fill 2p; total 8 elements (1s2 2s2 2p1-6).
- Third period (n = 3): Na → Ar involve filling 3s and 3p (also 8 elements) although the third shell can accommodate 18 electrons when 3d orbitals are involved in the next period.
- Fourth period (n = 4): starts with 4s filling (K), then 3d transition series begins before 4p completes; scandium is [Ar] 3d1 4s2, zinc completes 3d (3d10 4s2).
- Fifth period (n = 5): starts at 5s, includes 4d transition series and ends with filling 5p (ends at xenon).
- Sixth period (n = 6): involves 6s, then filling of 4f (lanthanides), 5d and 6p. Lanthanide series begins as electrons enter 4f (e.g., cerium).
- Seventh period (n = 7): includes 7s, 5f (actinides), 6d and 7p; many elements are radioactive and several are synthetic.
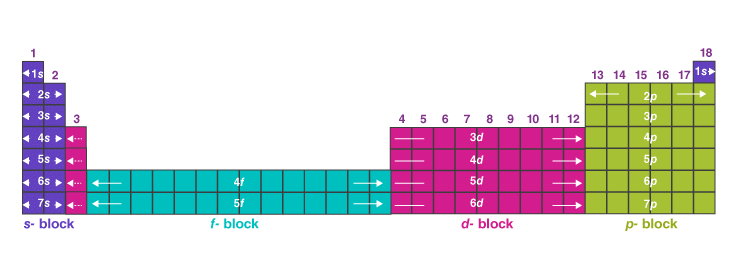
Blocks of the periodic table
- s-block: groups 1-2 (and helium) where valence electrons occupy s orbitals.
- p-block: groups 13-18 where valence electrons occupy p orbitals.
- d-block: transition elements where electrons occupy (n - 1)d orbitals.
- f-block: lanthanides and actinides where electrons occupy f orbitals.
Electronic configuration in groups
Elements in the same group generally have the same outer-shell (valence) electron configuration pattern, for example all group 1 elements end in ns1 and are highly reactive metals; group 17 elements end in ns2 np5 and are halogens.
Valence Electrons, Ions and Valency
- Valence electrons are electrons in the outermost shell and largely determine chemical bonding and reactivity.
- Valency (combining capacity) can often be predicted from valence electrons: main-group elements gain, lose or share electrons to attain a noble-gas configuration.
- When writing configurations for ions, add electrons for anions and remove electrons for cations. For main-group ions this usually affects only the valence shell; for transition-metal cations, ns electrons are removed before (n - 1)d electrons.
- Examples: Na → Na+ is 1s2 2s2 2p6 or [Ne]; Cl → Cl- is 1s2 2s2 2p6 3s2 3p6 or [Ar].
Applications and Consequences of Electron Configuration
- Prediction of chemical properties and typical oxidation states.
- Explanation of periodic trends: atomic and ionic sizes, ionisation enthalpy, electron affinity and electronegativity relate to valence configuration.
- Understanding magnetic behaviour: atoms with unpaired electrons (from configuration) are paramagnetic; those with all electrons paired are diamagnetic.
- Explanation of spectral lines: transitions between electronic energy levels give rise to characteristic emission or absorption spectra.
- Electronic configuration helps predict the most common oxidation states of elements, especially for main-group elements.
Common Exceptions and Practical Notes
- Exceptions to the simple filling order occur primarily among transition and heavier elements because of small energy differences between ns and (n - 1)d orbitals; chromium and copper are classic simple examples.
- For ions of transition metals, remove electrons from the ns orbital before the (n - 1)d orbital when forming cations.
- This rule applies even though the ns orbital is filled before the (n-1)d orbital in neutral atoms.
- This is because after bonding or ionisation, the ns orbital lies at a higher energy than the (n-1)d orbitals.
- Relativistic effects and electron correlation become important for very heavy elements; such effects alter expected configurations but are beyond basic syllabus detail.
Summary
Electron configurations give a concise description of how electrons occupy atomic orbitals and form the basis for understanding the structure of the periodic table and chemical behaviour of elements. The Aufbau principle, Pauli exclusion principle and Hund's rule together determine ground-state electronic arrangements, with some well-known exceptions among transition elements. Mastery of writing and interpreting electron configurations is essential for predicting valency, reactivity, magnetic properties and many periodic trends.
In NEET, questions commonly test correct writing of electronic configurations, identification of exceptions, calculation of unpaired electrons, magnetic behaviour, and prediction of valency.
FAQs on Electronic Configuration of Elements & Periodic Table
| 1. How do I write the electronic configuration of elements correctly using the Aufbau principle? |  |
| 2. What's the difference between Bohr model and quantum mechanical model when explaining electron placement? |  |
| 3. Why do some elements in the periodic table have irregular electronic configurations? |  |
| 4. How does the periodic table arrangement relate to electron configuration patterns? |  |
| 5. Can I quickly memorize electron configurations for NEET exams without writing them all out? |  |






