Structure of Electron Deficient Compounds involving Main Group Elements
What are electron deficient compounds explain with example?
Electrons plays a significant role in the formation of a compound. Electron rich and electron-deficient both have variant roles. Let us know about electron-deficient compounds.
➤ Definition- Electron deficient
An electron-deficient compound is one in which there is an insufficient number of electrons to complete the octet of the central atom. These compounds contain insufficient numbers of electrons to form normal electron-pair bonds between each pair of bonded atoms.
➤ Examples
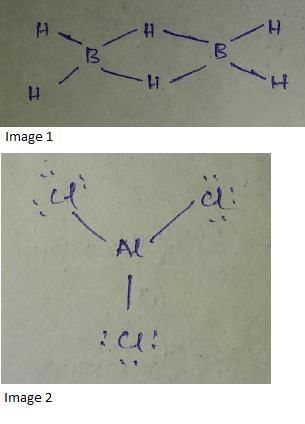
The compounds containing less than 8 electrons in the valence shells are called electron-deficient compounds such as B2F6, Al2Cl6, etc. On the other hand, the compounds containing more than 8 electrons in the valence shells may be called electron surplus compounds example, SF6, O8F8, etc.
➤ Boron family as an example
For example, boron which has electronic configuration [He], 2s22p1 forms compounds which are described as electron-deficient compounds.
- Boron has only three valence electrons but has four available orbitals and to accommodate these electrons.
- Other members of the boron family namely aluminium, gallium, indium and thallium show a tendency to lose their valence electrons to form ionic M3+ species in their compounds.
Try yourself: Which of the following pair has electron deficient compounds?
FAQs on Structure of Electron Deficient Compounds involving Main Group Elements
| 1. What are electron deficient compounds involving main group elements? |  |
| 2. How do electron deficient compounds form? |  |
| 3. What are some examples of electron deficient compounds involving main group elements? |  |
| 4. What are the properties of electron deficient compounds? |  |
| 5. What are the applications of electron deficient compounds involving main group elements? |  |