Chapter Notes: Solids, Liquids & Gases
What Is Matter?
Matter is everything around us that has mass and takes up space. It is what things are made of. You, your toys, the air, water and the food you eat are all examples of matter. Anything that you can touch, feel or see is made of matter.
Look around your classroom. Tick the things you see:
- Plant
- Chair
- Desk
- Blackboard
- Chalk
- Water
- Lizard
- Book
All the things above have two important features in common:
- They take up space.
- They have mass.
The space taken up by an object is called its volume. The amount of matter contained in an object is its mass. Anything that has mass and takes up space is called matter. Thus people, animals, plants, water, chairs, aeroplanes, kites and all other things around us are matter.
Does air matter? Yes. Air has mass and it takes up space, so air is also matter.
Try yourself: Which of the following is an example of matter?
What Is Matter Made Up of?
All matter is made up of very tiny particles called molecules. Molecules themselves are made of still smaller particles called atoms. The molecules of every substance are different and behave in different ways.

The States of Matter
Matter around us can be found mainly in three states: solid, liquid and gas. For example, a brick is a solid, water is a liquid and air is a gas. These three states have different properties and the arrangement of their molecules is different. The space between the molecules, called the intermolecular space, also varies in the three states.
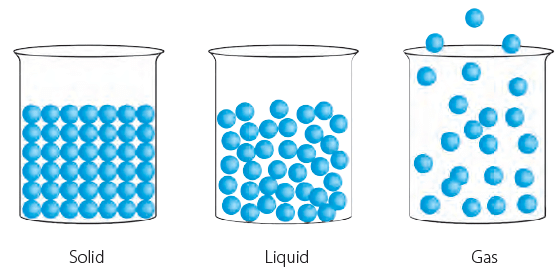 Intermolecular spaces in solids, liquids and gases
Intermolecular spaces in solids, liquids and gasesThere are three common states of matter.
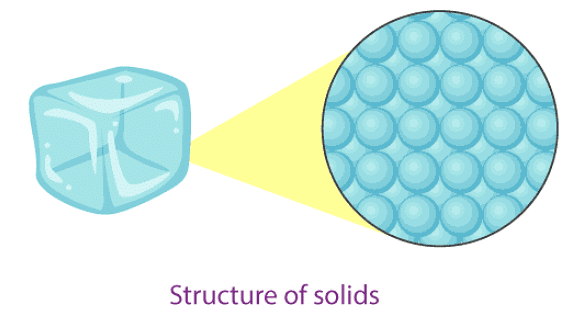
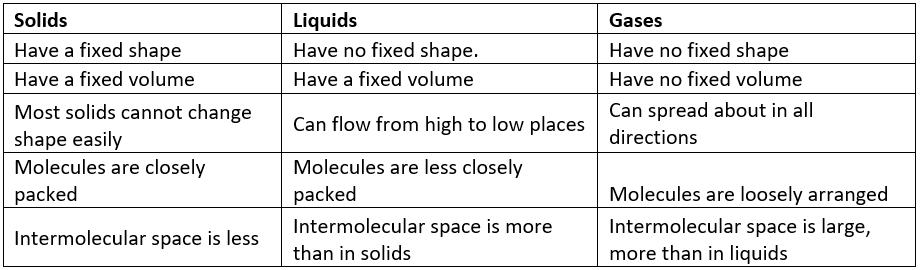
Solids
Examples of solids: a pencil, a slice of bread, a piece of wood and a pair of shoes.

Properties of solids
- In solids, particles are tightly packed together.
- The gaps between particles are very small, so solids are hard to compress.
- Solids have a fixed shape and a fixed volume.
- Particles in a solid can only vibrate about their mean positions; they cannot move freely.
- The force of attraction between particles in a solid is strong.
- The rate of diffusion in solids is very low (they do not spread quickly).
- Examples: ice (solid), sugar, rock, wood, etc.
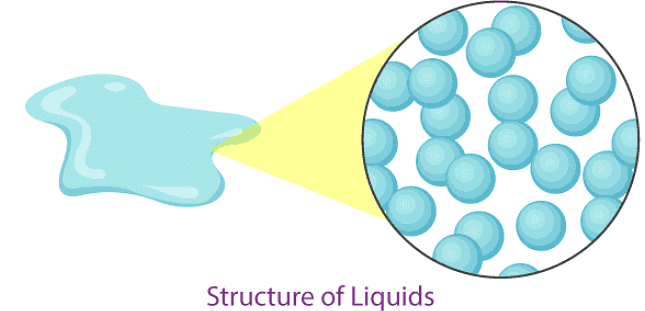
Liquids
Examples of liquids: water, milk, blood, coffee, etc.
- In liquids, particles are less tightly packed than in solids.
- Liquids take the shape of the container in which they are kept.
- Liquids are difficult to compress because particles still have little space between them.
- Liquids have a fixed volume but no fixed shape.
- The rate of diffusion in liquids is higher than in solids (liquids spread more readily).
- The force of attraction between particles in a liquid is weaker than in a solid.
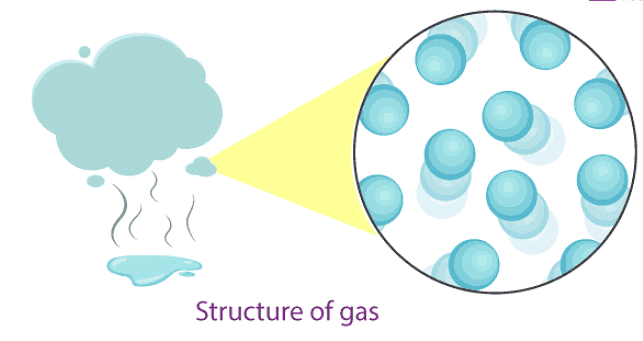
Gases
Examples of gases: air, helium, nitrogen, oxygen, carbon dioxide, etc.
- In gases, particles are far apart from each other.
- The force of attraction between particles is negligible, so particles move freely.
- Gases have neither a fixed volume nor a fixed shape.
- Gases are highly compressible - they can be squeezed into a smaller volume more easily than solids or liquids.
- The rate of diffusion in gases is higher than in solids and liquids (gases spread fastest).
- The kinetic energy of particles in gases is higher than in liquids and solids.
Change of States of Matter
A substance can change from one state to another when heating or cooling changes the energy of its particles. For example, water is a liquid but it can become a solid (ice) or a gas (water vapour). In each change, the arrangement and movement of molecules change.
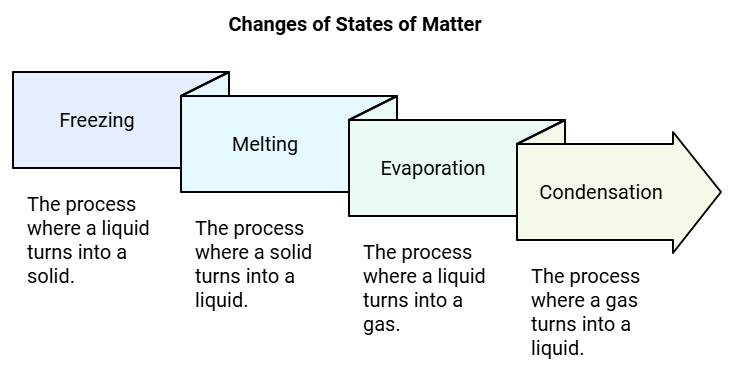
Freezing
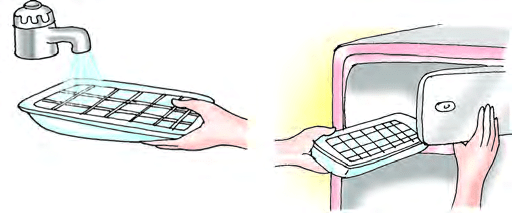
Freezing is the change from a liquid to a solid when a substance is cooled. For example, when you keep water in a freezer for a few hours, it changes into ice.
 Freezing
FreezingMelting
Melting is the change from a solid to a liquid when a substance is heated. For example, if you keep ice cubes in a pan and heat them gently, the ice melts and becomes water.
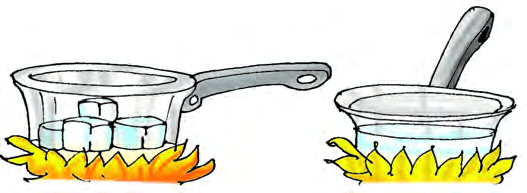 Melting
MeltingEvaporation
Evaporation is the change of a liquid into its vapour (gas) form when it is heated. For instance, when water is heated in a kettle, after some time it boils and steam (water vapour) rises from it.
 When water is boiled in a kettle, steam or gas appears.
When water is boiled in a kettle, steam or gas appears.Condensation
- Condensation happens when a gas cools down and changes back into a liquid.
- For example, steam from boiling water can turn into liquid water when it touches a cold surface.
- To see condensation, place a cold steel plate over the steam from a boiling kettle. The steam will touch the cold plate, cool down and become water droplets on the plate.
- We can see similar state changes in materials like butter and wax, which melt on heating and solidify on cooling.
 Condensation
Condensation Melting of butter
Melting of butterSoluble Substances
- Certain solids dissolve in water; these are called soluble substances.
- When you add a spoonful of sugar to water and stir, the sugar seems to disappear because it dissolves in the water.
- This means sugar is soluble in water. Other soluble substances include salt, soap powder and some fruit juices.
- When a solid dissolves, its tiny particles mix and occupy the spaces between the water molecules.
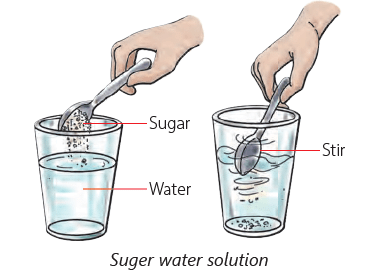
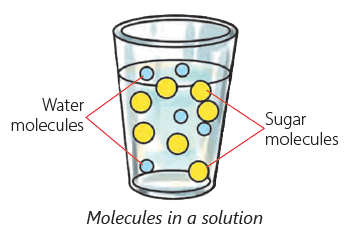
Solute, Solvent and Solution
- The substance that dissolves in a liquid is called the solute. Examples of solutes are salt and sugar.
- The liquid in which the solute dissolves is called the solvent. Water is a common solvent.
- A solution is the mixture formed when a solute dissolves in a solvent.
- We can write this as: Solvent + Solute = Solution
- Water is often called a universal solvent because it can dissolve many different substances such as salt, sugar and coffee.
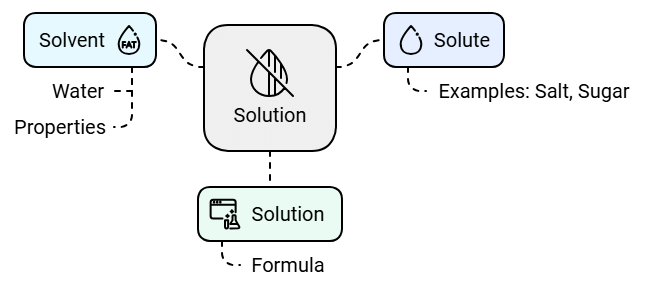
Edurev Tips:
Petrol and kerosene are also solvents. Kerosene is used to dissolve oil paint whereas petrol is used to dissolve grease.
Insoluble Substances
Some solids do not dissolve in water; these are called insoluble substances. For example, when making tea we mix water, milk, sugar and tea leaves. The sugar dissolves in the tea, but tea leaves do not. So, sugar is soluble while tea leaves are insoluble. We can separate insoluble solids from liquids using simple methods.
Filtration
Filtration is the method used to separate insoluble solids from a liquid. For example, to remove tea leaves from tea we pour the tea through a sieve or a strainer; the tea passes through while the leaves remain in the sieve.
 Filtration
FiltrationSedimentation and Decantation
Mix some sand in a glass of water. Does it dissolve? No - sand is insoluble in water. If you leave the sand-water mixture undisturbed, the sand particles settle at the bottom. This process is called sedimentation. After sedimentation, you can carefully pour the clear water into another container without disturbing the sand at the bottom. This process of pouring off the clear liquid is called decantation.
 Separating a mixture of sand and water using sedimentation and decantation
Separating a mixture of sand and water using sedimentation and decantationSummary
Matter is anything that has mass and occupies space. Matter is made of atoms and molecules and commonly exists as solids, liquids and gases. Substances can change state by heating or cooling (melting, freezing, evaporation, condensation). Some solids dissolve in water (soluble) while others do not (insoluble); solute, solvent and solution are important words to describe dissolving. Simple methods such as filtration, sedimentation and decantation help separate mixtures.
FAQs on Chapter Notes: Solids, Liquids & Gases
| 1. What's the difference between solids, liquids, and gases in science for Class 4? |  |
| 2. Why do gases spread out in all directions while solids stay in one place? |  |
| 3. How can you tell if something is a liquid or a gas just by looking at it? |  |
| 4. What happens to the shape and volume of water when you pour it into different containers? |  |
| 5. Can solids, liquids, and gases be examples of the same material, or are they always different things? |  |







