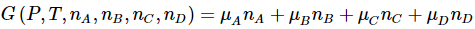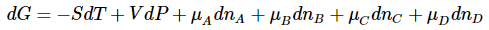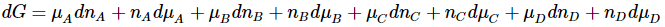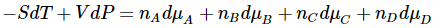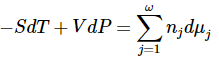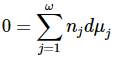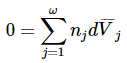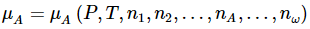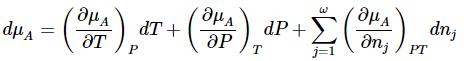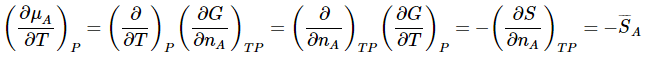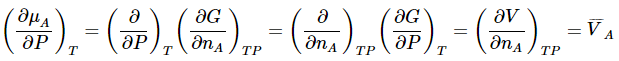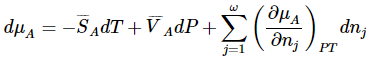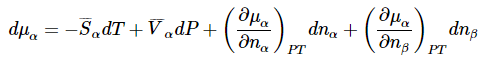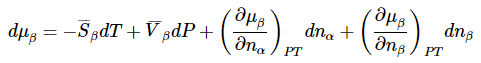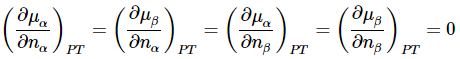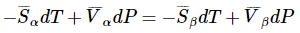Gibbs-Duhem Equation
An important relationship among the differentials of the chemical potentials for a system follows from the relationships we have just developed. From the fact that the Gibbs free energy, G(P,T,nA,nB,nC,nD) , is homogeneous of order one in the composition variables, we find that the Gibbs free energy of the system is related to its partial molar derivatives by
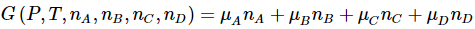
The differential of the left hand side is
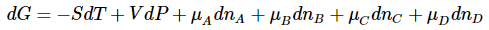
and the differential of the right hand side is
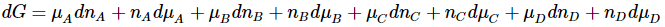
Since these differential expressions must be equal, we have
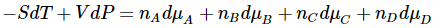
for any change in this system.
While we have considered the particular case of a system containing the species A, B, C, and D, it is clear that the same arguments apply to any system. For a system that contains ω species, we can write the result in general form as
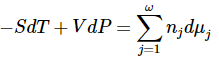
This relationship is known as the chemical-potential Gibbs-Duhem equation. It is a constraint on the dμj that must be satisfied when any change occurs in a system whose thermodynamic functions are continuous functions of its composition variables. If pressure and temperature are constant and this equation is satisfied, the system is at equilibrium-it is on a Gibbsian equilibrium manifold-and the chemical-potential Gibbs-Duhem equation becomes
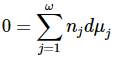
In the next two sections, we develop a particularly useful expression for dμj . We can obtain similar relationships for other partial molar quantities. These relationships are also called Gibbs-Duhem equations. Because the derivation requires only that the thermodynamic function be homogeneous of order one, the same relationships exist among the differentials of the partial molar derivatives of any extensive thermodynamic function. For partial molar volumes at constant pressure and temperature, we find
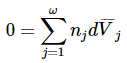
Dependence of Chemical Potential on Other Variables
The chemical potential of a substance in a particular system is a function of all of the variables that affect the Gibbs free energy of the system. For component A , we can express this by writing
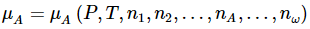
for which the total differential is
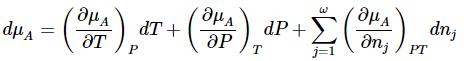
Recalling the definition of the chemical potential and the fact that the mixed second-partial derivatives of a state function are equal, we have
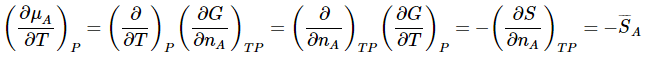
Similarly,
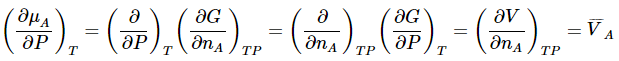
Thus, the total differential of the chemical potential for species A can be written as
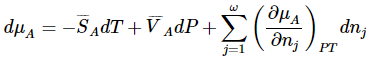
To illustrate the utility of this result, we can use it to derive the Clapeyron equation for equilibrium between two phases of a pure substance. We can now use the total differential of the chemical potential to present essentially the same derivation using a simpler argument. Letting the two phases be α and β, the total differentials for a system that contains both phases becomes
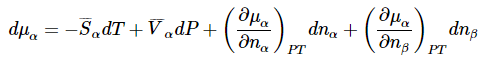
and
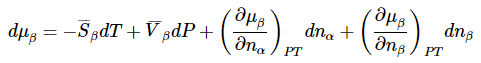
Since equilibrium between phases α and β means that μα=μβ, we have also that dμα=dμβ for any process in which the phase equilibrium is maintained. Moreover, α and β are pure phases, so that μα and μβ are independent of nα and nβ. Then
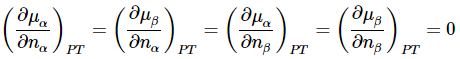
Hence,
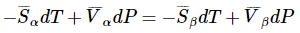
and the rest of the derivation follows as before.