Worksheet Solutions: Solids, Liquids & Gases - 1
Q1: Multiple Choice Questions (MCQs).
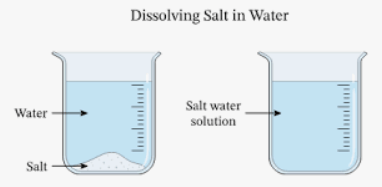
(i) The mixture of salt and water is called(a) solute
(b) solvent
(c) solution
(d) matter
Ans: (c)
Explanation: A solution is a uniform mixture of two or more substances. In a salt-water mixture, salt is the solute (the substance that dissolves) and water is the solvent (the substance that does the dissolving). The salt particles spread evenly among the water molecules, forming a clear mixture called a solution.
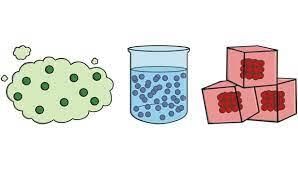
(ii) Which of these has a fixed shape and volume?
(a) Solid
(b) Liquid
(c) Gas
(d) All of these
Ans: (a)
Explanation: Solids have a fixed shape and a fixed volume because their particles are packed closely and only vibrate in place. Liquids do not have a fixed shape (they take the shape of their container) and gases have neither fixed shape nor fixed volume.
(iii) Which of the following is not always true for matter?
(a) Has weight
(b) Takes up space
(c) Can be seen
(d) Is made up of molecules
Ans: (c)
Explanation: Saying that matter "can be seen" is not always true. Some forms of matter, such as many gases, are invisible to our eyes even though they have weight, take up space and are made of molecules. Therefore "can be seen" is not always correct for matter.
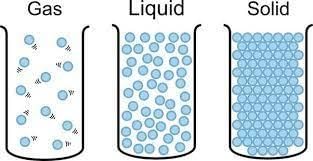
(iv) The distance between the molecules of which of these is the highest?
(a) Solid
(b) Liquid
(c) Gas
(d) Same in all of these
Ans: (c)
Explanation: In gases the particles are far apart and move freely; therefore the distance between molecules in a gas is much larger than in liquids or solids. Solids have the least intermolecular space, liquids are in between, and gases have the most.
Q2: True & False.
(i) Ice cube melts into water in freezing.
Ans: False
Explanation: Freezing is the change of a liquid into a solid when it becomes colder. Melting is the opposite process: a solid (like an ice cube) becomes a liquid when it warms up. So an ice cube melts during heating, not during freezing.

(ii) Insoluble substances can be separated from liquid by sedimentation only.
Ans: False
Explanation: Sedimentation (settling down) can separate large or heavy insoluble particles from a liquid, but it is not always enough. Often we use filtration or decantation after sedimentation to fully separate insoluble substances from the liquid.
(iii) Both solids and liquids have definite shapes.
Ans: False
Explanation: Solids have definite shapes because their particles are fixed in position. Liquids do not have a definite shape; they take the shape of the container that holds them, although they do have a definite volume.
(iv) In evaporation process, a liquid changes into solid.
Ans: False
Explanation: Evaporation is the process in which a liquid changes into a gas when it gains heat. A liquid does not become a solid during evaporation; becoming a solid is the result of freezing.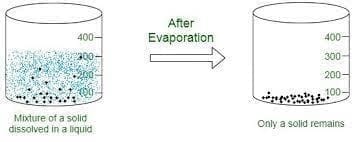
Q3: Fill in the blanks
(i) Intermolecular space is least in case of _____.Ans: Solids
In solids, the particles are packed very close together and there is very little space between them.The strong forces of attraction between particles in solids hold them in a fixed position, that is why solids keep their own shape and have a definite volume.
(ii) Liquids do not have definite_____.
Ans: Shape
Liquids have no fixed shape; they flow and take the shape of the container that holds them due to the relatively weak forces of attraction between their particles . However, liquids do have a definite volume because their particles remain close to each other.
(iii) Sand is _____ in water.
Ans: Insoluble
When we say that sand is insoluble in water, it means that sand does not readily dissolve in water. Sand consists of larger solid particles, and when mixed with water, it does not undergo a chemical reaction to form a solution. Instead, sand particles tend to settle down in water due to their insolubility, a process called sedimentation. This makes it possible to separate sand from water by allowing it to settle and then pouring off the water.
Q4: Match the following.

Ans: 1. Change of solid state into liquid - Melting
Melting is the process when a solid becomes a liquid on heating. For example, ice melts to become water.
2. Sugar in lemonade - solute
In lemonade, sugar is the solute because it dissolves in water (the solvent) to make a uniform mixture called a solution.
3. Change of liquid state into solid - Freezing
Freezing happens when a liquid becomes a solid on cooling. For example, water freezes to form ice.
4. Change of vapour into liquid - condensation
Condensation is when gas or vapour cools and changes into liquid droplets, such as tiny drops on a cold glass or on a window.
5. Conversation of liquid into vapour - Evaporation
Evaporation is the change of a liquid into a gas, usually at the surface of the liquid when it absorbs heat. Boiling is a faster form of vaporisation that happens throughout the liquid.
Q5: Answer the following questions in brief.
(i) What do you understand by the term freezing?Ans: Freezing is the change of a liquid into a solid when its temperature is lowered below its freezing point. For example, water becomes ice when it is cooled enough.
(ii) What is matter?
Ans: Matter is anything that has weight and takes up space. All living things (like animals and plants) and non-living things (like chairs, water and air) are made of matter.
(iii) What is condensation?
Ans: Condensation is the process in which water vapour (gas) cools and changes into tiny liquid drops. This is what forms dew on grass and drops of water on a cold glass.
(iv) Define the following:
(a) Solution
Ans: A solution is a uniform mixture of two or more substances in which the particles are very small( smaller than 1 nm) and spread evenly. Examples are sugar dissolved in water or salt dissolved in water.
(b) Solvent
Ans: A solvent is the substance that dissolves another substance (the solute). In many common solutions, water is the solvent because it dissolves many things.
(c) Solute
Ans: A solute is the substance that gets dissolved in a solution. For example, sugar is the solute when it dissolves in water.
(v) Write any two properties of solids.
Ans:
- A solid has a definite shape and a definite volume.
- Solids generally have higher density because their particles are closely packed.
- In solids, the forces between particles are strong, keeping them in fixed positions.
- Diffusion (mixing by motion of particles) in solids is extremely slow compared with liquids and gases.
FAQs on Worksheet Solutions: Solids, Liquids & Gases - 1
| 1. What are the three states of matter? |  |
| 2. How do solids, liquids, and gases differ from each other? |  |
| 3. Can a substance change from one state of matter to another? |  |
| 4. What causes the changes in state of matter? |  |
| 5. What are some examples of substances in each state of matter? |  |





