Class 10 Science: CBSE Sample Question Paper- Term I (2021-22) - 1
Class-X
Time: 90 Minutes
M.M: 40
General Instructions:
Read the following instructions very carefully and strictly follow them:
- The Question Paper contains three sections.
- Section A has 24 questions. Attempt any 20 questions.
- Section B has 24 questions. Attempt any 20 questions.
- Section C has 12 questions. Attempt any 10 questions.
- All questions carry equal marks.
- There is no negative marking.
Section - A
Q.1: Choose the correct statement about the reaction taking place below:
6CO2 + 6H2O → C6H12O6 + 6O2
(a) It is an endothermic reaction.
(b) It is an exothermic reaction.
(c) It releases large amount of energy.
(d) All of these
Ans: (a)
Explanation: This equation represents photosynthesis. In photosynthesis plants absorb light energy from the Sun and use it to convert carbon dioxide and water into glucose and oxygen. Because energy is absorbed and stored in the chemical bonds of glucose, the process is endothermic. It does not release energy; instead light energy is taken in and stored.
Q.2: Study the given equation: Na2CO3 + XHCl → 2NaCl + CO2 + H2O
What will be the value of X to make it balanced equation?
(a) 1
(b) 2
(c) 3
(d) 4
Ans: (b)
Explanation: Balance hydrogen and chlorine by using two molecules of HCl. The balanced equation is:
Na2CO3 + 2HCl → 2NaCl + CO2 + H2O.
Q.3: For a chemical reaction to take place:
(a) It must be accompanied with change in temperature and pressure.
(b) At least one of the reactants must be in a fixed quantity.
(c) It must follow the law of conservation of mass.
(d) All of the above
Ans: (c)
Explanation: Every chemical reaction must obey the law of conservation of mass. This means the total mass of reactants equals the total mass of products. Changes in temperature or pressure may occur in some reactions, but they are not necessary conditions for a reaction to take place; similarly, there is no requirement that a reactant be in a fixed quantity.
Q.4: The decomposition of silver chloride and silver bromide are carried out in the presence of:
(a) Electricity
(b) Sunlight
(c) Heat
(d) Water
Ans: (b)
Explanation: Silver chloride (AgCl) and silver bromide (AgBr) undergo photochemical decomposition; they break down when exposed to light. The light provides the energy needed to decompose these halides into silver and halogen, so sunlight (or another strong light source) is used.
Q.5: Choose the correct statement about the given reaction.
H2S + Cl2 → 2HCl + S
(i) Cl2 gets reduced to HCl.
(ii) H2S gets oxidized to S.
(iii) Cl2 is the oxidizing agent.
(iv) H2S is the oxidizing agent.
(a) I and II only
(b) II and III only
(c) I, II and III only
(d) All of these
Ans: (c)
Explanation: In the reaction Cl2 gains electrons to form HCl (reduction), while H2S loses electrons to form elemental sulfur (oxidation). Therefore Cl2 is the oxidizing agent (it causes oxidation of H2S), and H2S acts as the reducing agent. Statements (i), (ii) and (iii) are correct; (iv) is incorrect.
Q.6: The aqueous solution of sodium acetate is
(a) Neutral
(b) Weakly Acidic
(c) Strongly Acidic
(d) Alkaline
Ans: (d)
Explanation: Sodium acetate is the salt of a weak acid (acetic acid) and a strong base (sodium hydroxide). In water, the acetate ion hydrolyses to produce OH-, making the solution basic (alkaline).
Q.7: Which of the following is a mineral acid?
(a) HCl
(b) H2SO4
(c) HNO3
(d) All of these
Ans: (d)
Explanation: Mineral acids are acids obtained from minerals of the earth. Hydrochloric acid (HCl), sulphuric acid (H2SO4) and nitric acid (HNO3) are all examples of mineral acids, so option (d) is correct.
Q.8: A solution "X" gives pink colour with phenolphthalein. The pH is likely to be:
(a) 1
(b) 5
(c) 7
(d) 12
Ans: (d)
Explanation: Phenolphthalein is colourless in acidic and neutral solutions and turns pink in basic solutions (pH roughly above 8.2). A pink colour therefore indicates an alkaline solution; among the options, pH 12 is the only basic value given.
Q.9: In an experiment, Sonia found that an excess of dilute sulphuric acid reacts with both aqueous barium hydroxide and aqueous barium chloride respectively. In what way are the two reactions the same?
(a) A gas is evolved.
(b) An insoluble salt is formed
(c) Their final pH is 7
(d) A base is produced
Ans: (b)
Explanation: In both reactions a white insoluble precipitate of barium sulphate (BaSO4) is formed. The reactions are:
Ba(OH)2 + H2SO4 → BaSO4 + 2H2O
BaCl2 + H2SO4 → BaSO4 + 2HCl
In each case an insoluble salt (BaSO4) precipitates.
Q.10: Choose the correct equation for the metal which reacts vigorously with water.
(a) Ca(s) + 2H2O (l) → Ca(OH)2 (aq) + H2 (g)
(b) 2Na (s) + 2H2O (l) → 2NaOH (aq) + H2 (g)
(c) 2Al(s) + 3H2O(g) → Al2O3(s) + 3H2 (g)
(d) Mg(s)+ 2H2O(g) → Mg(OH) (aq)+ H2 (g)
Ans: (b)
Explanation: Alkali metals such as sodium react vigorously with water producing the hydroxide and hydrogen gas, often with enough heat to ignite the hydrogen. The correct balanced equation for sodium is:
2Na(s) + 2H2O(l) → 2NaOH(aq) + H2(g) (+ heat)
Q.11: The end product of fermentation in muscle cells is
(a) Lactic Acid
(b) Hydrochloric Acid
(c) Citric Acid
(d) Pyruvic Acid
Ans: (a)
Explanation: Under low oxygen conditions in muscle cells, pyruvate produced by glycolysis is converted into lactic acid. This allows some ATP to be produced anaerobically but leads to lactic acid accumulation, which can cause muscle fatigue and pain.
Q.12: Which of these statements is correct regarding the given figure of a heart.
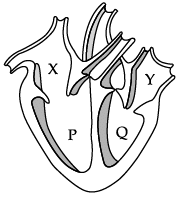
(a) The blood from Y reaches the lungs.
(b) The blood from the body enters the heart through P.
(c) Y receives blood from the lungs.
(d) Q receives blood from the body.
Ans: (c)
Explanation: In the labelled figure, Y is the left atrium, which receives oxygenated blood from the lungs. X (right atrium) receives deoxygenated blood from the body; P is the right ventricle (pumps to lungs); Q is the left ventricle (pumps oxygenated blood to the body).
Q.13: Haemoglobin has a maximum affinity for which of the following gases?
(a) Carbon Monoxide
(b) Carbon Dioxide
(c) Oxygen
(d) Nitrogen
Ans: (a)
Explanation: Haemoglobin binds carbon monoxide (CO) much more strongly than it binds oxygen - roughly 200 times stronger. CO forms carboxyhaemoglobin with haemoglobin, greatly reducing haemoglobin's capacity to carry oxygen.
Q.14: The metal __________ is associated with haemoglobin.
(a) Copper
(b) Iron
(c) Aluminium
(d) Potassium
Ans: (b)
Explanation: Iron is the central metal ion in haemoglobin. It binds oxygen in the lungs and helps transport it to tissues. A large fraction of the body's iron is present as haemoglobin in red blood cells and as myoglobin in muscles.
Q.15: Which of these statements is correct about transpiration?
(i) It causes absorption of mineral salts.
(ii) It regulates plant temperature.
(iii) It causes wilting and injury in plants.
(iv) It causes an ascent of sap.
(a) I and IV only
(b) I, II and III only
(c) I, II and IV only
(d) All of these
Ans: (d)
Explanation: Transpiration helps draw water and dissolved mineral salts upward from the roots (ascent of sap) and thus aids absorption. It cools the plant by evaporation, helping regulate temperature. Excessive transpiration, especially when water uptake cannot keep up, can cause wilting and damage. All four statements are correct.
Q.16: Which of these represent the correct pathway of carbon dioxide after the exchange of gases in the lungs?
(a) Alveolus → bronchiole → bronchus → trachea → nose
(b) Alveolus → bronchus → bronchiole → trachea → nose
(c) Trachea → bronchus → bronchiole → alveolus → nose
(d) Trachea → bronchiole → bronchus → alveolus → nose
Ans: (a)
Explanation: After gas exchange at the alveoli, the air (containing carbon dioxide) moves out along the same pathway it came in: from alveolus → bronchiole → bronchus → trachea → nose (or mouth). Option (a) shows this correct outward path.
Q.17: _____ mirrors can be used to burn a piece of paper using the reflected rays of the sun.
(a) A plane mirror
(b) A concave mirror
(c) A convex mirror
(d) All of the above
Ans: (b)
Explanation: A concave mirror converges parallel incoming rays (such as sunlight) to its focal point. If the paper is placed at the focus, the concentrated energy can produce enough heat to burn it. Plane or convex mirrors do not concentrate light in this way.
Q.18: What is the image formed when a convergent beam of light is incident on a plane mirror?
(a) Upright and Real
(b) Upright and Virtual
(c) Inverted and Virtual
(d) Inverted and Real
Ans: (b)
Explanation: A plane mirror always forms a virtual image that is upright (erect) relative to the object. Even if the incident rays are converging, the reflected rays from a plane mirror appear to come from a point behind the mirror, producing an upright virtual image.
Q.19: Read the given statements carefully.
(i) A light wave enters from the first medium into the second medium.
(ii) Its velocity in the second medium is double that in the first medium.
For the phenomenon of total internal reflection to take place, the angle of incidence must be greater than a certain value. This value is _________.
(a) 90°
(b) 60°
(c) 45°
(d) 30°
Ans: (d)
Explanation: Given the speed in medium 2 is twice that in medium 1, the refractive index μ = v1/v2 = 1/2. The critical angle C satisfies sin C = v2/v1 (or sin C = 1/μ). Therefore sin C = 1/2 ⇒ C = 30°.
Q.20: Under which of these cases, you will find an inverted image in a convex mirror?
(a) Under no circumstances
(b) When the object is very far from the mirror
(c) When the object is at a distance equal to the radius of curvature of the mirror
(d) When the distance of the object from the mirror is equal to the focal length of the mirror
Ans: (a)
Explanation: A convex mirror always forms a virtual, erect and diminished image, regardless of object distance. It never produces an inverted image. Only concave mirrors can produce inverted images under suitable object positions.
Q.21: When an object is at infinity from a concave mirror, then the image formed is __________.
(i) At the focus
(ii) Virtual and erect
(iii) Highly enlarged
(iv). Real and inverted
(a) I and IV only
(b) I, and II only
(c) I, II, III only
(d) I, III, IV only
Ans: (a)
Explanation: For a concave mirror when the object is at infinity, parallel rays are focused at the focal point. The image is formed at the focus and is real and inverted. It is highly diminished (not enlarged). So statements (i) and (iv) are correct.
Q.22: A converging lens has a focal length of 15 cm. If an object is placed 10 cm from the optical centre of the lens, the characteristics of the image formed will be:
(a) Real, inverted and diminished
(b) Real, inverted and of the same size as the object
(c) Real, inverted and enlarged
(d) Virtual, upright and enlarged
Ans: (d)
Explanation: Use the lens formula: 1/v - 1/u = 1/f (with sign convention: object u = -10 cm, f = +15 cm).
1/v = 1/f + 1/u = 1/15 + 1/(-10) = (2 - 3)/30 = -1/30 ⇒ v = -30 cm (negative v indicates image is on the same side as the object). A negative image distance for a converging lens means the image is virtual and erect. Magnification m = -v/u = -(-30)/(-10) = +3 → image is upright and enlarged. Hence the image is virtual, upright and enlarged.
Q.23: A real and inverted image of the same size is formed by a convex lens, when an object is placed ___________.
(a) At infinity
(b) At the focus of the lens
(c) At twice its focal length
(d) Between f and the lens
Ans: (c)
Explanation: For a convex (converging) lens, when the object is placed at 2f (twice the focal length), the image is real, inverted and of the same size as the object, formed at 2f on the other side.
Q.24: What is the frequency of yellow light of wavelength 550 nm, if the speed of light is 3 × 108 m/s?
(a) 5.4 × 1014 Hz
(b) 3.4 × 1012 Hz
(c) 3.0 × 1014 Hz
(d) 2.8 × 1012 Hz
Ans: (a)
Explanation: Frequency ν = speed / wavelength = (3 × 108 m/s) / (550 × 10-9 m) = 5.454... × 1014 Hz. Rounding to two significant figures gives 5.4 × 1014 Hz.
Section-B
Q.25: Marble's popularity began in ancient Rome and Greece, where white and off-white marble were used to construct a variety of structures, from hand-held sculptures to massive pillars and buildings.

The substance not likely to contain CaCO3 is
(a) Dolomite
(b) A marble statue
(c) Calcined Gypsum
(d) Sea Shells.
Ans: (c)
Explanation: Gypsum has the formula CaSO4·2H2O (calcium sulphate dihydrate). Calcined gypsum is heated gypsum and still based on CaSO4, so it does not contain calcium carbonate (CaCO3). Dolomite, marble and sea shells do contain carbonate minerals.
Q.26: A student added 10g of calcium carbonate in a rigid container, secured it tightly and started to heat it. After some time, an increase in pressure was observed, the pressure reading was then noted at intervals of 5 minutes and plotted against time, in a graph as shown below. During which time interval did maximum decomposition take place?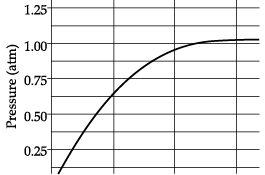
(b) 10-15 min
(c) 5-10 min
(d) 0-5 min
Ans: (d)
Explanation: Decomposition of CaCO3 produces CO2, which increases pressure. The maximum rate of change corresponds to the steepest rise on the pressure-time graph. From the graph the steepest increase occurs in the 0-5 minute interval, so maximum decomposition took place then.
Q.27: Gas A, obtained above is a reactant for a very important biochemical process that occurs in the presence of sunlight. Identify the name of the process -
(a) Respiration
(b) Photosynthesis
(c) Transpiration
(d) Photolysis
Ans: (b)
Explanation: Heating calcium carbonate gives calcium oxide and carbon dioxide:
CaCO3 → CaO + CO2.
The gas evolved is carbon dioxide, which is used by plants in the process of photosynthesis that requires sunlight.
Q.28: Marble statues are corroded or stained rainwater. Identify the main reason.

(a) Decomposition of calcium carbonate to calcium oxide
(b) Polluted water is basic in nature hence it reacts with calcium carbonate
(c) Polluted water is acidic in nature hence it reacts with calcium carbonate
(d) Calcium carbonate dissolves in water to give calcium hydroxide.
Ans: (c)
Explanation: Marble is mainly calcium carbonate (CaCO3). Polluted rainwater often contains dissolved oxides such as SO2 or NO2, which form acids (sulphuric, nitric) when dissolved in water. Acidic rain reacts with calcium carbonate to form soluble calcium salts, CO2 and water, causing corrosion and staining of marble.
Q.29: Calcium oxide can be reduced to calcium, by heating with sodium metal. Which compound would act as an oxidizing agent in the above process?
(a) Sodium
(b) Sodium oxide
(c) Calcium
(d) Calcium oxide
Ans: (d)
Explanation: The oxidizing agent is the species that gets reduced. In this process CaO is reduced to calcium metal (CaO → Ca), so CaO itself is the oxidizing agent because it accepts electrons (loses oxygen) and is reduced.
Q.30: Sodium hydrogen carbonate when added to acetic acid evolves a gas. Which of the following statements are true about the gas evolved?
(i) It turns lime water milky
(ii) It extinguishes a burning splinter
(iii) It dissolves in a solution of sodium hydroxide
(iv) It has a pungent odour
(a) (i) and (ii)
(b) (i), (ii) and (iii)
(c) (ii), (iii) and (iv)
(d) (i) and (iv)
Ans: (b)
Explanation: Sodium hydrogen carbonate (bicarbonate) reacts with acetic acid to produce carbon dioxide (CO2), water and sodium acetate. Carbon dioxide turns lime water milky, extinguishes a burning splinter, and reacts with/ dissolves in solutions of bases such as sodium hydroxide to form carbonate/bicarbonate species. It does not have a pungent odour.
Q.31: Directions: The consist of two statements. Assertion (A) and Reason (R). Answer these questions selecting the appropriate option given below:
Assertion: When an electric current is passed through water, it decomposes to give oxygen and hydrogen. This type of reaction is called electrolysis.
Reason : This reaction involves the use of electricity for the decomposition of the reactant molecules.
(a) Both A and R are true and R is the correct explanation of A.
(b) Both A and R are true but R is NOT the correct explanation of A.
(c) A is true but R is false.
(d) A is false and R is true.
Ans: (a)
Explanation:
(i) Assertion: When electric current is passed through water, it decomposes into hydrogen and oxygen - this is called electrolysis. (True.)
(ii) Reason: Electrolysis involves using electricity to decompose substances into their components. (True.)
(iii) Justification: The reason correctly explains the assertion because passage of current causes the decomposition of water into H2 and O2 at the electrodes; hence R is the correct explanation of A.
Q.32: Directions: The consist of two statements. Assertion (A) and Reason (R). Answer these questions selecting the appropriate option given below:
Assertion: An aqueous solution of acid conducts electricity.
Reason: An acid in the aqueous solution ionizes to produce H+ ions and the corresponding negative ions.
(a) Both A and R are true and R is the correct explanation of A.
(b) Both A and R are true but R is NOT the correct explanation of A.
(c) A is true but R is false.
(d) A is false and R is true.
Ans: (a)
Explanation:
(i) Assertion: An aqueous solution of an acid conducts electricity. (True.)
(ii) Reason: Acids ionise in water to produce H+ and the corresponding anion (e.g., HCl → H+ + Cl-). (True.)
(iii) Justification: The presence of mobile ions in the solution allows it to conduct electricity, so R correctly explains A.
Q.33: Directions: The consist of two statements. Assertion (A) and Reason (R). Answer these questions selecting the appropriate option given below:
Assertion: Based on the given experimental setup, it can be concluded that exhaled air contains more carbon dioxide than inhaled air.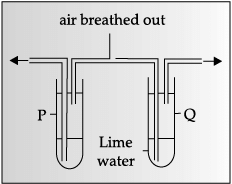
Reason: Carbon dioxide gas turns lime water milky.
(a) Both A and R are true and R is the correct explanation of A.
(b) Both A and R are true but R is NOT the correct explanation of A.
(c) A is true but R is false.
(d) A is false and R is true.
Ans: (a)
Explanation:
(i) Assertion: Exhaled air contains more carbon dioxide than inhaled air. (True.)
(ii) Reason: Carbon dioxide turns lime water milky. (True.)
(iii) Justification: In the experiment, exhaled air turns lime water milky while inhaled air does not, so R explains A and is the correct explanation.
Q.34: Directions: The consist of two statements. Assertion (A) and Reason (R). Answer these questions selecting the appropriate option given below:
Assertion: The focal length of a convex mirror increases when it is held underwater.
Reason: The focal length of a concave mirror is independent of the medium in which it is placed.
(a) Both A and R are true and R is the correct explanation of A.
(b) Both A and R are true but R is NOT the correct explanation of A.
(c) A is true but R is false.
(d) A is false and R is true.
Ans: (d)
Explanation:
(i) Assertion: The focal length of a convex mirror increases when held underwater. (False.)
(ii) Reason: The focal length of a mirror is independent of the medium in which it is placed. (True.)
(iii) Justification: Mirrors form images by reflection, and their focal length depends only on their shape (curvature). It does not change with surrounding medium. Therefore the assertion is false while the reason is true.
Q.35: Identify the correct representation of reaction occurring during chloralkali process
(A) 2NaCl(l) + 2H2O(l) → 2NaOH(l) + Cl2(g) + H2(g)
(B) 2NaCl(aq) + 2H2O(aq) → 2NaOH(aq) + Cl2(g) + H2(g)
(C) 2NaCl(aq) + 2H2O(l) → 2NaOH(aq) + Cl2(aq) + H2(aq)
(D) 2NaCl(aq) + 2H2O(l) → 2NaOH(aq) + Cl2(g) + H2(g)
Ans: (d)
Explanation: In the chloralkali process, electrolysis of aqueous sodium chloride (brine) produces aqueous sodium hydroxide, chlorine gas and hydrogen gas. The correct representation with appropriate phases is:
2NaCl(aq) + 2H2O(l) → 2NaOH(aq) + Cl2(g) + H2(g).
Q.36: What happens when calcium is treated with water?
(i) It does not react with water
(ii) It reacts violently with water
(iii) It reacts less violently with water
(iv) Bubbles of hydrogen gas formed stick to the surface of calcium
(a) (i) and (iv)
(b) (ii) and (iii)
(c) (i) and (ii)
(d) (iii) and (iv)
Ans: (d)
Explanation: Calcium reacts moderately (not violently like sodium or potassium) with water to give calcium hydroxide and hydrogen gas:
Ca + 2H2O → Ca(OH)2 + H2.
The reaction is less violent than with alkali metals, and bubbles of hydrogen often stick to the metal surface, so (iii) and (iv) are correct.
Q.37: The correct sequence of circulation of blood in the human body is :
(A) Lungs → Pulmonary vein → Left ventricle → Left atrium → Aorta → Body Organs → Vena Cava → Right Ventricle → Right atrium → Pulmonary artery → Lungs
(B) Lungs → Pulmonary vein → Left atrium → Left ventricle → Aorta → Body organs → Vena cava → Right atrium → Right ventricle → Pulmonary artery → Lungs.
(C) Lungs → Left atrium → Left ventricle → Body organs → Vena cava → Right atrium → Right ventricle → Pulmonary artery → Pulmonary vein → Aorta → Lungs.
(D) Lungs → Pulmonary artery → Pulmonary vein → Body organs → Vena cava → Right atrium → Right ventricle → Aorta → Left ventricle → Left atrium → Lungs
Ans: (b)
Explanation: Oxygenated blood from the lungs goes via the pulmonary vein to the left atrium, then to the left ventricle, and is pumped out through the aorta to body organs. Deoxygenated blood returns via the vena cava to the right atrium, then to the right ventricle and is sent through the pulmonary artery to the lungs for oxygenation. Option (b) correctly lists this sequence.
Q.38: Which of these statements is correct about peristaltic movement?
(A) It is the contraction and expansion of lungs.
(B) It is the contraction and expansion of walls of food pipe.
(C) It is the movement of limbs while walking.
(D) It is the movement of blood throughout the body.
Ans: (b)
Explanation: Peristalsis is the coordinated rhythmic contraction and relaxation of the muscles along the alimentary canal (food pipe and gut) that pushes food forward during digestion. It is not related to lung movement, limb movement or blood circulation.
Q.39: In the given experiment set-up, _______ gas is evolved as gas bubbles in the test tube. 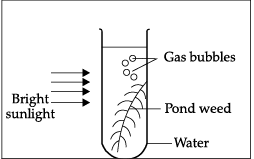
(A) Oxygen
(B) Carbon dioxide
(C) Nitrogen
(D) Hydrogen
Ans: (a)
Explanation: The experiment likely shows photosynthesis where aquatic plants release gas bubbles. The gas evolved during photosynthesis is oxygen, produced as water molecules are split during the light-dependent reactions.
Q.40: The blood sample _____ is most probably from a pulmonary artery.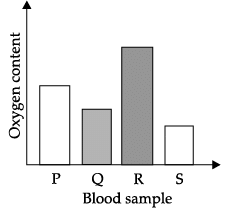
(A) P
(B) Q
(C) R
(D) S
Ans: (d)
Explanation: Pulmonary artery is the only artery that carries deoxygenated blood (rich in carbon dioxide) from the heart to the lungs. Most arteries carry oxygen-rich blood. The sample labelled S corresponds to deoxygenated (dark) blood and thus is most likely from the pulmonary artery.
Q.41: Identify Y and Z in the given flow chart.
(a) Y- Aerobic, Z- Anaerobic
(b) Y -Anaerobic, Z- Aerobic
(c) Y-Glycolysis, Z- Fermentation
(d) Y- Glycolysis, Z - Aerobic
Ans: (a)
Explanation: In the flow chart X represents glycolysis (initial breakdown of glucose). From glycolysis, if oxygen is present the pathway continues via aerobic respiration (Y), while in the absence of oxygen it proceeds via anaerobic pathways (Z). So Y = aerobic, Z = anaerobic.
Q.42: A 10 mm long awl pin is placed vertically in front of a concave mirror. A 5 mm long image of the awl pin is formed at 30 cm in front of the mirror. The focal length of this mirror is
(a) - 30 cm
(b) - 20 cm
(c) - 40 cm
(d) - 60 cm
Ans: (b)
Explanation: Given object height h = 10 mm, image height h' = 5 mm so magnification m = h'/h = 0.5. For mirrors m = -v/u. Given image distance v = -30 cm (real image in front of mirror is taken as negative here as per their sign convention), so -30/ u = 0.5 ⇒ u = -60 cm. Use mirror formula 1/v + 1/u = 1/f ⇒ (1/-30) + (1/-60) = 1/f ⇒ (-2 -1)/60 = 1/f ⇒ -3/60 = 1/f ⇒ f = -20 cm. The focal length is -20 cm (negative sign indicates concave mirror). (Option b)
Q.43: A child is standing in front of a magic mirror. She finds the image of her head bigger, the middle portion of her body of the same size and that of the legs smaller. The following is the order of combinations for the magic mirror from the top.
(a) Plane, convex, and concave
(b) Convex, concave, and plane
(c) Concave, plane, and convex
(d) Convex, plane, and concave
Ans: (c)
Explanation: A concave mirror (top) can make nearby parts appear larger (enlarged head). A plane mirror (middle) gives same-size images. A convex mirror (bottom) makes objects appear smaller. Thus the top-to-bottom order concave, plane, convex matches the observations.
Q.44: At which position, the object is needed to be placed to produce an image by a convex lens, at the position shown in the figure? 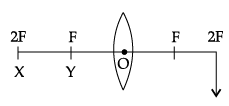
(a) Between Y and O
(b) Between X and Y
(c) At Y
(d) At X
Ans: (d)
Explanation: To produce an image at the given position (which corresponds to 2F), the object must be placed at the symmetric point, i.e., at X (which is at 2F). For a convex lens, an object at 2F produces an image at 2F on the opposite side, same size.
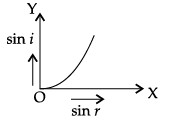
Q.45: The correct graphical relation between sine of angle of incidence (i) and sine of angle of refraction (r) for a given medium is :
(a)
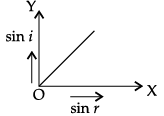
(b)
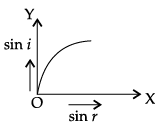
(c)
(d)
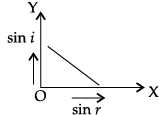
Ans: (a)
Explanation: Snell's law gives sin i / sin r = μ, a constant for a given pair of media. Therefore sin i is directly proportional to sin r, giving a straight line through the origin - option (a).
Q.46: Choose the correct diagram that depicts the reflection of a ray of light on a spherical mirror?
(a)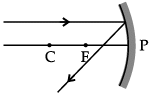
(b)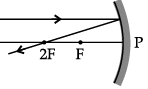
(c)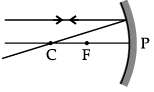
(d)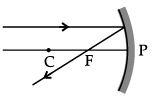
Ans: (c)
Explanation: A ray parallel to the principal axis after reflection from a concave mirror passes through the principal focus; for a convex mirror it appears to diverge from the principal focus. The diagram in option (c) correctly shows this behaviour for the given mirror.
Q.47: Vivaan placed an object O at the position shown in the given figure.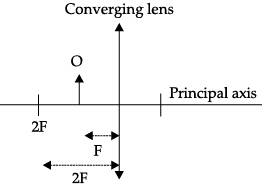
The characteristics of the image formed are:
(a) Real, inverted and diminished
(b) Real, inverted and enlarged
(c) Virtual, upright and enlarged
(d) No image is formed
Ans: (b)
Explanation: From the diagram the object is placed between the centre of curvature and the focus of a converging lens/mirror. In that region the image formed is real, inverted and enlarged, located on the opposite side. Thus option (b) correctly describes the image characteristics.
Q.48: In the given diagram, an incident ray strikes a rectangular glass block. 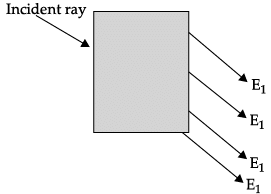
The correct emergent ray is :
(a) E1
(b) E2
(c) E3
(d) E4
Ans: (b)
Explanation: A rectangular (parallel-sided) glass block displaces a ray laterally but the emergent ray remains parallel to the incident ray. Among the options, E2 is the ray parallel to the incident ray after passing through the block, so it is the correct emergent ray.
Section-C
Q.49: Case:- The Salt Story
From: The New Indian Express 9 March 2021
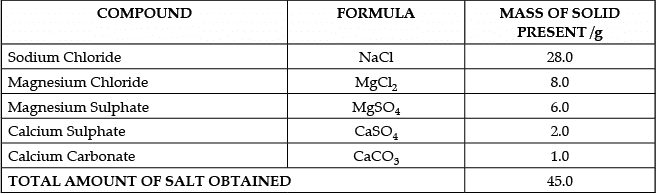
The salt pans in Marakkanam, a port town about 120 km from Chennai are the third largest producer of salt in Tamil Nadu. Separation of salt from water is a laborious process and the salt obtained is used as raw materials for manufacture of various sodium compounds. One such compound is Sodium hydrogen carbonate, used in baking, as an antacid and in soda acid fire extinguishers. The table shows the mass of various compounds obtained when 1litre of sea water is evaporated
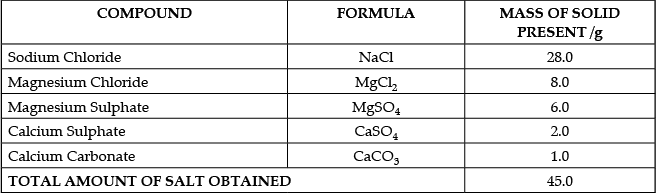
Which compound in the table reacts with acids to release carbon dioxide?
(a) NaCl
(b) CaSO4
(c) CaCO3
(d) MgSO4
Ans: (c)
- Explanation: Acids react with metal carbonates and hydrogen carbonates to form salt, carbon dioxide and water.
- From the options, calcium carbonate (CaCO3) is a carbonate and will react with acids to release CO2.
- Example: CaCO3 + 2HCl → CaCl2 + CO2 + H2O.
Q.50: Case:- The Salt Story
From: The New Indian Express 9 March 2021
The salt pans in Marakkanam, a port town about 120 km from Chennai are the third largest producer of salt in Tamil Nadu. Separation of salt from water is a laborious process and the salt obtained is used as raw materials for manufacture of various sodium compounds. One such compound is Sodium hydrogen carbonate, used in baking, as an antacid and in soda acid fire extinguishers. The table shows the mass of various compounds obtained when 1litre of sea water is evaporated
How many grams of Magnesium Sulphate are present in 135g of solid left by evaporation of sea water?
(a) 6g
(b) 12g
(c) 18g
(d) 24g
Ans: (c)
Explanation: From the table, 45 g of solid contains 6 g of MgSO4.
So per gram of solid there is 6/45 g of MgSO4.
For 135 g of solid: (6/45) × 135 = 6 × 3 = 18 g MgSO4.
Q.51: Case:- The Salt Story
From: The New Indian Express 9 March 2021
The salt pans in Marakkanam, a port town about 120 km from Chennai are the third largest producer of salt in Tamil Nadu. Separation of salt from water is a laborious process and the salt obtained is used as raw materials for manufacture of various sodium compounds. One such compound is Sodium hydrogen carbonate, used in baking, as an antacid and in soda acid fire extinguishers. The table shows the mass of various compounds obtained when 1litre of sea water is evaporated

What is the saturated solution of Sodium Chloride called?
(a) Brine
(b) Lime water
(c) Slaked lime
(d) Soda water
Ans: (a)
Explanation: A concentrated solution of sodium chloride is called brine. Brine is used in many processes including salt production and some chemical industries.
Q.52: Case:- The Salt Story
From: The New Indian Express 9 March 2021
The salt pans in Marakkanam, a port town about 120 km from Chennai are the third largest producer of salt in Tamil Nadu. Separation of salt from water is a laborious process and the salt obtained is used as raw materials for manufacture of various sodium compounds. One such compound is Sodium hydrogen carbonate, used in baking, as an antacid and in soda acid fire extinguishers. The table shows the mass of various compounds obtained when 1litre of sea water is evaporated

What is the pH of the acid which is used in the formation of common salt?
(a) Between 1 to 3
(b) Between 6 to 8
(c) Between 8 to 10
(d) Between 11 to 13
Ans: (a)
Explanation: Common salt (sodium chloride) is formed by the neutralisation of a strong acid (hydrochloric acid) with a strong base (sodium hydroxide). Strong acids typically have pH values between about 1 and 3, so option (a) is correct.
Q.53: The Figure shown below represents an activity to prove the requirements for photosynthesis. During this activity, two healthy potted plants were kept in the dark for 72 hours. After 72 hours, KOH is kept in the watch glass in setup X and not in setup Y. Both these setups are air tight and have been kept in light for 6 hours. Then, Iodine Test is performed with one leaf from each of the two plants X and Y.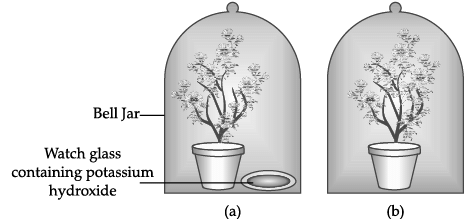
This experimental set up is used to prove essentiality of which of the following requirements of photosynthesis?
(a) Chlorophyll
(b) Oxygen
(c) Carbon dioxide
(d) Sunlight
Ans: (c)
Explanation: In setup X potassium hydroxide (KOH) is present and it absorbs carbon dioxide from the air inside the jar. As a result plant X cannot access CO2 while plant Y can. After exposing both to light and testing leaves with iodine, only the leaf from plant Y (which had CO2) will turn blue-black (starch present). This shows carbon dioxide is essential for photosynthesis.
Q.54: The Figure shown below represents an activity to prove the requirements for photosynthesis. During this activity, two healthy potted plants were kept in the dark for 72 hours. After 72 hours, KOH is kept in the watch glass in setup X and not in setup Y. Both these setups are air tight and have been kept in light for 6 hours. Then, Iodine Test is performed with one leaf from each of the two plants X and Y.
The function of KOH is to absorb
(a) Oxygen.
(b) Carbon dioxide.
(c) Moisture.
(d) Sunlight.
Ans: (b)
Explanation: Potassium hydroxide reacts with carbon dioxide to form potassium carbonate, thus removing CO2 from the air inside the jar. This prevents photosynthesis in the plant inside that jar if CO2 is essential.
Q.55: The Figure shown below represents an activity to prove the requirements for photosynthesis. During this activity, two healthy potted plants were kept in the dark for 72 hours. After 72 hours, KOH is kept in the watch glass in setup X and not in setup Y. Both these setups are air tight and have been kept in light for 6 hours. Then, Iodine Test is performed with one leaf from each of the two plants X and Y.
Which of the following statements shows the correct results of Iodine Test performed on the leaf from plant X and Y respectively?
(a) Blue - black colour would be obtained on the leaf of plant X and no change in colour on leaf of plant Y.
(b) Blue - black colour would be obtained on the leaf of plant Y and no change in colour on leaf of plant X.
(c) Red colour would be obtained on the leaf of plant X and brown colour on the leaf of plant Y.
(d) Red colour would be obtained on the leaf of plant Y and brown colour on the leaf of plant X.
Ans: (b)
Explanation: Iodine test detects starch: a blue-black colour indicates starch is present. Plant X had its CO2 absorbed by KOH and therefore cannot synthesise starch; its leaf will show no blue-black change. Plant Y had CO2 available and will produce starch, so its leaf turns blue-black on iodine test.
Q.56: The Figure shown below represents an activity to prove the requirements for photosynthesis. During this activity, two healthy potted plants were kept in the dark for 72 hours. After 72 hours, KOH is kept in the watch glass in setup X and not in setup Y. Both these setups are air tight and have been kept in light for 6 hours. Then, Iodine Test is performed with one leaf from each of the two plants X and Y.
Which of the following steps can be followed for making the apparatus air tight?
i. placing the plants on glass plate
ii. using a suction pump.
iii. applying vaseline to seal the bottom of jar.
iv. creating vacuum
(a) i and ii
(b) ii. and iii
(c) i. and iii
(d) ii. and iv
Ans: (c)
Explanation: To make the setup air tight we can place the plants on a glass plate to remove gaps at the base and apply Vaseline to seal the jar-glass contact. Using a suction pump or creating a vacuum would remove air but does not by itself make the apparatus sealed in the same simple way; these steps are not required for making it air tight in this practical setup. Therefore (i) and (iii) are correct.
Q.57: Noor, a young student, was trying to demonstrate some properties of light in her Science project work. She kept 'X' inside the box (as shown in the figure) and with the help of a laser pointer made light rays pass through the holes on one side of the box. She had a small butter-paper screen to see the spots of light being cast as they emerged.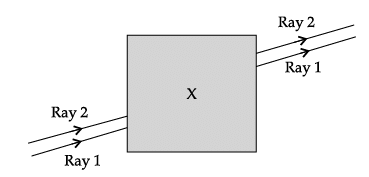
What could be the 'X' that she placed inside the box to make the rays behave as shown?
(a) a converging lens
(b) a parallel-sided glass block
(c) a plane mirror
(d) a triangular prism
Ans: (b)
Explanation: A parallel-sided glass block shifts the transmitted beams laterally but keeps the emergent rays parallel to the incident rays. The observed pattern of spots consistent with parallel displacement indicates a parallel-sided glass block was used.
Q.58: Noor, a young student, was trying to demonstrate some properties of light in her Science project work. She kept 'X' inside the box (as shown in the figure) and with the help of a laser pointer made light rays pass through the holes on one side of the box. She had a small butter-paper screen to see the spots of light being cast as they emerged.
She measured the angles of incidence for both the rays on the left side of the box to be 48.6°. She knew the refractive index of the material 'X' inside the box was 1.5. What will be the approximate value of angle of refraction?
(use the value: sin 48.6° ≈ 0.75)
(a) 45°
(b) 40°
(c) 30°
(d) 60°
Ans: (c)
Explanation: Refractive index n = sin i / sin r ⇒ 1.5 = 0.75 / sin r ⇒ sin r = 0.75 / 1.5 = 0.5 ⇒ r = 30°. Therefore the angle of refraction is approximately 30°.
Q.59: Noor, a young student, was trying to demonstrate some properties of light in her Science project work. She kept 'X' inside the box (as shown in the figure) and with the help of a laser pointer made light rays pass through the holes on one side of the box. She had a small butter-paper screen to see the spots of light being cast as they emerged.
Her friend noted the following observations from this demonstration:
i. Glass is optically rarer than air.
ii. Air and glass allow light to pass through them with the same velocity.
iii. Air is optically rarer than glass.
iv. Speed of light through a denser medium is faster than that of a rarer medium.
v. The ratio: sin of angle of incidence in the first medium to the ratio of sin of angle of refraction in the second medium, gives the refractive index of the second material with respect to the first one.
Which one of the combination of the above statements given below is correct.
(a) ii, iv and v are correct.
(b) iii and iv are correct.
(c) i, iv and v are correct.
(d) iii and v are correct.
Ans: (d)
Explanation: Air is optically rarer than glass (iii is correct). The ratio sin i / sin r gives the relative refractive index of the second medium with respect to the first (v is correct). Statements (i), (ii) and (iv) are incorrect: glass is not rarer than air, velocities in the two media are different, and light travels slower in a denser medium than in a rarer one.
Q.60: Noor, a young student, was trying to demonstrate some properties of light in her Science project work. She kept 'X' inside the box (as shown in the figure) and with the help of a laser pointer made light rays pass through the holes on one side of the box. She had a small butter-paper screen to see the spots of light being cast as they emerged.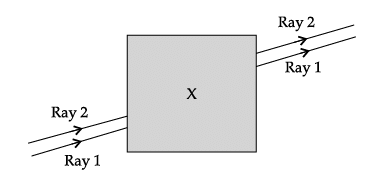
If the object inside the box was made of a material with a refractive index less than 1.5 then the
(a) lateral shift of the rays would have been less.
(b) lateral shift of the rays would have been more.
(c) lateral shift of the rays would remain the same as before.
(d) there is not enough information to comment on any of the above statements
Ans: (a)
Explanation: For a parallel-sided slab, lateral shift depends on the refractive index - larger refractive index generally leads to a greater change in direction inside the material and hence a larger lateral shift for the same thickness and angle of incidence. If the refractive index decreases (less than 1.5), the deviation inside the block is smaller and the lateral shift becomes less. Therefore option (a) is correct.