Is Matter Around Us Pure Class 9 Worksheet Science

Multiple Choice Questions
Q.1. Kerosene and Petrol are miscible liquids. The difference between their boiling points is more than 25°C. The two liquids can be separated from each other by _____.
(a) Simple distillation
(b) Steam distillation
(c) Fractional distillation
(d) Any of these
Correct Answer is Option (a)
By simple distillation. Vapours of the liquid which has low boiling point will be formed first and collected. The liquid having higher boiling point will remain in the vessel.
Q.2. How can a saturated solution be made unsaturated?
(a) By heating the solution
(b) By cooling the solution
(c) By increasing the amount of solute
(d) By centrifugation of the solution
Correct Answer is Option (a)
A saturated solution can be made unsaturated by increasing the temperature of solution by heating it or by increasing the amount of solvent in the solution i.e. diluting it.
Q.3. The cause of Brownian movement is:
(a) Heat changes in liquid state
(b) Convection currents
(c) Impact of molecules of dispersion medium on on dispersed phase
(d) Attractive forces between the particles of dispersed phase and dispersion medium.
Correct Answer is Option (c)
Zig-zag path of colloidal particles is called Brownian Movement. Zig-zag path of particles is due to collision of particles of dispersed phase and dispersion medium.
Q.4. In which of the following, dispersed phase is a liquid and dispersion medium is a gas?
(a) Cloud
(b) Smoke
(c) Gel
(d) Soap bubble
Correct Answer is Option (a)
In cloud, water drops are suspended in air.
Q.5. At room temperature, a non-metal which is a liquid is:
(a) Sulphur
(a) Bromine
(a) Chlorine
(a) Nitrogen
Correct Answer is Option (b)
Bromine is liquid at room temperature.
Fill in the Blanks
1. Common salt is _________.
Ans: Compound
2. A mixture contains more than ______ substance mixed in ______ proportion.
Ans: One, any
3. Properties of a __________ are different from its constituent elements, whereas a _______
shows the properties of its constituting elements.
Ans: Compound, mixture
4. A solution is defined as a mixture that is_________
Ans: Homogeneous
5. We can remove salts from a solution by using the process of _________
Ans: Evaporation
6. A pure substance has a fixed__________ or ______ at constant temperature.
Ans: Melting Point, Boiling Point
7. An element is made up of only one kind of _________.
Ans: Atoms
8. Miscible liquids are separated by ________ .
Ans: Fractional distillation
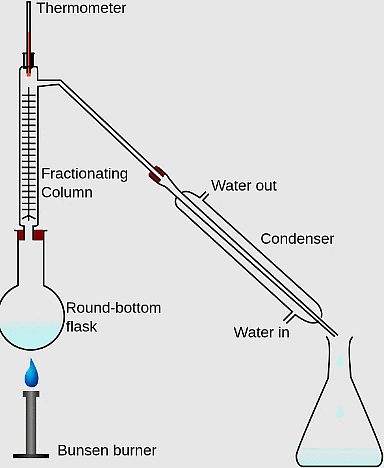
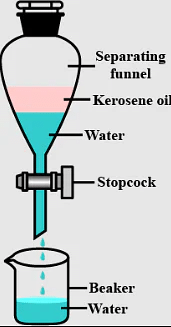
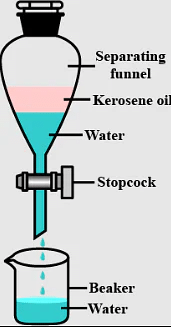
9. Immiscible liquids are separated by using a _______.
Ans: Separating funnel
10. Filtered tea is a _________ mixture.
Ans: Homogeneous
11. Alloy is a _______.
Ans: Solid solution
12. Sublimation of camphor is a _________ change.
Ans: Physical
13. Most common chemical change we observe in our routine life is rusting of______.
Ans: Iron
Very Short Answer Question
Q.1. Classify the substances given in below figure into elements and compounds
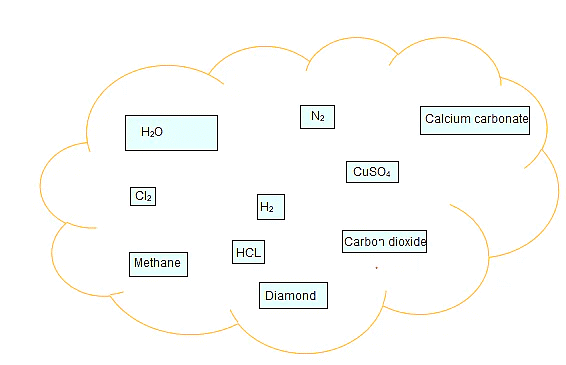
Ans:
Q.2. Give one example each of homogeneous and heterogeneous mixture.
Ans: Homogeneous :Brass Heterogenous: Sand and water
Q.3. Name the apparatus by which mixture of oil and water can be separated.
Ans: Separating funnel
 Q.4. Is brass a mixture or a compound?
Q.4. Is brass a mixture or a compound?
Ans: Mixture
Q.5. What type of solution is an alloy? Liquid solution or solid solution
Ans: Solid solution
Q.6. A mixture consisting of two miscible liquids 'A' and 'B' whose boiling points differ by 50 C can be separated by which process?
Ans: Fractional distillation
Q.7. Give one example of solid- liquid homogeneous mixture.
Ans: Salt in water solution
Q.8. What is a Aqua regia?
Ans: Aqua regia is a highly-corrosive mixture of - nitric acid and hydrochloric acid. The mixture is formed by freshly mixing concentrated nitric acid and hydrochloric acid, usually in a volume ratio of 1:3
Q.9. Which method is used to separate two immiscible liquids?
Ans: Separating funnel
Q.10. Name two elements which are in liquid state at room temperature?
Ans: The only liquid elements at standard temperature and pressure arebromine (Br) and mercury (Hg). Although, elements caesium (Cs),rubidium (Rb), Francium (Fr) and Gallium (Ga) become liquid at or just above room temperature.
Short Answer Types Questions
Q.1. Try segregating the things around you as pure substances or mixtures.
You can do it by yourself like try mixing chalk powder and water then separate them.
Q.2. What is meant by a substance?
Substance can be defined as that kind of matter where constituent particles cannot be separated from each other by any physical process since they are all similar in chemical properties.
Q.3. What type of mixtures are separated by the technique of crystallisation?
From impure samples of solids, pure solid crystals can be obtained by the method of crystallization for eg to obtain pure sugar from impure sample of the same.
Q.4. What is tyndall effect? Which kinds of solution show it?
The scattering of a beam of light by particles of solution when light is passed through it is called tyndall effect. Those solutions where size of the particle is very small for e.g. colloidal solution shows tyndall effect.
Q.5. What is centrifugation? Where it is used?
Centrifugation is a technique used for separation of constituents of mixture and is based upon the principle that denser particles stay at bottom and lighter particles stays at the top when spun rapidly. It is used separate cream from milk.
Q.6. What is crystallization? Where is it used? Why is this better than simple evaporation technique?
Crystallization is a process that separates a pure solid in the form of crystals from its solution. It is used to purify solids. For e.g. salt from sea water is purified using crystallization.
It is a better technique than simple evaporation because:
(a) Some solid may decompose or get charred on heating to dryness during evaporation.
(b) On evaporation, some of the impurities still remain dissolved in the solution.
Q.7. What is a colloid? What are its various properties?
Colloids are the heterogeneous mixture of substances in which the particle size is too small and cannot be seen by naked eyes.
(a) It is a heterogeneous mixture, but appears homogenous.
(b) The size of particles is too small to be individually seen by naked eyes.
(c) They scatter beam of light passing through it and makes its path visible.
(d) The particles of colloid do not settle down when left undisturbed.
Q.8. Write a method to separate different gases from air.
Air is a homogeneous mixture of various gases. It can be separated from its various components by fractional distillation.
(a) First compress and cool the air by increasing the pressure and decreasing the temperature.
(b) We obtain the liquid air; now allow the liquid air to warm up slowly in fractional distillation column.
(c) The various gases separate from each other according to their boiling points at various heights of the fractionally column.
Q.9. Explain the following giving examples.
(a) saturated solution
(b) pure substance
(c) colloid
(d) suspension
(a) saturated solution: It is a solution in which no more solute particles can be dissolved at a particular temperature.
(b) pure substance: Such substance that has a uniform composition i.e. has particles with identical properties is called pure substance eg sugar, salt, water, nitrogen etc.
(c) colloid: It is a kind of heterogeneous mixture/solution in which particle size is between 1nm and 1000nm. Colloids have dispersion medium and dispersed phase.eg smoke, milk, shaving cream, jelly, cheese etc.
(d) suspension: It is a kind of heterogeneous mixture in which insoluble solid particles remain suspended in the medium and dispersion particles are visible to the unaided eyes.eg muddy river water, chalk powder in water, dust storm, sand in water etc.
Q.10. Write the steps you would use for making tea. Use the words solution, solvent, solute, dissolve, soluble, insoluble, filtrate and residue.
Take more amount of solvent (water) in a pan and after heating it add little amount of solute (sugar) to the solvent. Solute will dissolve completely in the solvent forming true solution, then add tea leaves that are insoluble along with another soluble liquid milk. After boiling allow filtration with a sieve so the filtrate you obtain is tea while the residue has tea leaves that are thrown away.
Crossword Puzzle
Across
1. hydrogen ______ is a color gas with a smell of rotten eggs
5. The major components in solution
6. Melting point and boiling point are _______ properties
7. Two elements are liquid at room temperature are mercury and _______
Down
2. In colloids ,The particles are called the ______ phase and the medium in which they are distributed is called the dispersion medium.
3. amount of solute present per unit volume or mass of the solution or solvent
4. denser particles are forced to the bottom and the lighter particles stay at the top when spun rapidly
Ans:
1. sulphide
2. dispersed
3. concentration
4.centrifugation
5. solvent
6. physical
7. bromine
|
88 videos|369 docs|67 tests
|
FAQs on Is Matter Around Us Pure Class 9 Worksheet Science
| 1. What is the difference between a pure substance and a mixture? |  |
| 2. How can you determine if something is a pure substance or a mixture? |  |
| 3. Give an example of a pure substance and a mixture. |  |
| 4. Can mixtures be classified into different types? |  |
| 5. How can mixtures be separated into their individual components? |  |

|
Explore Courses for Class 9 exam
|

|



















