Worksheet Solutions: Chemical Effects of Electric Current
Q1: Which effect of electric current is utilized for detecting the flow of current through a solution when a compass is used?
Ans: Magnetic effect of electric current is utilised for detecting the flow of current through a solution when a compass is used. When current flows through the solution, it produces a magnetic field around the conductor; the compass needle detects this magnetic field and deflects, showing that current is flowing.
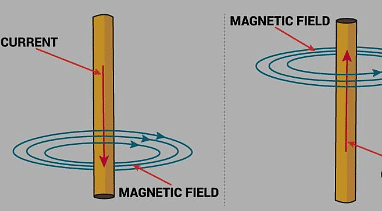 Magnetic effect of Electric Current
Magnetic effect of Electric Current Ans: The passage of an electric current through a conducting liquid causes chemical changes at the electrodes. These are called the chemical effects of electric current. Example: When electric current is passed through a copper sulphate solution during electroplating, copper ions are deposited on the cathode as a metal coating.
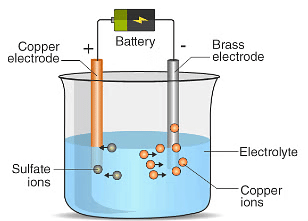
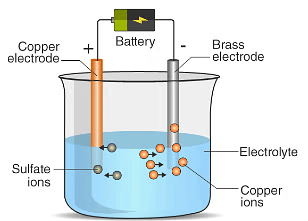 Chemical Effect of Electric Current
Chemical Effect of Electric CurrentAns: When electric current is passed through copper sulphate solution, the compound dissociates into copper ions and sulphate ions. The positively charged copper ions move towards the electrode connected to the negative terminal (the carbon rod) and get deposited there as copper metal. At the same time, copper atoms from the copper plate (connected to the positive terminal) dissolve into the solution to replace the lost copper ions. As a result, copper is transferred from the copper plate (anode) to the carbon rod (cathode).
Ans: Silver and gold are usually electroplated on cheaper metals to give the appearance and some properties of the precious metal while reducing cost.
 Silver and Gold
Silver and GoldAns: Due to the heating effect of current, the filament of the bulb becomes very hot and starts glowing, producing light. If the current is too weak, the filament does not heat up enough and the bulb will not glow.
 Bulb
BulbAns: Distilled water is a poor conductor of electricity because it does not contain dissolved salts or impurities that provide ions to carry current. If a little common salt is dissolved in distilled water, it becomes a good conductor. Tap water usually contains dissolved salts and minerals, so it conducts electricity.
Q7: The process that you saw in Activity 14.7 is used for purification of copper. A thin plate of pure copper and a thick rod of impure copper are used as electrodes. Copper from impure rod is sought to be transferred to the thin copper plate. Which electrode should be attached to the positive terminal of battery and why?
Ans: The thick rod of impure copper should be connected to the positive terminal of the battery (anode). The thin plate of pure copper should be connected to the negative terminal (cathode). On passing electric current, metal from the impure anode dissolves into the electrolyte as ions and these ions are deposited on the pure copper cathode. This transfers pure copper onto the thin plate, leaving impurities behind.
Q8: Does pure water conduct electricity? If not, what can we do to make it conducting?
Ans: Pure water, being free of dissolved salts, is a poor conductor of electricity. We can make it conducting by adding a small amount of a soluble salt (for example, common salt), which provides ions that carry electric current.
Q9: In case of a fire, before the firemen use the water hoses, they shut off the main electrical supply for the area. Explain why they do this.
Ans: Water conducts electricity when it contains dissolved salts or impurities. If the electrical supply is still on, water sprayed by firemen may carry current and they could get electrocuted by wet electrical fittings, wires or appliances. Switching off the supply reduces this risk.
Q10: What happens when electric current is passed through acidified water?
Ans: When electrodes are immersed in acidified water and electric current is passed, oxygen gas is formed at the positive electrode (anode) and hydrogen gas is formed at the negative electrode (cathode). These gases appear as bubbles at the respective electrodes.
Q11: A child staying in a coastal region tests the drinking water and also the seawater with his tester. He finds that the compass needle deflects more in the case of seawater. Can you explain the reason?
Ans: The compass needle deflects more with seawater because seawater contains a large amount of dissolved salts, so it is a much better conductor of electricity than drinking water. A better conducting liquid allows a stronger current to flow, producing a larger magnetic effect that deflects the needle more.
Q12: What is the advantage of using LED in testing the electrical conductivity of liquids?
Ans: An LED glows even when a weak electric current flows through it. This makes it useful for testing electrical conductivity of liquids because it can indicate small currents. Additionally, LEDs consume little power and give a clear visible indication.
Q13: For electroplating copper on an iron object, which terminal of the battery (positive or negative) is connected to the iron object?
Ans: In electroplating, the object to be coated is made the cathode (negative) and the metal used for coating is made the anode (positive). Therefore, the iron object to be plated is connected to the negative terminal of the battery.
Q14: Is it safe for the electrician to carry out electrical repairs outdoors during heavy downpour? Explain.
Ans: It is not safe because rainwater and wet surfaces conduct electricity. Working outdoors in heavy rain increases the risk of electric shock or electrocution if live wires or fittings are touched while wet.
Q15: Which is the polluting waste generated by electroplating factories?
Ans: The conducting solution (electrolyte) used in the electroplating process, which contains dissolved metal ions and chemicals, is the polluting waste produced by electroplating factories. If released untreated into the environment, these solutions can contaminate water and soil.
FAQs on Worksheet Solutions: Chemical Effects of Electric Current
| 1. What is the chemical effect of electric current? |  |
| 2. How does electric current cause chemical changes? |  |
| 3. What are some examples of chemical effects of electric current? |  |
| 4. How is electrolysis related to the chemical effects of electric current? |  |
| 5. Can the chemical effects of electric current be harmful? |  |






