Grade 10 Exam > Grade 10 Notes > Chemistry for Grade 10 > Introduction to Chromatography
Introduction to Chromatography | Chemistry for Grade 10 PDF Download
About Chromatography
- Chromatography is used to separate substances and provide information to help identify them
- The components have different solubilities in a given solvent (e.g. different coloured inks that have been mixed to make black ink) and different adhesion to the supporting medium - usually paper
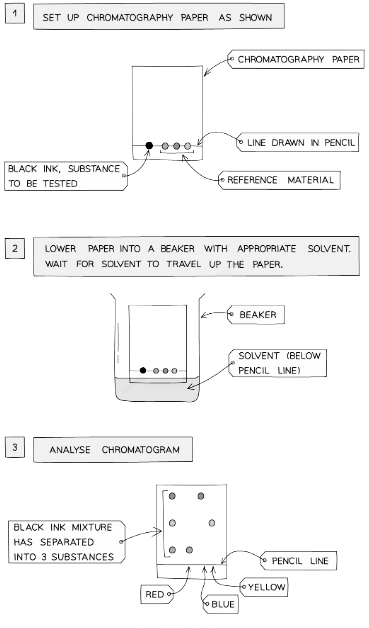
- A pencil line is drawn on chromatography paper and spots of the sample are placed on it
- Pencil is used for this as ink would run into the chromatogram along with the samples
- The paper is then lowered into the solvent container, making sure that the pencil line sits above the level of the solvent so the samples don’t wash into the solvent container
- The solvent travels up the paper by capillary action, taking some of the coloured substances with it
- Different substances have different solubilities so will travel at different rates, causing the substances to spread apart
- Those substances with higher solubility will travel further than the others
 The pigments in ink can be analysed using paper chromatography
The pigments in ink can be analysed using paper chromatography
- All chromatography techniques use two phases called the mobile phase and the stationary phase
- In paper chromatography:
- The mobile phase is the solvent in which the sample molecules can move, which in paper chromatography is liquid e.g. water or ethanol
- The stationary phase in paper chromatography is the actual chromatography paper itself
- The substances which are more soluble in the solvent will travel further up the paper because they spend more time in the mobile phase and are thus carried further up the paper than the less soluble components
Exam Tip
Paper chromatography is the name given to the overall separation technique while a chromatogram is the name given to the visual output of a chromatography run. This is the piece of chromatography paper with the visibly separated components after the run has finished.
Distinguishing Pure & Impure Substances
- Pure substances will produce only one spot on the chromatogram
- If two or more substances are the same, they will produce identical chromatograms
- If the substance is a mixture, it will separate on the paper to show all the different components as separate spots
- An impure substance therefore will produce a chromatogram with more than one spot
 Diagram showing the analysis of a mixture and pure substances using chromatography
Diagram showing the analysis of a mixture and pure substances using chromatography
Identifying Substances Using Rf Values
- These values are used to identify the components of mixtures
- The Rf value of a particular compound is always the same but it is dependent, however, on the solvent used
- If the solvent is changed then the value changes
- Calculating the Rf value allows chemists to identify unknown substances because it can be compared with Rf values of known substances under the same conditions
- These values are known as reference values
Calculation
- Retention factor, Rf, is calculated by the equation:

- The Rf value is a ratio and therefore has no units and will be less that 1
 Using Rf values to identify components of a mixture
Using Rf values to identify components of a mixture - The Rf value of the substances in the chromatogram above can be calculated by:

Exam Tip
For the Rf calculations, both distances are measured from the baseline.
The document Introduction to Chromatography | Chemistry for Grade 10 is a part of the Grade 10 Course Chemistry for Grade 10.
All you need of Grade 10 at this link: Grade 10
|
78 videos|87 docs|11 tests
|

|
Explore Courses for Grade 10 exam
|

|
Signup for Free!
Signup to see your scores go up within 7 days! Learn & Practice with 1000+ FREE Notes, Videos & Tests.
Related Searches



















