Exercises: Acids, Bases & Salts
A. Fill in the blanks with the correct words.
Q.1. ____ (Strong/Weak) acids are highly corrosive and can cause severe burns.
Strong acids are highly corrosive and can cause severe burns.
Q.2. ____ (Acids/Bases) are slippery to touch.
Bases are slippery to touch.
Q.3. ____ (Nitric/Sulphuric) acid is used in the manufacture of paints, dyes and drugs.
Sulphuric acid is used in the manufacture of paints, dyes and drugs.
Q.4. ____ (Sodium hydroxide/Milk of magnesia) is used to unblock drains.
Sodium hydroxide is used to unblock drains.
Q.5. ____ (Red/Blue) litmus changes to ____ (red/blue) in acids.
Red litmus changes to blue in acids.
B. Choose the correct option.
Try yourself: Which of the following is a characteristic property of most of the salts?
Try yourself: Which acid is called the 'King of chemicals?
Try yourself: Which acid is present in vinegar?
Try yourself: Which of the following is a weak mineral acid?
Try yourself: Which of the following is used to neutralize bee stings?
Try yourself: Which of the following cannot be used as an indicator?
Try yourself: Which of the following is a salt?
Try yourself: Which acid is used to remove deposits from the inside of the boilers?
Try yourself: Which of the following is a strong base?
Try yourself: Which of the following pH values represents a strong alkali?
C. Match the following.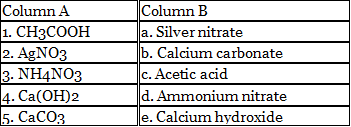
1. CH3COOH is the chemical formula for acetic acid.
2. AgNO3 is the chemical formula for silver nitrate.
3. NH4NO3 is the chemical formula for ammonium nitrate.
4. Ca(OH)2 is the chemical formula for calcium hydroxide.
5. CaCO3 is the chemical formula for calcium carbonate.
II. Very short answer type questions
Give one word for the following.
Q.1. Bases that dissolve in water
Alkalis
Q.2. A substance that shows a change in colour when brought in contact with an acid or a base
Indicator
Q.3. Universal indicator that is soaked into paper
Litmus
Q.4. Substance formed when a strong acid reacts with a strong base
Salt
Q. 5. Salts with water of crystallization
Hydrated salts
III. Short answer type questions
Q.1. State any two properties of acids and bases.
Properties of acids include sour taste, ability to turn blue litmus paper red, and the ability to react with metals to release hydrogen gas. Properties of bases include bitter taste, ability to turn red litmus paper blue, and slippery or soapy texture.
Q.2. Why acids or bases are sometimes added to soil?
Acids or bases are added to soil to adjust the pH level. This can help plants absorb nutrients more efficiently and also prevent soil erosion.
Q.3. What is a neutralization reaction? Give an example.
Neutralization reaction is a chemical reaction between an acid and a base to form a salt and water. An example of a neutralization reaction is the reaction between hydrochloric acid and sodium hydroxide to form sodium chloride (salt) and water.
Q.4. How can a hydrated salt be converted into anhydrous salt? Give an example.
A hydrated salt can be converted into an anhydrous salt by heating it to remove the water molecules. For example, hydrated copper sulfate (CuSO4.5H2O) can be heated to convert it into anhydrous copper sulfate (CuSO4).
IV. Long answer type questions
Q.1. How are natural indicators prepared? Give examples of a flower, root, stem and leaf that can be used as an indicator. What are the colour changes observed with red cabbage juice in acidic, neutral and basic medium?
Natural indicators are prepared by extracting pigments from plants and using them to test for acidity or alkalinity. Examples of natural indicators are:
Flower: Petals of hibiscus flowers can be used as an indicator. They turn acidic solutions into a bright red colour and alkaline solutions into a greenish-yellow colour.
Root: Beetroot is a good indicator. Its juice turns acidic solutions pink and alkaline solutions yellow.
Stem: Turmeric powder can be used as an indicator. It turns acidic solutions yellow and alkaline solutions reddish-brown.
Leaf: Red cabbage leaves can be boiled in water to extract their pigments, which can be used as an indicator. The juice turns acidic solutions pink, neutral solutions purple, and alkaline solutions greenish-yellow.
The colour changes observed with red cabbage juice in acidic, neutral and basic medium are as follows:
Acidic: pink
Neutral: purple
Basic: greenish-yellow
Q.2. Explain the following:
a. Vinegar is used in packaged food items.
Vinegar is used in packaged food items as a preservative and flavouring agent. It helps to prevent the growth of harmful bacteria and fungi and enhances the taste of the food.
b. Slaked lime is added to factory waste.
Slaked lime is added to factory waste to neutralize the acidic waste and make it less harmful to the environment. It also helps to precipitate the heavy metals present in the waste, making it easier to dispose of.
c. Magnesium hydroxide is used as an antacid.
Magnesium hydroxide is used as an antacid because it is a mild base that can neutralize excess stomach acid. It helps to relieve the symptoms of heartburn, indigestion, and acid reflux.
d. Strong acids and bases should be handled carefully.
Strong acids and bases should be handled carefully because they can cause severe burns and damage to the skin, eyes, and respiratory system. They should be stored in a safe place and handled with protective equipment such as gloves and goggles.
Q.3. What is water of crystallization? Explain with the help of an example.
Water of crystallization is the water molecules that are trapped inside the crystal lattice of a compound. These water molecules are chemically bonded to the compound and are essential for its stability. For example, copper sulfate pentahydrate (CuSO4.5H2O) is a blue crystalline compound that contains five water molecules. When heated, these water molecules are released and the compound turns white and powdery. The water of crystallization can also affect the physical properties of a compound, such as its solubility and melting point.
FAQs on Exercises: Acids, Bases & Salts
| 1. What's the difference between acids and bases in simple terms? |  |
| 2. How do I know if something is acidic or basic without using litmus paper? |  |
| 3. What exactly are salts and how do acids and bases make them? |  |
| 4. Why do my stomach antacids work when I have acidity problems? |  |
| 5. What happens when I mix an acid and a base together-will it be dangerous? |  |