Worksheet: Solids, Liquids and Gases - 1
Q1: Multiple Choice Questions (MCQs).
(i) Which state of matter has a definite shape and volume?
(a) Solid
(b) Liquid
(c) Gas
(d) Plasma

(ii) Which state of matter takes the shape of the container it is in?
(a) Solid
(b) Liquid
(c) Gas
(d) Plasma
(iii) Which state of matter has particles that move the fastest?
(a) Solid
(b) Liquid
(c) Gas
(d) Plasma
(iv) The process of a solid changing into a liquid is called:
(a) Melting
(b) Freezing
(c) Evaporation
(d) Condensation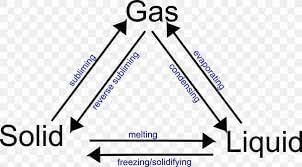
(v) The process of a gas changing into a liquid is called:
(a) Melting
(b) Freezing
(c) Evaporation
(d) Condensation
Q2: Fill in the blanks.
(i) The three states of matter are _______, _______, and _______.
(ii) Particles in a solid are packed closely together, making it _______.
(iii) Liquids do not have a fixed shape, instead, they take the shape of the _______.
(iv) Gases have no fixed shape or volume and can be compressed or _______.
(v) The change of a solid into a liquid is called _______.
Q3: Match the column.

Q4: True or False.
(i) Solids have a fixed shape.
(ii) Particles in a gas are loosely packed.
(iii) The process of a liquid turning into a solid is called evaporation.
(iv) The particles in a liquid are more tightly packed than in a solid.
(v) The particles in a gas have more energy than those in a solid or liquid.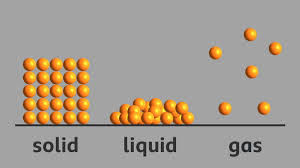
Q5: Short Answer Questions.
(i) What are the three main states of matter?
(ii) How do the particles in a solid, liquid, and gas differ from each other?
(iii) What is the process called when a liquid changes into a gas?
(iv) What happens to the particles of a substance when it is heated?
(v) What is the process called when a liquid changes into a solid?
You can find Worksheets Solutions here: Worksheet Solutions: Solids, Liquids and Gases - 1
FAQs on Worksheet: Solids, Liquids and Gases - 1
| 1. What are the main differences between solids, liquids, and gases for Class 5 science? |  |
| 2. Why do gases spread out so quickly compared to solids and liquids? |  |
| 3. Can matter change from one state to another, and what causes these changes? |  |
| 4. How can you identify whether something is a solid, liquid, or gas based on its properties? |  |
| 5. What happens to particles when a solid melts into a liquid or a liquid boils into a gas? |  |






