What are Titrations?
Introduction
Titration, in the context of chemistry, is an experimental technique used to determine the concentration of an unknown acid or alkali solution.
During a titration, a neutralization reaction occurs when an acid and alkali react together. By conducting a titration, we can measure the amount of acid required to neutralize a standard solution of alkali (with a known concentration), or vice versa, if the concentration of the acid is unknown.
Steps of Titration
The following are the steps involved in a typical titration:
- Prepare the burette: Fill a burette with the acid of unknown concentration. A burette is chosen for its precision in delivering the acid solution.
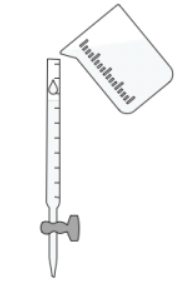
- Prepare the conical flask: Use a pipette to add a fixed volume of the standard alkali solution into a conical flask. A volumetric pipette is preferred for its accuracy.
- Add an indicator: Introduce an indicator into the alkali solution in the conical flask. The indicator causes a color change, which helps detect the endpoint of the titration.
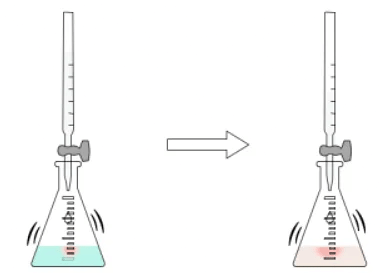
- Perform the titration: Gradually add the acid from the burette into the alkali solution in the conical flask while swirling the flask to ensure proper mixing of the reactants.
- Observe the indicator color change: As the acid is added, monitor the color change in the solution. When the indicator's color changes, slow down the addition of acid to a drop-by-drop rate.
- Stop the titration: Cease adding acid when a permanent color change is observed in the indicator.
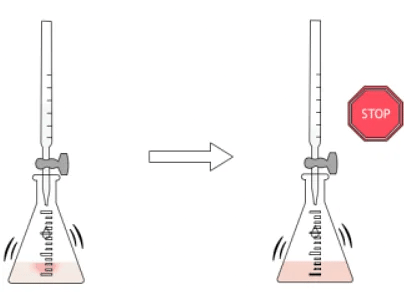
- Record the volume of acid used: Note the volume of acid (called the "titre") delivered from the burette. This volume is a crucial measurement for calculating the unknown concentration.
- Repeat the titration: Repeat the titration process until consistent and concordant titres are obtained, usually within a range of 0.1 cm³.
By analyzing the volume of acid required to neutralize the alkali and applying the principles of stoichiometry, the concentration of the unknown acid or alkali solution can be calculated.
Determining the Concentration of Acid
The concentration of an unknown acid can be calculated using the following steps:
- The known volume and concentration of the alkali solution allow the moles to be determined.
- The balanced symbol equation provides the moles ratio between the acid and alkali. This allows the number of moles of acid to be calculated.
- The mean titre, which is the volume of the acid solution used in the titration, is obtained. By combining the moles and volume information, the concentration of the unknown acid can be determined.





