Specific Heat Capacity
Specific Heat Capacity is a fundamental characteristic of matter that quantifies the quantity of heat energy needed to raise the temperature of 1 kg of matter by 1 degree Celsius. The SI unit for Specific Heat Capacity is J/(kgK), although it can also be expressed as cal/(gK) or J/(g°C). The specific heat capacity of a substance is influenced by its composition, density, and physical state. For example, the specific heat capacity of water varies across its different states.
Heat Capacity
The amount of heat energy required to increase the temperature of a given quantity of matter by 1°C, is known as the Heat Capacity of that matter. Heat Capacity is an extensive property of matter as it varies with the size and quantity of the matter.
Heat capacity is a measure of a system's total internal energy. This comprises the system's overall kinetic energy as well as the potential energy of the molecules. It has been demonstrated that a system's internal energy may be altered by either giving heat energy to it or doing work on it. The internal energy of a system is shown to grow as the temperature rises. This rise in internal energy is affected by temperature differences and the amount of substance present in the system Mathematically, the heat capacity is given by:
c = Q /ΔT
where,
- Q is the amount of heat energy necessary to cause a temperature change of ΔT
- c is the heat capacity of the system under consideration.
Unit of Heat Capacity
The SI unit of heat capacity is Joule per Kelvin (J/K) or Joule per degree Celsius (J/°C).
Specific Heat Capacity
Specific Heat Capacity is a physical characteristic of the system and is defined as the amount of energy required to increase the temperature of 1 unit substance by 1°C.
It is also an example of an extensive property because its value is proportional to the size of the system under consideration. Specific heat capacity, in general, is a measure of how much energy it takes to change the temperature of a system. However, it is critical to understand that the energy intake must be through heating. If work is done on the system, the temperature will rise; however, attempting to compute the temperature rise using the heat capacity and the quantity of work done on it is inaccurate.
Another thing to consider is the limitation that the system is held to. Because the latter does work on its surroundings as it expands, the specific heat capacity of a system kept at constant volume differs from that of a system held at constant pressure. Such discrepancies are typically overlooked when dealing with solids, but they are critical when working with gases.
Specific Heat Capacity Formula
A solid's or liquid's specific heat is the amount of heat required to increase the temperature of the unit mass of the solid by 1° C. It is represented by the symbol C.
If ΔQ is the amount of heat necessary to raise the temperature of mass m through ΔT, then the formula for specific heat is:
C = ΔQ ⁄ m ΔT
ΔQ = m c ΔT
Unit of Specific Heat Capacity
As it is the amount of heat required to increase the temperature of 1 kg of matter by 1 K. Thus,
- SI unit of Specific Heat Capacity is always J kg-1 K-1.
- In the cgs system, the unit of Specific Heat Capacity, Cal/(g°C) or J/(g°C)
- Dimensional Formula of Specific Heat Capacity is [M0 L2 T-2 K-1]
Specific Heat Capacity of Various Substances
The specific Heat Capacity of various substances in their different states of matter is shown in the following table.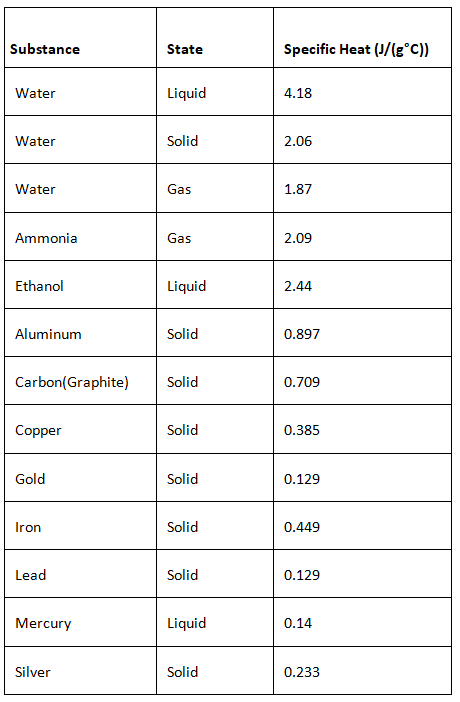
Molar Specific Heat
The molar specific heat of a matter is an amount of heat required to raise the temperature of one mole of substance by 1 °C or 1 °K.
Molar Specific Heat Capacity is denoted by the third alphabet of the English language "c"(lower case) and its unit is J/(mol K) or J/(mol °C). Mathematically Molar Specific Heat is given by the following formula:
c = Q/(n ΔT)
Q = ncΔT
where,
- Q is the amount of energy required to chagne temperature by ΔT
- n is the number of moles of the substance
- c is the specific heat of the system under considertation
Specific Heat of Water
Specific heat capacity of water at NTP(normal pressure and temperature) is approximately 4.2 J ⁄ (g°C) or 1 Cal ⁄(g°C) which implies that we need 4.2 joules of energy to raise the temperature of 1 gm of water by1 °C.
As water is not a conductor of heat, thus this number is actually pretty high compared to the other substances. Even water vapor has a higher specific heat capacity than many other materials at normal temperatures. Water vapor's specific heat capacity at normal pressure and temperature is approximately 1.9 J ⁄ (g°C). For the solid form of water i.e., ice, the specific heat capacity is 2.06 J ⁄ (g°C).
Water has a high specific heat capacity, which means that its temperature changes more slowly compared to other liquids when it absorbs or releases heat. This property allows water to absorb heat without causing an instant temperature increase and to retain its temperature for a longer period than many other substances. In the human body, we use this property of water to maintain a stable body temperature. A lower specific heat value of water would result in a higher risk of underheating and overheating in our bodies.
Specific Heat at Constant Pressure and Constant Volume
The specific heat at constant pressure and constant volume is discussed below in the article.
Specific Heat at Constant Pressure
The specific heat capacity at constant pressure (CP) is the amount of heat energy required to increase the temperature of a substance by one degree Celsius or Kelvin while keeping the pressure constant. CP includes the heat energy required to raise the temperature of the substance as well as the work done by the substance as it expands against the constant pressure.
Specific Heat at Constant Volume
The specific heat capacity at constant volume (CV) is the amount of heat energy required to increase the temperature of a substance by one degree Celsius or Kelvin while keeping the volume constant. CV only includes the heat energy required to raise the temperature of the substance since there is no expansion work being done against a constant volume.
Relation between CP and CV
The way gas is heated affects the behavior of the gas, the volume and pressure change in temperature, and the amount of heat necessary to increase the temperature of 1gm of gas by 1° C. We can heat the gas with a variety of P and V values.
As a result, the specific heat value is limitless. If we don't deliver a steady quantity of heat, the gas's specific heat will change. As a result, we will need a constant pressure or volume of specific heat.
For an ideal gas,
CP - CV = n R
where,
- CV is heat capacity at constant volume
- CP is heat capacity at constant pressure
- R is the molar gas constant
- n is amount of substance
The value of the Gas Constant, R = 8.3145 J mol-1 K-1
Heat Capacity Ratio: CP ⁄ CV
The adiabatic index is also known as the heat capacity ratio or ratio of specific heat capacities (CP:CV) in thermodynamics. The ratio of heat capacity at constant pressure (CP) to heat capacity at constant volume (CV) is defined as the heat capacity ratio.
The isentropic expansion factor, commonly known as heat capacity ratio, is indicated by γ for an ideal gas (gamma). As a result, specific heat ratio, γ is equal to ratio of CP to CV, i.e.
γ = CP ⁄ CV
Why is CP Greater than CV?
The specific heats of an ideal gas are represented by CP and CV. This is the amount of heat required to raise the temperature of unit mass by 1° C. By the first law of thermodynamics,
ΔQ = ΔU + ΔW
where,
- ΔQ is the amount of heat that is given to the system
- ΔU is the change in internal energy
- ΔW is the work done
At constant pressure, heat is absorbed to raise internal energy and do any work on the system. On the other side, heat is absorbed just to raise internal energy at constant volume, not to do any work on the system. As a result, the specific heat under constant pressure is greater than that at constant volume, i.e. Cp > Cv.
Uses of Specific Heat Capacity
The concept of specific heat capacity is used for the following below purposes,
- Insulators make use of materials with a high specific heat capacity. Take, for instance, wood. Houses built of wood are better suitable for areas with high or low temperatures.
- Swimming pool water used to be cold in comparison to the temperature outdoors due to the high specific heat of the water.
- Cooking utensils are made of a low-specific-heat material. You can immediately heat their bottoms. This is due to their polished aluminum or copper bottoms. To maintain the heat and safeguard our hands, the handles of these utensils are made of high-specific heat material.
Solved Example
Example: Determine the temperature difference if 40 Kg of water absorbs 400 K J of heat.
Solution:
Given:
Mass of water, m = 40 Kg
Heat transfer, Q = 400 KJ,
Specific heat of water, c = 4.2 × 103 J ⁄ Kg °C
The formula for specific heat capacity is given as:
c = ΔQ ⁄ m ΔT
Rearrange the formula in terms of ΔT.
ΔT = ΔQ ⁄ c m
= (400 × 103) ⁄ (4.2 × 103 × 40) °C
= 2.38 °C
Hence, the temperature difference is 2.38 °C.


