Practice Questions with Solutions: Structure of the Atom
Multiple Choice Questions
Q1: Maximum number of electrons present in 'N' shell is
(a) 18
(b) 32
(c) 2
(d) 8
Ans: (b)
Explanation: N-shell corresponds to principal quantum number n = 4. The maximum number of electrons that can be accommodated in any shell is given by 2n2. For n = 4, this gives 2 × 42 = 2 × 16 = 32 electrons. Hence option (b) is correct.
Q2: In 1932. J. Chadwick discovered another sub-atomic particle which had no charge and a mass nearly equal to that of a proton. It was eventually named as
(a) proton
(b) neutron
(c) electron
(d) α-particle
Ans: (b)
Explanation: In 1932, J. Chadwick discovered the neutron, a neutral sub-atomic particle with mass nearly equal to that of a proton. Neutrons carry no electrical charge and reside in the nucleus along with protons, so option (b) is correct.
Q3: Which of the following is a property of isotopes?
(a) They have the same number of electrons.
(b) They have different numbers of protons.
(c) They have different chemical properties.
(d) They have the same mass number.
Ans: (a)
Explanation: Isotopes are atoms of the same element, so they have the same number of protons. For neutral atoms, this also means they have the same number of electrons. Because chemical behaviour depends mainly on the electronic structure, isotopes generally show the same chemical properties. However, isotopes differ in the number of neutrons and thus have different mass numbers.
Q4: Valency of oxygen is
(a) 1
(b) 2
(c) 3
(d) 4
Ans: (b)
Explanation: Electronic configuration of oxygen is 2, 6. To achieve a stable octet (8 electrons) in the outer shell, oxygen needs 2 more electrons. Therefore its valency is 2, so option (b) is correct.
Q5: In the nucleus of 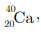 , there are
, there are
(a) 40 protons and 20 electrons
(b) 20 protons and 40 electrons
(c) 20 protons and 20 neutrons
(d) 20 protons and 40 neutrons
Ans: (c)
Explanation: The element shown is calcium with atomic number Z = 20 and mass number A = 40. Number of protons = Z = 20. Number of neutrons = A - Z = 40 - 20 = 20. In a neutral atom, electrons = protons = 20. Thus the nucleus contains 20 protons and 20 neutrons, so option (c) is correct.
Q6: The number of neutrons in the element 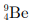 is
is
(a) 4
(b) 5
(c) 9
(d) 13
Ans : (b)
Explanation: Number of neutrons = mass number - atomic number. From the image data A = 9 and Z = 4, so neutrons = 9 - 4 = 5. Hence option (b) is correct.
Q7: Which of the following elements contains only two electrons in the outermost shell?
(a) Helium
(b) Beryllium
(c) Magnesium
(d) All of these
Ans: (d)
Explanation: Helium has two electrons (1s2) and its outermost (and only) shell holds two electrons. Beryllium (2, 2) and magnesium (2, 8, 2) each have two valence electrons in their outermost shell. Therefore all three have two electrons in the outermost shell and option (d) is correct.
Q8: The charge on the atom having 17 protons, 18 electrons is
(a) +1
(b) -1
(c) -2
(d) zero
Ans : (b)
Explanation: The atom has 17 protons (positive charges) and 18 electrons (negative charges). Since electrons outnumber protons by one, the net charge = 17(+1) + 18(-1) = -1. Therefore the atom carries a charge of -1, so option (b) is correct.
Q9: Proton was discovered by
(a) Thomson
(b) Rutherford
(c) Chadwick
(d) Goldstein
Ans: (d)
Explanation: E. Goldstein observed positively charged rays (called canal rays) in gas discharge experiments. These positive particles were later identified as protons. Goldstein's work thus led to the discovery of the proton, so option (d) is correct.
Q10: Which of the following is a property of isotopes?
(a) They have the same number of electrons.
(b) They have different numbers of protons.
(c) They have different chemical properties.
(d) They have the same mass number.
Ans : (a)
Explanation: Isotopes are atoms of the same element, so they have the same number of protons and, for neutral atoms, the same number of electrons. This is why their chemical properties are similar. However, isotopes differ in the number of neutrons and thus have different mass numbers.
Q11: Isotopes have
(a) same physical and chemical properties
(b) same physical properties but different chemical properties
(c) same chemical properties but different physical properties
(d) different physical and chemical properties
Ans: (c)
Explanation: Chemical properties depend mainly on the number and arrangement of electrons; isotopes of an element have the same number of electrons and therefore have similar chemical properties. Physical properties (such as mass, density or boiling point) can vary because isotopes have different mass numbers. Hence isotopes have same chemical but different physical properties, so option (c) is correct.
Q12: The part of an atom where nearly whole mass is concentrated is called
(a) extra-nuclear part
(b) nucleus
(c) atom
(d) neutron
Ans : (b)
Explanation: Nearly all the mass of an atom is concentrated in the nucleus, which contains protons and neutrons. Electrons orbiting the nucleus contribute very little to the total mass.
Q13: Which of the following elements has same number of protons, electrons and neutrons?
(a) Al
(b) Mg
(c) P
(d) Cl
Ans: (b)
Explanation: Magnesium (Mg) is shown as
with atomic number 12 and mass number 24. Number of protons = 12, number of neutrons = 24 - 12 = 12, and for a neutral atom electrons = 12. Thus protons, neutrons and electrons are all equal (=12), so option (b) is correct.
Q14: Atom X and atom Y have similar chemical properties. If the proton number of atom X is 12, What is the likely proton number of atom Y ?
(a) 5
(b) 10
(c) 14
(d) 20
Ans : (d)
Explanation: Atom X has proton number 12, electronic configuration 2, 8, 2, so it has 2 valence electrons. For two atoms to have similar chemical properties they must have the same number of valence electrons. Option (d), proton number 20 (calcium), has electronic configuration 2, 8, 8, 2 and thus also has 2 valence electrons. Therefore the likely proton number of atom Y is 20 and option (d) is correct.
Q15: The ion of an element has 2 positive charge. Mass number of the atom is 24 and the number of neutrons is 12. What is the number of electrons in the ion?
(a) 8
(b) 10
(c) 12
(d) 24
Ans : (b)
Explanation: Given mass number A = 24 and neutrons n = 12, so number of protons p = A - n = 24 - 12 = 12. A neutral atom would have 12 electrons. The ion has a 2+ charge, which means it has lost two electrons. Therefore the number of electrons in the ion = 12 - 2 = 10. Option (b) is correct.
Fill in the blanks
Q16: The subatomic particle not present in a hydrogen atom is .........
Ans : Neutron
In the most common form of hydrogen (protium), the atom contains one proton and one electron and no neutron. (Note: hydrogen has isotopes such as deuterium and tritium that do contain neutrons.)
Q17: Almost all the mass of an atom is concentrated in a small region of space called the .........
Ans : Nucleus
The nucleus is the tiny central region of an atom that contains protons and neutrons. These particles account for almost all of the atom's mass; electrons contribute very little mass in comparison.
Q18: The number of neutrons in the nucleus of an atom can be calculated by ......... the atomic number from its mass number.
Ans : subtracting
The number of neutrons = mass number - atomic number. In words, you find the number of neutrons by subtracting the atomic number from the mass number.
Q19: An atom of an element has 11 protons, 11 electrons and 12 neutrons. The atomic mass of the atom is .........
Ans : 23
Atomic mass (mass number) is the sum of protons and neutrons. Here protons = 11 and neutrons = 12, so mass number = 11 + 12 = 23.
Q20: Cathode rays are a beam of fast moving ..........
Ans : electrons
Cathode rays are streams of fast-moving electrons produced in discharge tubes when a high voltage is applied between the electrodes. These rays travel from the cathode to the anode.
True/False
Q21: α -particles are same thing as helium atoms.
Ans : False
Explanation: An α-particle is the nucleus of a helium atom (two protons and two neutrons) and carries a +2 charge. A neutral helium atom includes two electrons as well. Because an α-particle lacks these electrons it is not the same as a helium atom; it is the helium nucleus.
Q22: An electron has a mass that is much less than a proton.
Ans : True
Explanation: An electron is much lighter than a proton. A proton's mass is roughly 1836 times the mass of an electron, so the statement is true.
Q23: Atoms of an element may have more or less neutrons or electrons than other atoms of the same element.
Ans : True
Explanation: Atoms of the same element always have the same number of protons, but they can have different numbers of neutrons (these are called isotopes). Atoms can also gain or lose electrons to form ions, so the number of electrons can vary. Thus the statement is true.
Q24: Thomson proposed that the nucleus of an atom contains protons and neutrons.
Ans : False
Explanation: J. J. Thomson proposed the plum-pudding model in which electrons were embedded in a positive sphere; he did not propose a dense nucleus. It was Ernest Rutherford who, from scattering experiments, proposed that an atom has a small dense nucleus containing protons (and later neutrons were discovered). Therefore the statement is false.
Q25: There is no particle of matter smaller than an atom.
Ans : False
Explanation: Atoms are made up of smaller particles - protons, neutrons and electrons. Scientific research has also shown still smaller particles (for example quarks and leptons). Therefore it is incorrect to say there are no particles smaller than an atom; the statement is false.
Matching Questions
Direction : In the section, each question has two matching lists. Choices for the correct combination of elements from List-I and List-II are given as options (a), (b), (c) and (d) out of which one is correct.
Q26:
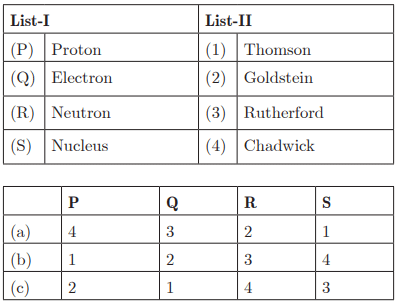
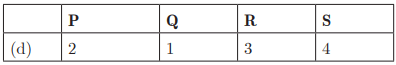
Ans: (c) P - 2, Q - 1, R - 4, S - 3
Q27:
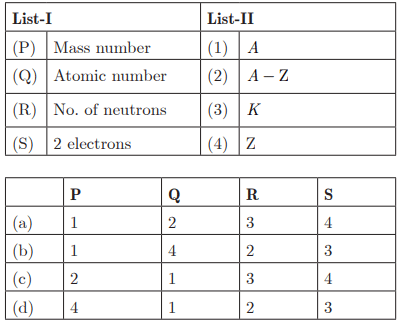
Ans: (b) P - 1, Q - 4, R - 2, S - 3
Q28:
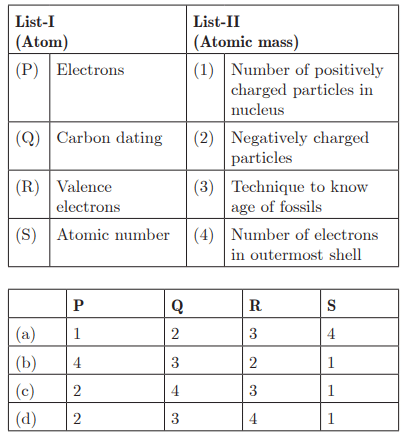
Ans: (d) P - 2, Q - 3, R - 4, S - 1
Q29:
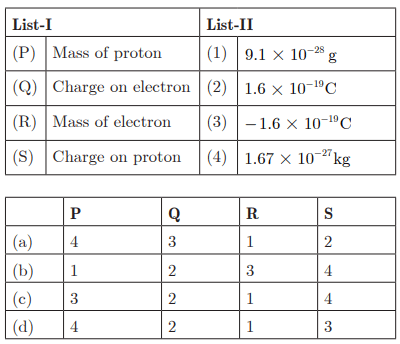
Ans : (a) P - 4, Q - 3, R - 1, S - 2
Q30:
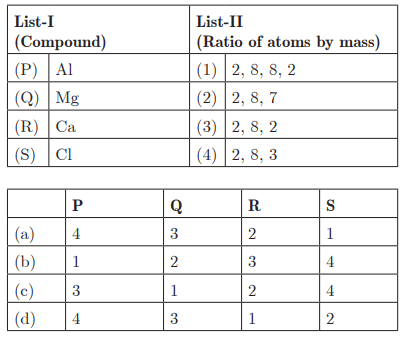
Ans : (d) P - 4, Q - 3, R - 1, S - 2
FAQs on Practice Questions with Solutions: Structure of the Atom
| 1. What are the main components of an atom? |  |
| 2. How is the atomic number of an element defined? |  |
| 3. What is the significance of isotopes in chemistry? |  |
| 4. Can you describe the structure of the nucleus of an atom? |  |
| 5. What role do electrons play in chemical bonding? |  |





