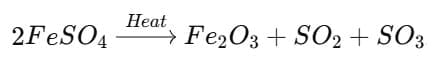Time: 1 hour
M.M. 30
Attempt all questions.
- Question numbers 1 to 5 carry 1 mark each.
- Question numbers 6 to 8 carry 2 marks each.
- Question numbers 9 to 11 carry 3 marks each.
- Question number 12 & 13 carry 5 marks each.
Q1: Which of the following reactions is an endothermic reaction? (1 Mark)
(a) Burning of coal.
(b) Decomposition of vegetable matter into compost.
(c) Process of respiration.
(d) Decomposition of calcium carbonate to form quicklime and carbon dioxide.
Ans: (d)
Explanation: An endothermic reaction is one that absorbs heat from the surroundings. The decomposition of calcium carbonate (CaCO3) to give quicklime (CaO) and carbon dioxide (CO2) requires heat to break bonds and so it absorbs energy. Burning of coal and respiration are exothermic (they release heat). Composting is a slow biological process that is largely exothermic due to microbial activity.
Q2: Which of the following reactions is BOTH a combination reaction and an oxidation reaction? (1 Mark)
(a) CaO + H₂O → Ca(OH)₂
(b) 2Mg + O₂ → 2MgO
(c) Fe + CuSO₄ → FeSO₄ + Cu
(d) NaOH + HCl → NaCl + H₂O
Ans: (b)
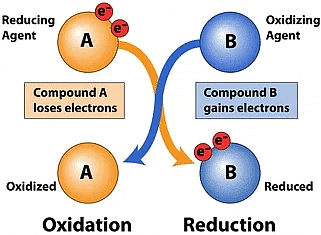
Explanation: In the reaction 2Mg + O₂ → 2MgO, magnesium and oxygen combine to form a single product, magnesium oxide, so it is a combination reaction. At the same time, magnesium gains oxygen, which means magnesium is oxidised. Therefore, this reaction is both a combination reaction and an oxidation reaction, making option (b) the correct answer.
Q3: Why is respiration considered an exothermic reaction even though energy is not released suddenly? (1 Mark)
Ans: Respiration is an exothermic reaction because glucose reacts with oxygen to form carbon dioxide and water while releasing energy. The energy is released slowly in a controlled manner to meet the energy requirements of the body instead of being released suddenly as heat.
Q4: The reaction Fe₂O₃ + 2Al → Al₂O₃ + 2Fe is an example of which type of reaction and why? (1 Mark)
Ans: It is a displacement (redox) reaction because aluminium, being more reactive, displaces iron from iron(III) oxide. Aluminium is oxidised and iron oxide is reduced.
Q5: Why do silver articles become black after some time when exposed to air? (1 Mark)
Ans: Silver articles tarnish because silver reacts with sulphur compounds (present in air as hydrogen sulphide traces) to form silver sulphide (Ag2S), which is black. This surface reaction causes the darkening.
Q6: In the equations given below, state giving reasons, whether substances have been oxidised or reduced. (2 Marks)
(i) PbO + CO → Pb + CO2
(ii) H2S + Cl2 → 2HCl + S
Ans:
(i) In the reaction PbO + CO → Pb + CO2 :
Ans: Carbon monoxide (CO) is oxidised to carbon dioxide (CO2) because it gains oxygen. Lead in lead(II) oxide (PbO) is reduced to metallic lead (Pb) because it loses oxygen (or gains electrons). Thus CO is the reducing agent and PbO is the oxidising agent.
(ii) In the reaction H2S + Cl2 → 2HCl + S :
Ans: Hydrogen sulphide (H2S) is oxidised to sulphur (S) because it loses hydrogen (or loses electrons). Chlorine (Cl2) is reduced to chloride ions in hydrogen chloride (HCl) because each chlorine atom gains an electron (and appears as Cl- in HCl). Therefore H2S is the reducing agent and Cl2 is the oxidising agent.
Q7: Can rancidity retard by storing foods away from the light? (2 Marks)
Ans: Yes. Rancidity of fats and oils mainly occurs by oxidation, a process that is accelerated by light (especially ultraviolet). Storing foods away from light (in dark or opaque containers) slows down the photo-oxidation of fats and thus retards the development of rancidity. Other measures such as refrigeration and using antioxidants also help.
Q8: What are the different ways can make more informative about the chemical equation? (2 Marks)
Ans:
(i) By indicating the physical states of reactants and products, for example (s), (l), (g) or (aq).
(ii) By indicating the heat changes taking place in the reaction, for example Δ (heat added) or writing "exothermic/ endothermic".
(iii) By indicating the conditions under which the reaction takes place, such as catalysts, temperature, pressure or presence of light.
Q9: The action of heat on ferrous sulphate is an example of a decomposition reaction. (3 Marks)
Ans: On heating, ferrous sulphate crystals (FeSO4·7H2O) first lose their water of crystallisation to give anhydrous ferrous sulphate (FeSO4). This causes a change in appearance. On further heating, anhydrous FeSO4 decomposes to form ferric oxide (Fe2O3), sulphur dioxide (SO2) and sulphur trioxide (SO3). The overall decomposition is a chemical breakdown of one substance into simpler substances, so it is a decomposition reaction.
Equation:
2FeSO4 → Δ Fe2O3 + SO2 + SO3
Q10: When a green coloured crystalline salt 'X' is heated strongly in a dry test tube, it first loses water of crystallisation and then decomposes, giving a brown residue and two gases.
(a) Identify the salt 'X'.
(b) Name the two gases evolved during the reaction.
(c) Write the balanced chemical equation for the reaction. (3 Marks)
Ans. (a) Ferrous sulphate crystals, FeSO₄·7H₂O
(b) Sulphur dioxide (SO₂) and sulphur trioxide (SO₃)
(c) 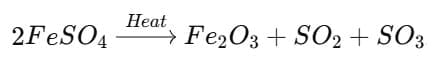
Q11: Write the balanced chemical equations for the following reactions (3 Marks)
(a) Sodium carbonate on reaction with hydrochloric acid in equal molar concentrations gives sodium chloride and sodium hydrogencarbonate.
(b) Sodium hydrogencarbonate on reaction with hydrochloric acid gives sodium chloride, water and liberates carbon dioxide.
(c) Copper sulphate on treatment with potassium iodide precipitates cuprous iodide (CuI), liberates iodine gas and also forms potassium sulphate.
Ans:
(a) Na2CO3 + HCl → NaCl + NaHCO3
(b) NaHCO3 + HCl → NaCl + H2O + CO2
(c) 2CuSO4 + 4KI → 2CuI(s) + 2K2SO4 + I2
Q12: Identify the type of chemical reaction in the following statements and define each of them :
(i) Digestion of food in our body
(ii) Rusting of iron
(iii) Heating of manganese dioxide with aluminum powder.
(iv) Blue color of copper sulphate solution disappears when iron filings are added to it.
(v) Dilute hydrochloric acid is added to sodium hydroxide to form sodium chloride and water. (5 Marks)
Ans. (i) Digestion of food in our body
Type: Decomposition reaction
Definition: Digestion is a chemical process in which complex food substances are broken down into simpler substances (like glucose, amino acids, etc.) with the help of enzymes, so it is a decomposition reaction.(ii) Rusting of iron
Type: Oxidation reaction (corrosion)
Definition: Rusting is an oxidation process in which iron reacts with oxygen in the presence of moisture to form hydrated iron(III) oxide (rust). Here, iron gets oxidised.
(iii) Heating of manganese dioxide with aluminium powder
Type: Displacement (redox) reaction
Definition: In this reaction, aluminium, being more reactive, displaces manganese from manganese dioxide. Aluminium is oxidised and manganese dioxide is reduced.
(iv) Blue colour of copper sulphate solution disappears when iron filings are added to it
Type: Displacement reaction
Definition: Iron displaces copper from copper sulphate solution because iron is more reactive than copper, forming iron sulphate and copper metal.
(v) Dilute hydrochloric acid reacts with sodium hydroxide to form sodium chloride and water
Type: Neutralisation reaction (double displacement)
Definition: A neutralisation reaction is one in which an acid reacts with a base to form salt and water. Here, HCl reacts with NaOH to form NaCl and H₂O.
Q13: Analyse the following reactions and answer the questions given below:
(a) Identify the substances that are oxidised and reduced in the reaction:
Zn + CuSO₄ → ZnSO₄ + Cu
(b) Explain why this reaction does not occur when copper is placed in zinc sulphate solution.
(c) State the role of the reactivity series in predicting such reactions.
(d) Name the type of chemical reaction involved.
(e) Write one more example of a similar reaction. (5 Marks)
Ans:
(a) Zinc is oxidised (loses electrons) and copper(II) ions are reduced (gain electrons).
(b) Copper is less reactive than zinc and therefore cannot displace zinc from its compound.
(c) The reactivity series helps us predict whether a metal can displace another metal from its compound in a chemical reaction.
(d) Displacement reaction (also a redox reaction).
(e) Example:
Fe + CuSO₄ → FeSO₄ + Cu