Long Question Answer: Is Matter Around Us Pure?
Q1: Explain the sublimation process with a labelled diagram.
Ans: Sublimation is the process in which a solid directly changes into the gaseous state without passing through the liquid state, and vice versa. It is a physical and reversible change. Sublimation occurs when a solid has appreciable vapour pressure at a temperature lower than its melting point, so it converts to vapour on heating and reforms as solid on cooling.
Substances that show sublimation: Camphor, ammonium chloride, naphthalene, iodine, dry ice (solid CO2), etc.
Activity to demonstrate sublimation (using ammonium chloride):
Take a small amount of ammonium chloride and crush it to increase the surface area.
Place it in a china dish.
Cover the dish with an inverted funnel.
Plug the narrow stem of the funnel with cotton to prevent the vapour from escaping into the room.
Gently heat the dish and observe.
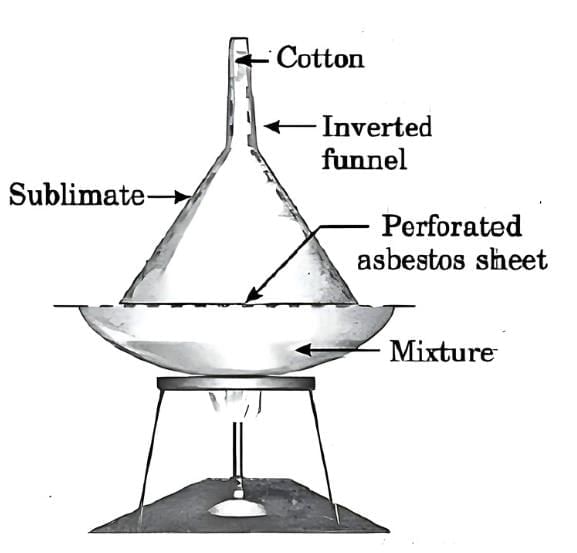
Observation:
White fumes of ammonium chloride rise and deposit as white crystals on the upper inner side of the funnel. The solid changes directly into vapour on heating and then deposits back as solid on the cooler part of the funnel without forming a liquid.
Conclusion:
This confirms that ammonium chloride undergoes sublimation and can be separated from a non-sublimable impurity by this method.
Equation:
NH4Cl (solid) ⇌ NH4Cl (vapour)
Q2: What are colloids? Give their characteristics.
Ans: A colloid is a type of heterogeneous mixture in which tiny particles of one substance (the dispersed phase) are evenly distributed throughout another substance (the dispersion medium). The size of colloidal particles lies between that of true solutions and suspensions; typically it ranges from 1 nm to 1000 nm. These particles are not visible to the naked eye but are large enough to scatter light.
Examples of colloids:
Milk, fog, smoke, butter, jelly, and ink.
Characteristics of colloids:
- Heterogeneous mixture: Colloids appear uniform to the naked eye but consist of two distinct phases on a microscopic scale - the dispersed phase and the dispersion medium.
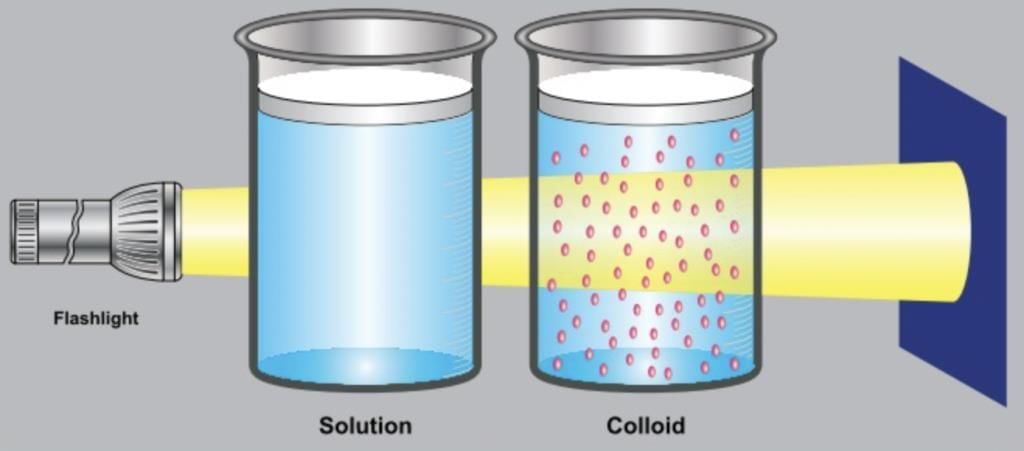
- Tyndall Effect: When a beam of light passes through a colloid, the path of light becomes visible because light is scattered by the colloidal particles. This scattering is called the Tyndall effect and helps to distinguish colloids from true solutions.
- Brownian Motion: Colloidal particles show random, zigzag motion caused by continuous collisions with the molecules of the dispersion medium. This motion helps suspend the particles and prevents settling.
- Stability: Colloidal particles do not settle down on standing, so colloids are relatively stable. Stability often arises from electrical charges on the particles which cause mutual repulsion.
- Cannot be filtered easily: Colloidal particles cannot be separated using ordinary filter paper because they are too small. They can, however, be separated by methods like centrifugation, ultrafiltration, or coagulation.
- Particle size: The particle size is larger than that in a true solution but much smaller than in a suspension, which gives colloids their intermediate behaviour.
Thus, colloids are an important class of mixtures found in everyday life. Their distinct properties such as the Tyndall effect and Brownian motion make them easily recognisable and useful in many applications, for example, in food, medicines and fog sprays.
Q3: How much water should be mixed with 12 ml of alcohol to obtain 12% of alcohol? Calculate.
Ans: Given:
Volume of solute (alcohol) = 12 ml
Desired concentration = 12%
Let the final volume of solution = x ml
Using the formula for concentration:
Concentration (%) = (Volume of solute / Volume of solution) × 100
So, (12 / x) × 100 = 12
Therefore, 12 / x = 12 / 100 = 0.12
Hence, x = 12 ÷ 0.12 = 100 ml
Volume of water required = Final volume - Volume of alcohol = 100 ml - 12 ml = 88 ml
Thus, to obtain a 12% alcohol solution, 88 ml of water should be mixed with 12 ml of alcohol.
Q4: Give the difference between colloidal solutions and suspensions.
Ans:
| Feature | Colloidal Solution | Suspension |
|---|---|---|
| Particle size | 1 nm to 1000 nm | More than 1000 nm |
| Appearance | Appears homogeneous but is heterogeneous | Clearly heterogeneous |
| Visibility of particles | Particles not visible to the naked eye | Particles are visible |
| Stability | Stable, does not settle on standing | Unstable, settles on standing |
| Filtration | Cannot be filtered by filter paper | Can be separated by filtration |
| Tyndall effect | Shows Tyndall effect | May or may not show Tyndall effect |
In summary, the main differences are particle size and stability: colloids have much smaller particles and remain dispersed for long periods, while suspensions contain larger particles that settle on standing and can be separated by ordinary filtration.
Q5: Give the difference between true solutions and colloidal solutions.
Ans:
| Feature | True Solution | Colloidal Solution |
|---|---|---|
| Particle size | Less than 1 nm | Between 1 nm and 1000 nm |
| Appearance | Homogeneous | Appears homogeneous but is heterogeneous |
| Visibility of particles | Particles not visible | Particles not visible but scatter light |
| Stability | Very stable | Relatively stable |
| Tyndall effect | Does not show | Shows Tyndall effect |
| Filtration | Cannot be filtered | Cannot be filtered with normal filter paper |
To conclude, the key distinction is that true solutions have extremely small particles and do not scatter light, whereas colloidal solutions contain larger particles that scatter light (showing the Tyndall effect) and behave differently from true solutions in separation techniques.
FAQs on Long Question Answer: Is Matter Around Us Pure?
| 1. What is meant by 'pure matter'? |  |
| 2. How can we determine if a substance is pure? |  |
| 3. What are the different types of mixtures? |  |
| 4. Why is it important to understand the purity of matter? |  |
| 5. Can pure substances be separated into simpler substances? |  |





