Important Diagrams: Chemical Reactions & Equations
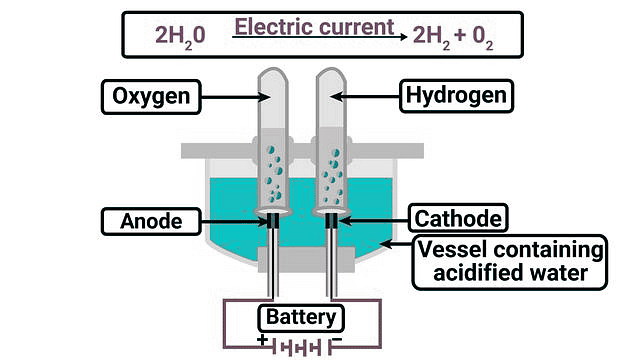
Electrolytic Decomposition
Definition: Electrolytic decomposition is a decomposition reaction in which a compound breaks down into simpler substances when electric current is passed through it.
Diagram Description: The diagram shows electrolysis of water using a battery and electrodes dipped in acidulated water. When electric current is passed, hydrogen gas is collected at the cathode and oxygen gas at the anode. This shows decomposition of water into its elements.
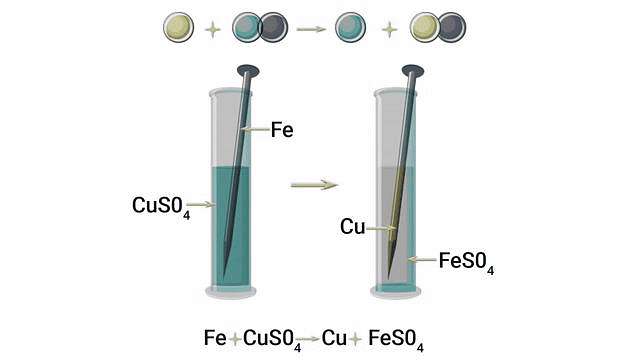
Displacement Reaction
Definition: A displacement reaction is a chemical reaction in which a more reactive element displaces a less reactive element from its compound.
Diagram Description: The diagram shows an iron nail dipped in copper sulphate solution. After some time, copper gets deposited on the iron and the blue solution turns green. This shows iron displacing copper from copper sulphate.
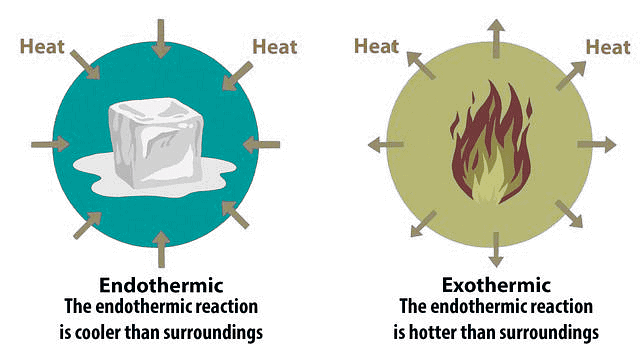
Exothermic and Endothermic Reaction
Definition: Exothermic reaction: A reaction in which heat is released. Endothermic reaction: A reaction in which heat is absorbed.
Diagram Description: The diagram shows heat flow. In an exothermic reaction, heat is released to the surroundings (arrows going out). In an endothermic reaction, heat is absorbed from the surroundings (arrows coming in).
Rancidity
Definition: Rancidity is the process in which fats and oils get oxidised, producing unpleasant smell and taste.
Diagram Description: The diagram shows food (like apples) getting spoiled over time due to exposure to air. This represents oxidation of fats and oils, leading to bad smell and taste.

FAQs on Important Diagrams: Chemical Reactions & Equations
| 1. What are the main types of chemical reactions shown in diagrams for Class 10 CBSE? |  |
| 2. How do I read and interpret chemical equation diagrams correctly? |  |
| 3. Why do some chemical reactions produce heat or light in the diagrams? |  |
| 4. What's the difference between balanced and unbalanced chemical equations in diagram form? |  |
| 5. How can I use chemical reaction diagrams to prepare better for CBSE exams? |  |






