HOTS Questions: Structure of the Atom
Q1: Both helium (He) and beryllium (Be) have two valence electrons. Whereas He represents a noble gas element, Be does not. Assign reason.
Ans: The element He (Z = 2) has two electrons present in the only shell, the K-shell. Since this shell can hold a maximum of two electrons only, the K-shell of helium is complete and the atom is stable; hence He is a noble gas.
In beryllium (Z = 4) the electronic configuration is 2, 2. Although the outer (L) shell has two electrons, it is not full - the L-shell can accommodate up to eight electrons. Because Be does not have a complete outer shell, it does not show the inert (noble gas) behaviour of helium and is chemically reactive compared with He.
Q2: Which of the two will be chemically more reactive ; element X with atomic number 17 or element Y with atomic number 16 ?
Ans: The electronic configurations of the two elements are:
X (Z = 17): K (2), L (8), M (7).
Y (Z = 16): K (2), L (8), M (6).
The element X needs only one electron in the outermost shell to attain the noble gas configuration of argon (Ar, Z = 18), whereas Y needs two electrons. Therefore X has a greater tendency to gain one electron and is chemically more reactive (in terms of gaining an electron) than Y.
Q3: An atom of an element has three electrons in the third shell which is the outermost shell. Write
- the electronic configuration
- the atomic number
- number of protons
- valency
- the name of the element
- its nature whether metal or non-metal.
Ans: The third shell is the M shell. If the atom has three electrons in the M shell, the K and L shells must already be filled.
- Electronic configuration : 2, 8, 3.
- Atomic number = Total number of electrons = 13.
- Number of protons = Atomic number = 13.
- Valency = 3 (the atom has three valence electrons and can lose these three to attain the nearest noble gas configuration, or it may share them in covalent bonding).
- The element with Z = 13 is aluminium (Al).
- Nature: It is a metal.
Q4: Study the data given below and answer the questions which follow :
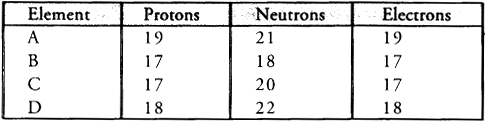
- Write the mass number and atomic number of the particles A, B, C and D.
- Which represent a pair of isotopes ?
Ans:
- Particle A : Mass number = 7 ; Atomic number = 3.
- Particle B : Mass number = 17 ; Atomic number = 9.
- Particle C : Mass number = 16 ; Atomic number = 8.
- Particle D : Mass number = 18 ; Atomic number = 8.
Particles C and D represent a pair of isotopes because they have the same atomic number (8) but different mass numbers (16 and 18).
Q5: The number of protons, neutrons and electrons in particles from A to E are given below:

- Which one is a cation ?
- Which one is an anion ?
- Which represent pair of isotopes ?
Ans:
- B is a monovalent cation (B+) - it has one more proton than electrons, so it carries a positive charge.
- E is a monovalent anion (E-) - it has one more electron than protons, so it carries a negative charge.
- A and D represent a pair of isotopes - they have the same number of protons (same atomic number) but different numbers of neutrons, hence different mass numbers.
Q6: One electron is present in the outermost shell of the atom of an element 'Z'.
(a) What will be the nature of this element?
(b) What will be the value of charge of the ion formed, if this electron is removed from the outermost shell?
Ans:
(a) Element 'Z' will be a metal. An atom with a single electron in its outermost shell is typically electropositive and tends to lose that electron to form a positively charged ion.
(b) On removal of one electron, the atom will form a singly charged positive ion, Z → Z+ + e-. The ion has a charge of +1.
Q7: An atom 'M' of an element reacts with oxygen to form M2O3. Calculate the valency of the element 'M'.
Ans: Two atoms of element M combine with three atoms of oxygen to form M2O3.
Valency of oxygen = 2. Total negative charge contributed by 3 oxygen atoms = 3 × 2 = 6 (in terms of valence units).
This must be balanced by the total positive valency from 2 atoms of M. Let valency of M = x.
Therefore, 2 × x = 6 ⟹ x = 3.
Hence, the valency of element M is 3.
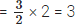
Q8: Explain why chlorine, whether as the element or its compounds, always has relative atomic mass of about 35.5.
Ans: The relative atomic mass of an element is the weighted average of the masses of its naturally occurring isotopes, taking their abundances into account.
Natural chlorine exists mainly as two isotopes: 35Cl and 37Cl. Their approximate natural abundances are about 75% for 35Cl and 25% for 37Cl.
Relative atomic mass = (fraction of 35Cl × 35) + (fraction of 37Cl × 37).
= (0.75 × 35) + (0.25 × 37) = 26.25 + 9.25 = 35.5 u.
Therefore, the relative atomic mass of chlorine is about 35.5 u.
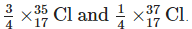
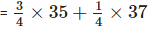
Q9: How many electrons will weigh 1 g?
Ans: Mass of one electron = 9.11 × 10-31 kg.
Mass of 1 g = 1 × 10-3 kg.
Number of electrons in 1 g = (1 × 10-3 kg) ÷ (9.11 × 10-31 kg per electron).
= 1 × 10-3 / 9.11 × 10-31 ≈ 1.098 × 1027 electrons.
Therefore, about 1.10 × 1027 electrons weigh 1 g.
Q10: Composition of the nuclei of two atomic species 'X' and 'Y' are given below:

Give the mass number of 'X' and 'Y'. What is the relationship between the two species?
Ans: (i) Mass number of X = Number of protons + Number of neutrons = 8 + 8 = 16 u.
(ii) Mass number of Y = Number of protons + Number of neutrons = 8 + 10 = 18 u.
Relationship: Both X and Y have the same atomic number (8 protons) but different mass numbers. Therefore, X and Y are isotopes of the same element.
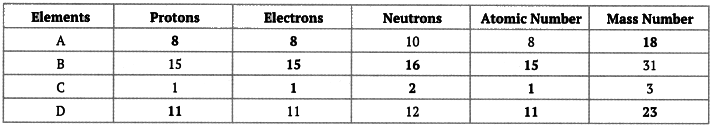
Q11: Complete the following gaps in the given table:

Ans: We know the following relationships:
- Number of protons = Atomic number.
- Number of electrons = Number of protons (for a neutral atom).
- Mass number = Number of protons + Number of neutrons.
Using these relationships, the blanks in the table can be filled by calculating the missing quantity from the others given.
Q12: An element 'X' has mass number 4 and atomic number 2. Write the valency of this element. Will it react with other atoms of different elements?
Ans: Atomic number 2 means the atom has two electrons; its electronic configuration is 2 (K-shell full). The valency is zero because the outer shell is complete and the atom is already stable. Such an atom (helium, He) does not react readily with other atoms; it is inert and shows almost no tendency to form chemical bonds.
FAQs on HOTS Questions: Structure of the Atom
| 1. What are the main components of an atom? |  |
| 2. How do protons and neutrons contribute to the atomic mass? |  |
| 3. What role do electrons play in chemical bonding? |  |
| 4. How did the discovery of the electron change our understanding of atomic structure? |  |
| 5. What is the significance of isotopes in understanding atomic structure? |  |





