Worksheet Solutions: Solids, Liquids & Gases - 2
Q1: Fill in the blanks.
(i) All matter has _____ and _____.
Ans: mass, takes up space
All matter has mass, which is the amount of matter in an object, and takes up space (volume), meaning every object occupies some space.
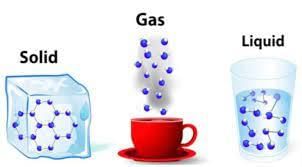
(ii) Matter is made up of tiny particles called _____.
Ans: molecules
Matter is made of tiny particles called molecules. Each molecule is made of even smaller parts called atoms.
(iii) The three states of matter are _____, _____, and _____.
Ans: solid, liquid, gas
The three states of matter are solid, liquid and gas. Each state has different properties because the particles are arranged and move differently.
(iv) Liquids take the shape of the _____ they are poured into.
Ans: container
Liquids do not have a fixed shape; they take the shape of the container they are poured into while keeping the same volume.
(v) Gases do not have a definite shape or _____.
Ans: volume
Gases have neither a definite shape nor a definite volume. Their particles are far apart and they spread to fill the space available.
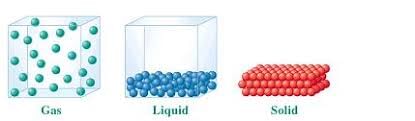
Q2: Match the column.
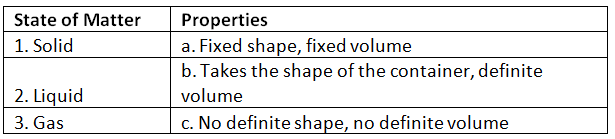
Ans: 1. Solid - Fixed shape, fixed volume
Solids keep their shape and volume because their particles are held closely together.
2. Liquid - Takes the shape of the container, definite volume
Liquids take the shape of the container they are in but keep the same amount of space (volume).
3. Gas - No definite shape, no definite volume
Gases do not have a fixed shape or volume; they spread out to fill any space available.
Q3: True or False.
(i) Solids have a definite shape and volume.
Ans: True
Explanation: Solids keep their own shape and volume because their particles are tightly packed and do not move freely.
(ii) Gases are often visible.
Ans: False
Explanation: Gases are usually invisible; we cannot see the air around us even though it is a gas.
(iii) Melting is the process of changing a liquid to a solid.
Ans: False
Explanation: Melting is when a solid turns into a liquid on heating (for example, ice melting to water).
(iv) Water can exist in all three states: solid, liquid, and gas.
Ans: True
Explanation: Water can be ice (solid), liquid water, or water vapour (gas) depending on the temperature.
(v) Soluble substances dissolve in water.
Ans: True
Explanation: Substances that dissolve in water, like sugar or salt, are called soluble; others, like tea leaves, do not dissolve and are insoluble.

Q4: Multiple Choice Questions (MCQs).
(i) What is the state of matter of water at room temperature?
(a) Solid
(b) Liquid
(c) Gas
(d) None of these
Ans: (b)
Explanation: At room temperature, water is a liquid. Its particles can move past each other but stay close together, so it flows and takes the shape of its container.
(ii) Which of the following substances is a solute?
(a) Water
(b) Salt
(c) Coffee
(d) None of these
Ans: (b)
Explanation: A solute is the substance that dissolves in a solvent. Salt is a common solute that dissolves in water to make a salt solution.

(iii) What happens to water when it is boiled in a kettle?
(a) It freezes into ice
(b) It evaporates into steam
(c) It turns into a solid
(d) None of these
Ans: (b)
Explanation: When water is boiled, it changes into steam (water vapour), which is the gas form of water and leaves the kettle as bubbles and vapour.
(iv) How can insoluble solids be separated from liquids?
(a) Filtration
(b) Evaporation
(c) Condensation
(d) None of these
Ans: (a)
Explanation: Filtration separates insoluble solids from liquids because the liquid passes through the filter while the solid is left behind on the filter paper.
(v) Which of the following is NOT a property of gases?
(a) Definite shape
(b) No definite volume
(c) Often invisible
(d) None of these
Ans: (a)
Explanation: Gases do not have a definite shape; they spread out to take the shape of the space available. So having a definite shape is not a property of gases.
Q5: Short Answer Questions.
(i) Explain the three states of matter with examples.
Ans: The three states of matter are solid, liquid and gas.
- Solids: Solids have a fixed shape and fixed volume. Examples: a pencil, a slice of bread, a piece of wood.
- Liquids: Liquids take the shape of the container they are in and have a definite volume. Examples: water, juice, milk.
- Gases: Gases have no definite shape or volume and often cannot be seen. Examples: the air we breathe, the gas inside a balloon.
(ii) Describe the process of evaporation.
Ans: Evaporation is when a liquid changes into a gas upon heating. When water is heated, the heat energy provided to it overcomes the forces of attraction between the water molecules, allowing them to escape into the air as gas. For example, water in a kettle boils and produces steam, which is water in the gas form.
(iii) What is a solution? Give an example.
Ans: A solution is a uniform mixture formed when a solute dissolves in a solvent. The solute is the substance that dissolves and the solvent is the liquid that does the dissolving. Example: salt (solute) dissolved in water (solvent) makes a saltwater solution.
(iv) How can insoluble solids be separated from liquids?
Ans: Insoluble solids can be separated from liquids by two common methods:
- Filtration: Pour the mixture through filter paper or a sieve. The liquid passes through while the solid particles are left on the filter , resulting in the separation of the insoluble solid from the liquid.
- Sedimentation and decantation: Let the mixture stand for some time so the solid settles at the bottom due to gravity. Then carefully pour the clear liquid away without disturbing the settled solid.
FAQs on Worksheet Solutions: Solids, Liquids & Gases - 2
| 1. What are the three states of matter? |  |
| 2. What is the main difference between solids and liquids? |  |
| 3. How do solids, liquids, and gases differ in terms of particles? |  |
| 4. Can matter change from one state to another? Give an example. |  |
| 5. How does temperature affect the states of matter? |  |





