Case Based Question Answer: Atoms and Molecules
(I) Read the following passage and answer the questions based on the passage and related studied concepts.
Chemical reactions follow laws of chemical combination such as law of conservation of mass, law of constant (definite) proportion. Atom is smallest particle of an element that retains all its chemical properties and takes part in chemical reaction. Molecule is made of elements or compounds, capable of independent existence. It shows all properties of substance. A chemical formula of compound show its elements and number of atoms of each element. Cluster of atoms act as polyatomic ions having fixed charge on them and value helps to decide chemical formula. Atoms of C-12 are assigned relative atomic mass-12 and relative mass of all other atoms are determined with the help of C-12. The Avogadro's number 6.022 × 1023 is defined as the number of atoms in exactly 12 g of carbon 12. Mole is amount of substance that contains same number of atom as 12 g of C-12. Mass of 1 mole of substance is called its molar mass.
Q1: Calculate the percentage of oxygen in CO2. [Atomic mass of C = 12 u, O = 16 u]
Ans: Molar mass of CO2 = 12 + (16 × 2) = 44 u.
Mass of oxygen in one mole of CO2 = 16 × 2 = 32 u.
% of oxygen = (32 ÷ 44) × 100 = 72.727...% ≈ 72.73%.
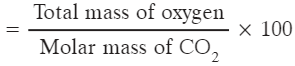
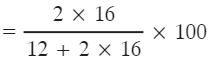
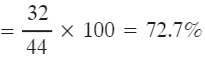
Q2: How many mole of atoms are present in 10 g of calcium atom? [Ca = 44u]
Ans: Number of moles = mass ÷ molar mass = 10 g ÷ 44 g mol-1 = 0.2273 mol (to four significant figures).
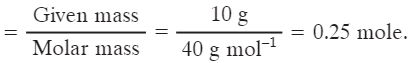
Number of atoms = Number of moles × 6.022 × 1023 = 0.2273 × 6.022 × 1023 = 1.37 × 1023 atoms.
Q3: 0.25 mole of an element 'X' is 9.75 g. What is X ?
Ans: 0.25 mole of X = 9.75 g.
1 mole of X = 9.75 ÷ 0.25 = 39.0 g mol-1.
The element with atomic mass about 39 is Potassium (K).
Q4: What is empirical formula of a compound with composition 80% copper and 20% sulphur. [At. mass of Cu = 64 u, S = 32 u]
Ans:
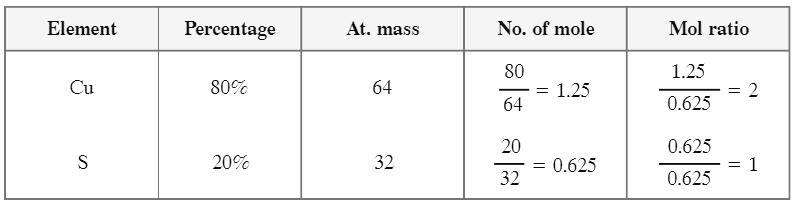
Assume 100 g of the compound: Cu = 80 g and S = 20 g.
Moles of Cu = 80 ÷ 64 = 1.25; Moles of S = 20 ÷ 32 = 0.625.
Ratio Cu : S = 1.25 : 0.625 = 2 : 1.
The empirical formula of the compound is Cu2S.
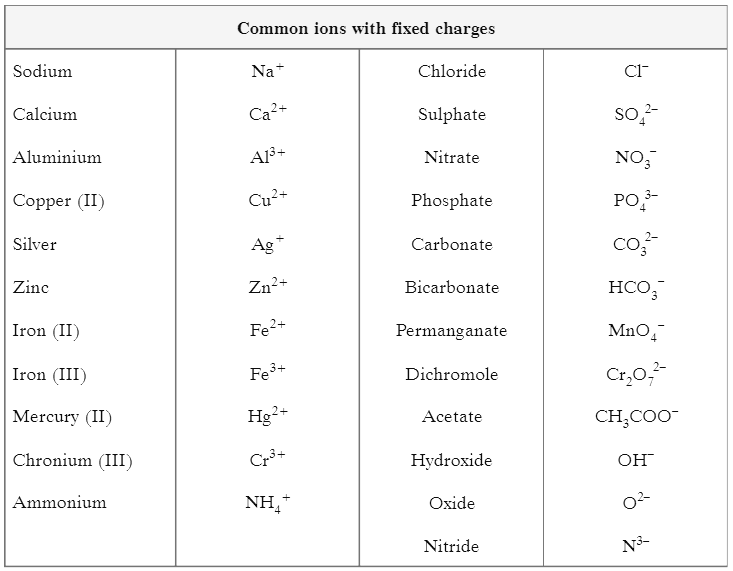
(II) Table shows common ions with fixed charges Answer the questions based on this table and related studied concepts.
Q1: What is formula of magnesium nitride?
Ans:
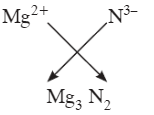
Magnesium forms Mg2+ and nitride is N3-.
To balance charges, 3 Mg2+ (total +6) combine with 2 N3- (total -6).
Formula: Mg3N2.
Q2: Calculate the molar mass of Iron (II) Sulphate. [Fe = 56 u, S = 32 u, O = 16 u]
Ans:
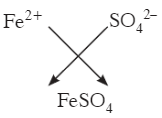
Molar mass of FeSO4 = Fe + S + (O × 4) = 56 + 32 + (16 × 4).
16 × 4 = 64; 56 + 32 + 64 = 152 g mol-1.
Q3: Write name of (NH4)2SO4
Ans: Ammonium sulphate.
Q4: Give one example of polyatomic anion.
Ans: CO32- (Carbonate).
FAQs on Case Based Question Answer: Atoms and Molecules
| 1. What are atoms and how do they relate to molecules? |  |
| 2. What is the difference between a compound and a mixture? |  |
| 3. How are atomic and molecular masses calculated? |  |
| 4. What are the different types of chemical bonds? |  |
| 5. Why is the concept of the mole important in chemistry? |  |





