JEE Exam > JEE Notes > Chemistry Main & Advanced > Mind Map: Isomerism
Mind Map: Isomerism
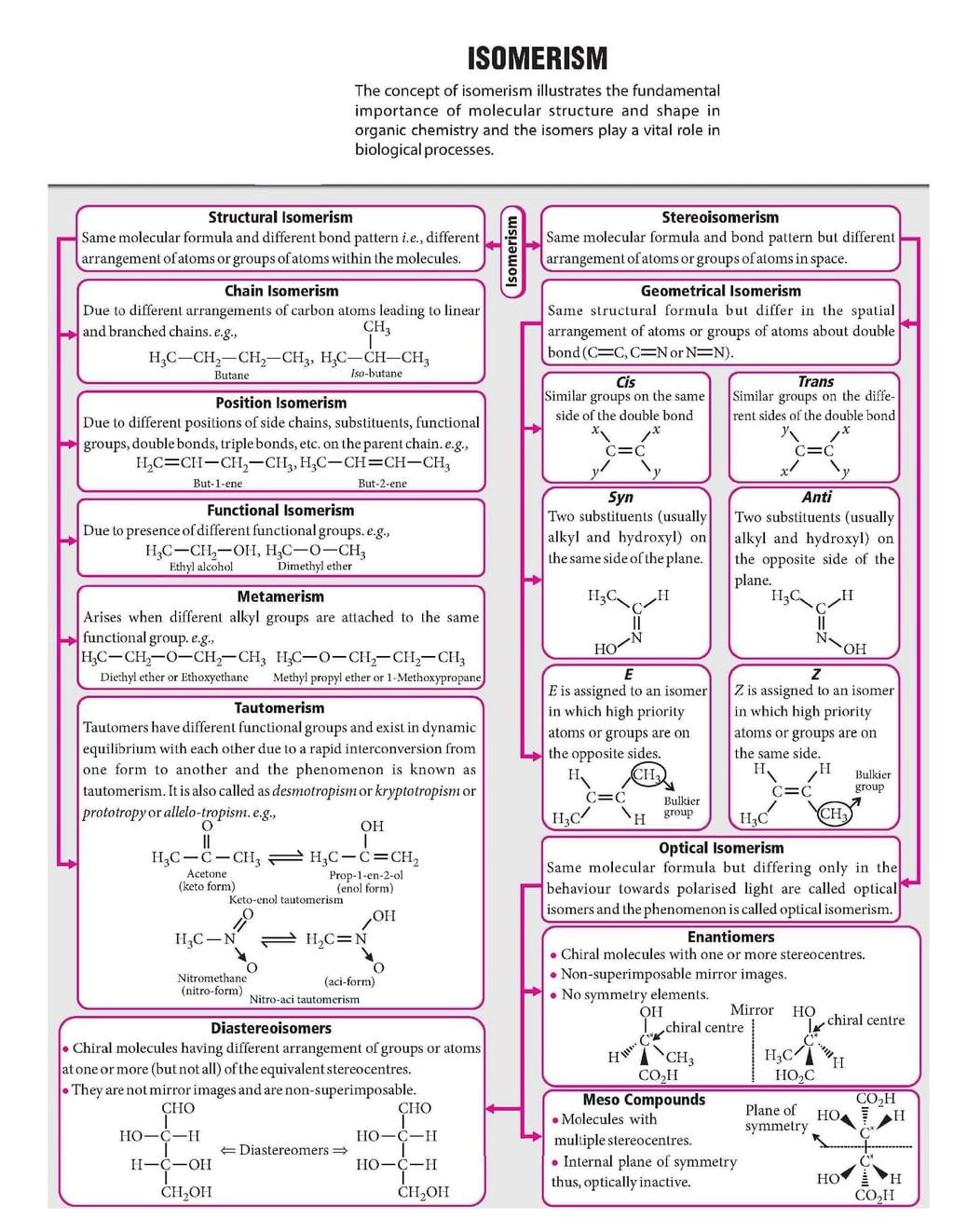
The document Mind Map: Isomerism is a part of the JEE Course Chemistry for JEE Main & Advanced.
All you need of JEE at this link: JEE
FAQs on Mind Map: Isomerism
| 1. What's the difference between structural isomers and stereoisomers in organic chemistry? |  |
Ans. Structural isomers have identical molecular formulas but different atom arrangements, while stereoisomers have the same atom connectivity but differ in spatial orientation. Structural isomers include chain, position, and functional group variants. Stereoisomers encompass geometric (cis-trans) and optical isomerism, where atoms occupy different three-dimensional positions. Both types are crucial for understanding molecular behaviour in JEE Chemistry.
| 2. How do I identify optical isomerism and what's the deal with chiral centres? |  |
Ans. Optical isomers are non-superimposable mirror images produced by chiral centres-atoms bonded to four different groups. Molecules with chiral centres rotate plane-polarized light differently (dextrorotatory or laevorotatory). To identify them, look for carbon atoms with four distinct substituents. Stereoisomerism through chirality is essential for JEE Main and Advanced problem-solving involving enantiomers and racemic mixtures.
| 3. What's the difference between cis-trans isomerism and E-Z nomenclature? |  |
Ans. Cis-trans isomerism describes geometric arrangement around double bonds or rings (cis = same side, trans = opposite side). E-Z nomenclature uses priority rules for substituents when cis-trans fails. E-Z systematically names stereoisomers regardless of position, making it more reliable for complex molecules. Understanding both classification systems strengthens answers in JEE examinations involving unsaturated compounds.
| 4. Can a molecule have both structural and stereoisomers at the same time? |  |
Ans. Yes-compounds can simultaneously exhibit structural isomerism (different atom arrangements) and stereoisomerism (different spatial configurations). For example, butene has position isomers (1-butene vs 2-butene), and 2-butene itself shows geometric isomerism. This dual classification occurs when molecular formulas remain identical but both connectivity and three-dimensional orientation differ, complicating structure-property relationships.
| 5. Why do geometric isomers have different physical properties despite the same molecular formula? |  |
Ans. Geometric isomers differ in spatial arrangement, causing variations in dipole moments, boiling points, and intermolecular forces. Cis-isomers are typically polar with higher boiling points due to stronger dipole-dipole interactions, while trans-isomers are less polar with lower boiling points. These property differences arise from distinct molecular geometry, making stereoisomerism critical for predicting reactivity and behaviour in JEE Chemistry problems.
Related Searches
Mind Map: Isomerism, Summary, practice quizzes, study material, Important questions, pdf , shortcuts and tricks, Extra Questions, Previous Year Questions with Solutions, ppt, Mind Map: Isomerism, Mind Map: Isomerism, Free, mock tests for examination, Sample Paper, Semester Notes, past year papers, MCQs, Exam, Objective type Questions, Viva Questions, video lectures;





