IGCSE Class 10 > Class 10 Notes > Biology for GCSE/ > Enzymes & pH
Enzymes & pH
Enzymes & pH: Extended
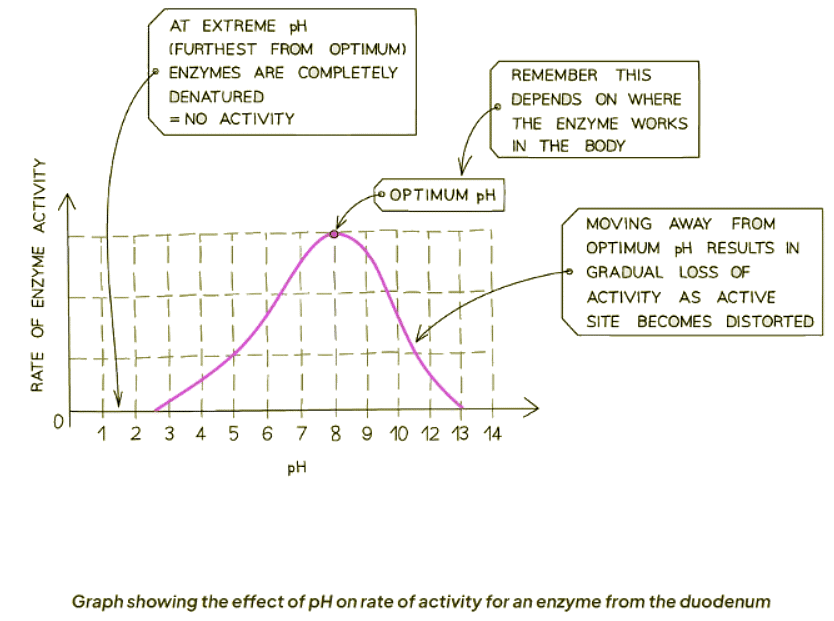
- Enzymes typically function optimally at a pH of 7, though exceptions exist based on their origin. For instance, enzymes from acidic environments like the stomach operate best at lower pH levels (around 2), while those from alkaline environments like the duodenum prefer higher pH levels (around 8 or 9).
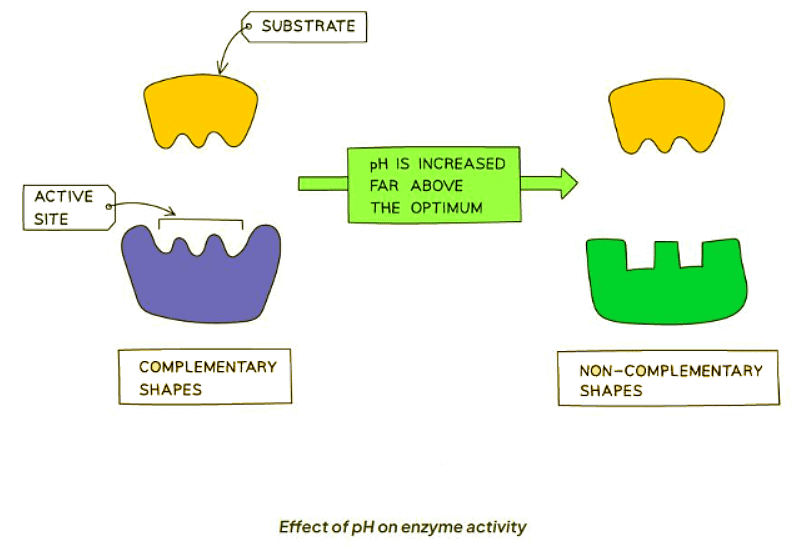
- pH levels significantly diverging from the optimal range can disrupt the bonds stabilizing the protein's amino acid chain.
- This disruption alters the active site's shape, rendering it incompatible with the substrate, thus diminishing enzymatic activity.
- Extremes in pH can lead to denaturation, where the enzyme loses its native conformation and ceases to function.
MULTIPLE CHOICE QUESTIONTry yourself: At what pH level do enzymes from acidic environments like the stomach operate optimally?
The document Enzymes & pH is a part of the Class 10 Course Biology for GCSE/IGCSE.
All you need of Class 10 at this link: Class 10
FAQs on Enzymes & pH
| 1. How does pH affect enzyme activity? |  |
Ans. Enzymes have an optimal pH at which they function most effectively. Changes in pH can alter the shape of the enzyme's active site, affecting its ability to bind with the substrate and catalyze the reaction. Extreme pH levels can denature the enzyme, rendering it inactive.
| 2. What happens to enzyme activity if the pH is too high or too low? |  |
Ans. If the pH is too high or too low, the enzyme may become denatured, leading to a loss of its three-dimensional structure and functionality. This can result in a decrease or complete loss of enzyme activity, as the active site is no longer able to interact with the substrate.
| 3. Can enzymes work in any pH environment? |  |
Ans. Enzymes have specific pH ranges in which they function optimally. While some enzymes may be able to tolerate slight fluctuations in pH, extreme pH levels can disrupt their structure and activity. Therefore, enzymes typically work best within a specific pH range.
| 4. How can pH be controlled in a laboratory setting to optimize enzyme activity? |  |
Ans. In a laboratory setting, pH can be controlled by using buffer solutions that resist changes in pH. By adjusting the pH of the reaction environment to match the optimal pH for the enzyme being used, the enzyme's activity can be optimized for the desired reaction.
| 5. What are some examples of enzymes that are sensitive to pH changes? |  |
Ans. Examples of enzymes that are sensitive to pH changes include pepsin, which functions best in the acidic environment of the stomach, and trypsin, which works optimally in the slightly alkaline environment of the small intestine. Changes in pH can significantly impact the activity of these enzymes.
Related Searches
MCQs, ppt, Semester Notes, Enzymes & pH, Summary, Important questions, mock tests for examination, study material, pdf , Exam, Viva Questions, Previous Year Questions with Solutions, Enzymes & pH, Extra Questions, practice quizzes, Enzymes & pH, Free, past year papers, video lectures, shortcuts and tricks, Objective type Questions, Sample Paper;





