IGCSE Year 10 > Year 10 Notes > Chemistry for GCSE/ > Proton Transfer, Strong and Weak Acid
Proton Transfer, Strong and Weak Acid
Proton transfer
- The previous definitions of acids and bases can be expanded upon. When we consider proton transfer, we delve deeper into how each substance interacts with protons.
- In the context of proton transfer, we can elaborate on how substances behave regarding proton interactions.
Acids
- Acids act as proton donors by ionizing in a solution, releasing protons (H⁺ ions). These H⁺ ions contribute to the acidic nature of the solution.
- Proton donors supply H⁺ ions, thereby increasing the acidity of the solution.
Bases
- Bases function as proton acceptors; they receive protons donated by acids.
- Proton acceptors take in protons provided by acids, playing a crucial role in chemical reactions.
- Diagram showing the role of acids and bases in the transfer of protons - here water acts as a base as it accepts a proton:
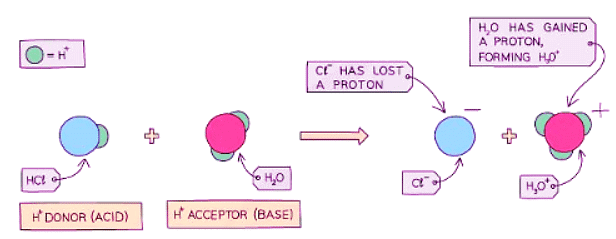
Strong Acids
- Acids can be categorized as strong or weak based on the number of H+ ions they release in water.
- Strong acids completely dissociate in water, resulting in solutions with very low pH levels.
- Examples of strong acids are hydrochloric acid (HCl) and sulfuric acid (H2SO4).
- For instance, when hydrochloric acid dissolves in water, it ionizes into H+ and Cl- ions: HCl (aq) → H+ (aq) + Cl- (aq).
Weak Acids
- Weak acids only partially dissociate in water, leading to pH values closer to the middle of the pH scale, below 7.
- Organic acids like ethanoic acid (CH3COOH) are examples of weak acids.
- When weak acids are added to water, an equilibrium is established between the molecules and their ions.
- For example, propanoic acid undergoes partial ionization in water: CH3CH2COOH ⇌ H+ + CH3CH2COO-.
- The equilibrium favors the left side, indicating a high concentration of intact acid molecules with a low concentration of H+ ions in the solution.
Effect of Concentration on Strong and Weak Acids
- A concentrated acid solution has a higher number of acid molecules per dm3 of solution. It does not necessarily denote strength, as it could be from a weak acid that doesn't fully dissociate.
- For instance, a dilute HCl solution can be more acidic than a concentrated ethanoic acid solution, as most HCl molecules dissociate compared to CH3COOH.
MULTIPLE CHOICE QUESTIONTry yourself: What is the role of bases in proton transfer?
The document Proton Transfer, Strong and Weak Acid is a part of the Year 10 Course Chemistry for GCSE/IGCSE.
All you need of Year 10 at this link: Year 10
FAQs on Proton Transfer, Strong and Weak Acid
| 1. What is the difference between a strong acid and a weak acid? |  |
Ans. A strong acid completely dissociates in water to release all its protons, while a weak acid only partially dissociates, resulting in fewer protons being released.
| 2. How does proton transfer occur in acids? |  |
Ans. Proton transfer in acids involves the transfer of a hydrogen ion (proton) from the acid to a base, forming the conjugate base of the acid and the conjugate acid of the base.
| 3. Can you provide an example of a strong acid and a weak acid? |  |
Ans. Hydrochloric acid (HCl) is an example of a strong acid, while acetic acid (CH3COOH) is a weak acid.
| 4. Why are strong acids considered to be more corrosive than weak acids? |  |
Ans. Strong acids release more protons, leading to a higher concentration of H+ ions in solution, which can cause more severe chemical burns and corrosion compared to weak acids.
| 5. How does the equilibrium expression change for a weak acid compared to a strong acid? |  |
Ans. For a weak acid, the equilibrium expression includes both the dissociation of the acid and the recombination of the ions formed, while for a strong acid, the equilibrium expression only considers the dissociation of the acid.
Related Searches
Objective type Questions, pdf , ppt, Previous Year Questions with Solutions, shortcuts and tricks, Proton Transfer, mock tests for examination, MCQs, Important questions, Exam, practice quizzes, Proton Transfer, Free, past year papers, video lectures, Semester Notes, Sample Paper, Viva Questions, study material, Summary, Extra Questions, Strong and Weak Acid, Strong and Weak Acid, Proton Transfer, Strong and Weak Acid;






