IGCSE Year 10 > Year 10 Notes > Chemistry for GCSE/ > Preparation of Insoluble Salts
Preparation of Insoluble Salts
Preparing Insoluble Salts
- Insoluble salts are formed using a precipitation reaction.
- The solid salt obtained is known as the precipitate. To ensure success, the solid salt formed must be insoluble in water, and the reactants must be soluble.
When two soluble reactants are used, a precipitation reaction occurs. The diagram below illustrates the filtration process to separate the precipitate:
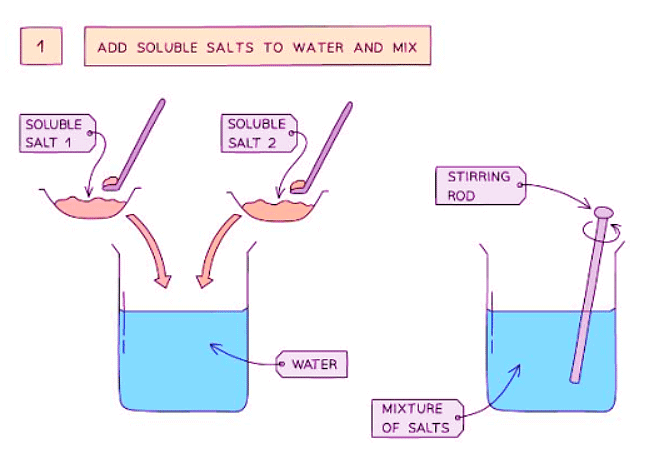
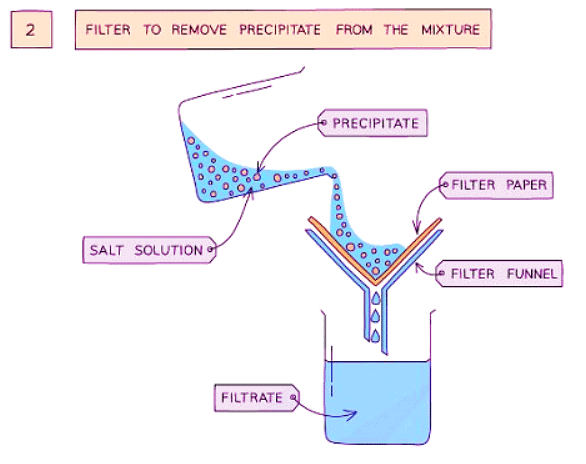
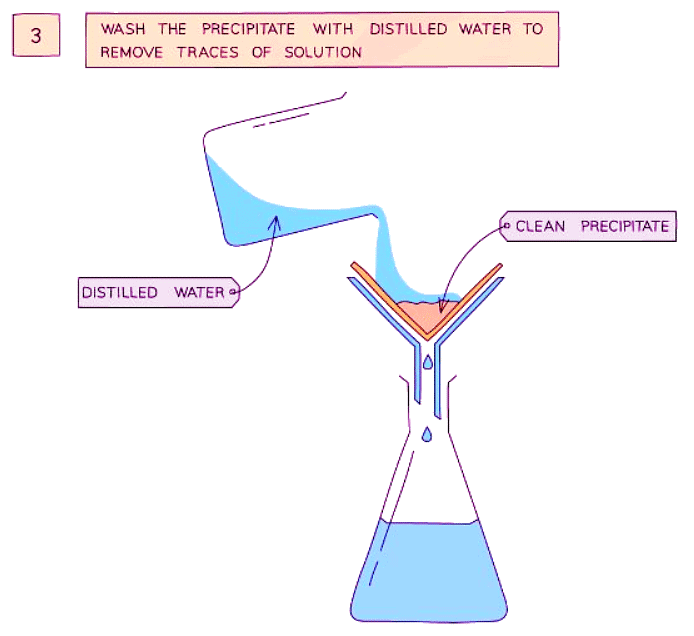
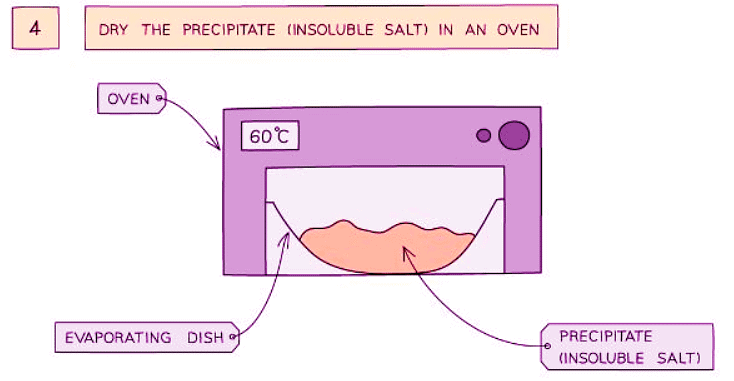
Method
Follow these steps to prepare insoluble salts:
- Mix two soluble reactants in a solution.
- A precipitation reaction will occur, forming an insoluble salt as a solid.
- Filter the mixture to separate the precipitate from the solution.
- Collect and dry the precipitate to obtain the insoluble salt.
MULTIPLE CHOICE QUESTIONTry yourself: What is the purpose of the filtration process in preparing insoluble salts?
Example: Preparation of pure, dry lead(II) sulfate crystals using a precipitation reaction
Soluble Salts Used
- Soluble Salt 1: lead(II) nitrate
- Soluble Salt 2: potassium sulfate
Method
- Dissolve lead(II) nitrate and potassium sulfate in water. Mix them well in a beaker using a stirring rod.
- Filter the mixture to separate the precipitate.
- Wash the precipitate with distilled water to remove any traces of potassium nitrate solution.
- Allow the washed precipitate to dry in an oven.
Equation of reaction
lead(II) nitrate + potassium sulfate → lead(II) sulfate + potassium nitrate
Pb(NO3)2(aq) + K2SO4(aq) → PbSO4(s) + 2KNO3(aq)
The document Preparation of Insoluble Salts is a part of the Year 10 Course Chemistry for GCSE/IGCSE.
All you need of Year 10 at this link: Year 10
FAQs on Preparation of Insoluble Salts
| 1. What are insoluble salts? |  |
Ans. Insoluble salts are salts that do not dissolve in water or any other solvent to a significant extent.
| 2. How can insoluble salts be prepared? |  |
Ans. Insoluble salts can be prepared by mixing two solutions of soluble salts that react to form an insoluble salt, which can then be precipitated out.
| 3. Can insoluble salts be separated from a solution? |  |
Ans. Yes, insoluble salts can be separated from a solution through a process called precipitation, where the insoluble salt forms a solid that can be filtered out.
| 4. What are some common examples of insoluble salts? |  |
Ans. Some common examples of insoluble salts include silver chloride (AgCl), lead(II) sulfate (PbSO4), and calcium carbonate (CaCO3).
| 5. Why are insoluble salts useful in chemistry? |  |
Ans. Insoluble salts are useful in chemistry for various purposes, such as forming precipitates for qualitative analysis, as catalysts, or as pigments in paints and dyes.
Related Searches
past year papers, video lectures, ppt, Viva Questions, Summary, Preparation of Insoluble Salts, Free, Extra Questions, Important questions, practice quizzes, Exam, pdf , MCQs, study material, Sample Paper, Previous Year Questions with Solutions, mock tests for examination, Preparation of Insoluble Salts, Semester Notes, shortcuts and tricks, Preparation of Insoluble Salts, Objective type Questions;





