IGCSE Class 10 > Class 10 Notes > Physics for GCSE/ > Molecular Matter
Molecular Matter
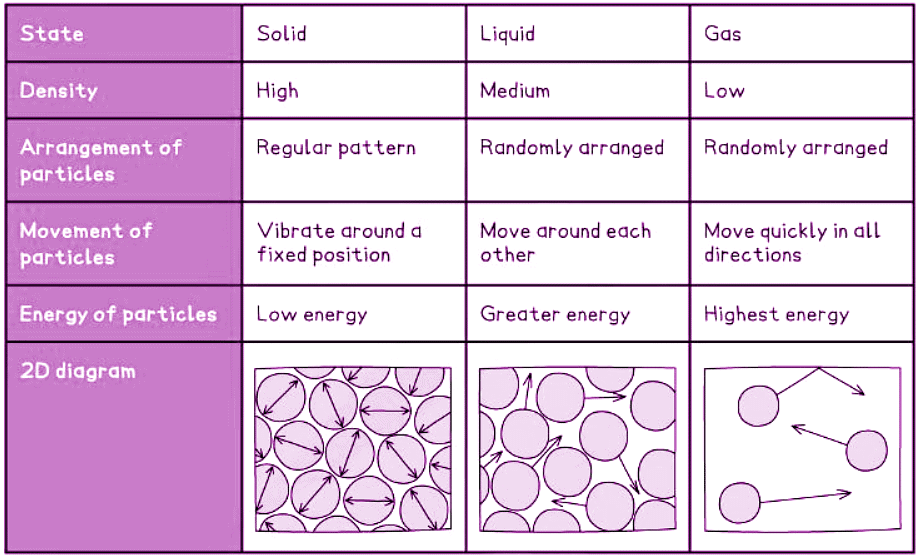
Arrangement & Motion of Particles
In a solid
- Molecules are tightly packed and organized in a consistent structure.
- Molecules oscillate around fixed positions.
In a liquid
- Molecules remain close together without gaps but lack a uniform arrangement.
- Molecules can glide past each other.
In a gas
- Molecules are considerably spaced apart, approximately ten times farther in each direction.
- Molecules exhibit random movement at rapid velocities.
The Forces & Distances between Molecules
- The interactions among particles impact the state of matter. These interactions play a crucial role in determining the arrangement and movement of particles within a substance, influencing its properties such as shape, volume, and flow characteristics.
- The strength of these forces directly influences how particles behave in terms of their relative positions and movements. This, in turn, affects the substance's ability to undergo changes in shape, volume, and flow.
- Changes in shape: The ability of a substance to alter its form without altering its volume.
- Changes in volume: The capacity of a substance to occupy a specific amount of space.
- Flow: The property of a substance to move or deform continuously under an applied force.
- Matter is composed of various types of particles, including atoms, molecules, ions, and electrons. These constituents collectively contribute to the properties and behavior of the substance.
- Atoms: The basic units of matter, consisting of a nucleus surrounded by electrons.
- Molecules: Combinations of atoms held together by chemical bonds.
- Ions: Charged particles formed when atoms gain or lose electrons.
- Electrons: Negatively charged subatomic particles orbiting the atomic nucleus.
Intermolecular Forces and Motion of Particles
Solids
- Molecules in solids are tightly bound by strong intermolecular forces, causing them to vibrate in place.
- The distance between molecules remains constant, giving the solid its rigid structure and fixed volume.
Liquids
- Molecules in liquids possess enough energy to surpass intermolecular forces, yet they remain closely packed.
- Liquid volume mirrors that of solids, but molecules can shift position by sliding past one another.
- This fluidity allows liquids to change shape and flow.
Gases
- Molecules in gases possess higher energy levels and move chaotically at rapid speeds.
- They have overcome the forces keeping them close, resulting in significant spaces between molecules.
- Gases can readily compress and expand, and they flow freely.
MULTIPLE CHOICE QUESTIONTry yourself: What is the characteristic of particles in a solid?
Temperature & Energy of Particles
- The pressure exerted by a gas on its container hinges on the gas's temperature.
- This is because particles acquire kinetic energy with rising temperature.
- As the gas's temperature declines, so does the pressure on the container.
- In 1848, Lord Kelvin, a renowned Mathematician and Physicist, deduced the existence of a temperature at which gas particles cease exerting pressure.
- At this temperature, they cease motion entirely, thus no longer colliding with their container.
- This temperature is termed absolute zero and measures -273 °C.
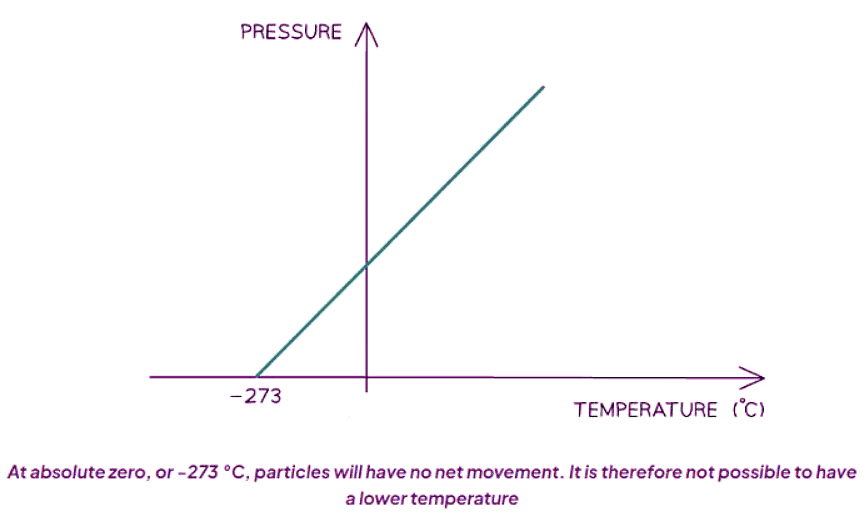
- The unit Kelvin is abbreviated as K.
- It's important to note the absence of the degree symbol, unlike Celsius, which is represented as °C.
The document Molecular Matter is a part of the Class 10 Course Physics for GCSE/IGCSE.
All you need of Class 10 at this link: Class 10
FAQs on Molecular Matter
| 1. How does the arrangement and motion of particles affect the properties of a substance? |  |
Ans. The arrangement and motion of particles determine the physical state and properties of a substance. In solids, particles are tightly packed and vibrate in fixed positions. In liquids, particles are more loosely packed and can move around each other. In gases, particles are far apart and move freely. These differences in arrangement and motion lead to variations in density, shape, and volume of substances.
| 2. What role do forces and distances between molecules play in determining the behavior of matter? |  |
Ans. The forces between molecules, such as hydrogen bonding or van der Waals forces, affect the strength of interactions between particles. The distances between molecules determine how closely packed they are and influence the substance's physical properties. Strong intermolecular forces result in higher boiling and melting points, while weak forces lead to lower points.
| 3. How does temperature impact the energy of particles in a substance? |  |
Ans. Temperature is a measure of the average kinetic energy of particles in a substance. As temperature increases, the particles gain more energy and move faster, leading to an increase in the substance's overall energy. This increased energy can cause changes in the state of matter, such as melting or boiling, as well as alterations in other physical properties.
| 4. Why is understanding the molecular structure of matter important in various scientific fields? |  |
Ans. Understanding the molecular structure of matter is crucial in fields such as chemistry, physics, and biology because it helps explain the behavior and properties of substances. By knowing how molecules are arranged and interact, scientists can predict how materials will behave under different conditions, develop new compounds, and design efficient processes in industries like pharmaceuticals and materials science.
| 5. How can knowledge of particle behavior be applied in everyday life and technology? |  |
Ans. Knowledge of particle behavior is used in various applications, such as designing efficient heating and cooling systems, developing new materials with specific properties, and understanding the effects of pollution on the environment. For example, understanding how particles move and interact in fluids is essential for optimizing the performance of engines and designing effective drug delivery systems.
Related Searches
Sample Paper, pdf , Exam, practice quizzes, ppt, Important questions, Molecular Matter, Semester Notes, Molecular Matter, study material, past year papers, video lectures, Free, Molecular Matter, MCQs, mock tests for examination, Extra Questions, Objective type Questions, Viva Questions, shortcuts and tricks, Summary, Previous Year Questions with Solutions;





