IGCSE Class 10 > Class 10 Notes > Physics for GCSE/ > Evaporation
Evaporation
Cooling by Evaporation
- Evaporation denotes the transition of a liquid into a gas state.
- It takes place:
- Regardless of temperature.
- Solely from the surface of the liquid.
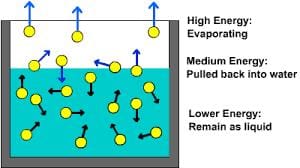
- Molecules within a liquid possess varying energy levels, ranging from high to low.
- The average energy level correlates with the liquid's temperature.
- Evaporation transpires when highly energetic molecules near the liquid's surface possess adequate energy to break free.
- Consequently, the liquid's average energy diminishes.
- Thus, evaporation leads to the cooling of liquids.
Temperature, Surface Area & Air Movements
These factors all affect the rate of evaporation:
- Heightened temperature boosts the kinetic energy of liquid molecules.
- Molecules possessing greater energy are likelier to surpass the intermolecular forces that keep them in the liquid state and break free from the surface.
- Hence, elevated temperature results in a swifter rate of evaporation.
- Molecules can only break free from the intermolecular forces of attraction at the liquid's surface.
- Consequently, a larger surface area facilitates a quicker evaporation rate.
- Air movement carries off recently evaporated water vapor, drying the air and enabling more water molecules to escape.
- Therefore, boosting air movement (referred to as draughts indoors) enhances the rate of evaporation.
Evaporation & Cooling
- Evaporation has a cooling effect, making it useful for lowering temperatures.
- When a substance evaporates, it absorbs heat from its surroundings, including nearby objects.
- This cooling effect can be utilized in refrigeration and air conditioning systems.
- Objects in contact with evaporating liquids experience cooling as the liquid transitions to a gas state.
- Refrigerators and air conditioners exploit this phenomenon to maintain lower temperatures within their compartments.
Boiling vs Evaporation
- Boiling represents the transition of a substance from a liquid to a gas state.
- Boiling occurs specifically at the boiling point of the substance.
- During boiling, the change of state occurs uniformly throughout the liquid, often evidenced by the formation of bubbles, such as those observed in boiling water.

MULTIPLE CHOICE QUESTIONTry yourself: How does evaporation lead to the cooling of liquids?
The document Evaporation is a part of the Class 10 Course Physics for GCSE/IGCSE.
All you need of Class 10 at this link: Class 10
FAQs on Evaporation
| 1. How does cooling by evaporation work? |  |
Ans. Cooling by evaporation works by utilizing the heat energy in a liquid to transform it into a gas, which results in a decrease in temperature due to the loss of heat energy during the phase change.
| 2. What factors affect the rate of evaporation? |  |
Ans. The rate of evaporation is influenced by factors such as the surface area of the liquid exposed to the air, the temperature of the liquid, the humidity of the surrounding air, and the presence of air movement.
| 3. Why does evaporation cause cooling? |  |
Ans. Evaporation causes cooling because as the liquid molecules absorb heat energy from their surroundings to change into a gas, the remaining liquid molecules lose kinetic energy, resulting in a decrease in temperature.
| 4. How is evaporation different from boiling? |  |
Ans. Evaporation is a slow process that occurs at the surface of a liquid, while boiling is a rapid process that happens throughout the liquid when it reaches its boiling point and forms bubbles of vapor.
| 5. Can evaporation be used for cooling purposes in everyday life? |  |
Ans. Yes, evaporation is commonly used for cooling purposes in everyday life, such as through the use of evaporative coolers, sweat evaporating from the skin to cool the body, and in the cooling effect of wet clothes on a hot day.
Related Searches
Important questions, Exam, Free, past year papers, shortcuts and tricks, Summary, MCQs, Sample Paper, video lectures, Viva Questions, Evaporation, mock tests for examination, Extra Questions, Objective type Questions, ppt, practice quizzes, Semester Notes, pdf , Previous Year Questions with Solutions, study material, Evaporation, Evaporation;





