Year 7 Exam > Year 7 Notes > Exothermic and Endothermic Reactions
Exothermic and Endothermic Reactions
Introduction
- When a chemical reaction occurs, there is a transfer of energy to or from the surroundings.
- An exothermic reaction releases energy to the surroundings, often resulting in the feeling of heat.
- An endothermic reaction absorbs energy from the surroundings, leading to a sensation of cold.
Exothermic reactions
- Exothermic reactions involve chemical reactions where new substances are formed by breaking and making chemical bonds between atoms. This process releases energy into the surroundings, often in the form of heat.
- Handwarmers are a common example of exothermic reactions where heat is released into the environment.
- Exothermic reactions can also release energy in the form of light. For instance, glowsticks emit light without a significant increase in temperature.

Example of an Exothermic Reaction
When a chemical reaction releases energy in the form of heat, it is termed as an exothermic reaction. Let's understand this concept with an example:
- When sodium hydroxide solution is added to hydrochloric acid in a beaker, the initial temperature is recorded as room temperature. This mixture results in the formation of sodium chloride and water.
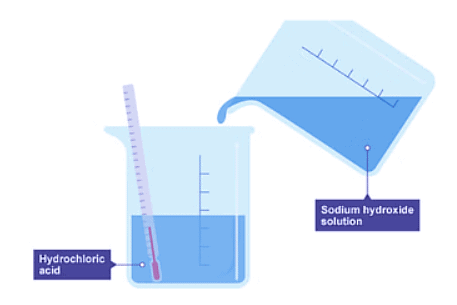
- As the reaction progresses, the temperature in the beaker increases, indicating that heat is being released, making it an exothermic reaction.
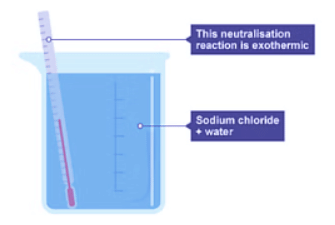
Summary
During an exothermic reaction, such as the neutralization between sodium hydroxide and hydrochloric acid, heat is released to the surroundings. This results in a temperature increase as the reaction progresses, indicating an exothermic process.
Key Points
- An exothermic reaction releases heat to the surroundings.
- The temperature rises during an exothermic reaction, indicating the release of energy.
- Exothermic reactions are common in various chemical processes and are essential in understanding energy changes in reactions.
Importance of Exothermic Reactions
- Understanding exothermic reactions is crucial in various fields such as chemistry, biology, and physics.
- It helps in comprehending energy transfer, heat release, and the behavior of substances during chemical reactions.
MULTIPLE CHOICE QUESTIONTry yourself: Which of the following best describes an exothermic reaction?
Endothermic reactions

- Endothermic reactions involve absorbing energy from the surroundings.
- This absorbed energy, often in the form of heat, causes the surroundings to become cooler.
Photosynthesis
- Photosynthesis is a key endothermic reaction where plants utilize light energy to convert carbon dioxide and water into glucose and oxygen.
- This process results in the production of oxygen as a by-product. Plants, algae, and certain bacteria perform photosynthesis.
Thermal decomposition
- Thermal decomposition reactions are also endothermic as they absorb energy when the compounds are subjected to high temperatures.
- This process involves breaking down a compound into simpler substances or its elemental components.
Example of an endothermic reaction
When a reaction absorbs heat from its surroundings, it's termed an endothermic reaction. Here's a detailed breakdown: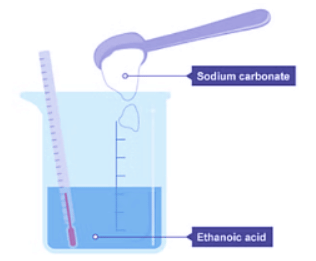
- Sodium carbonate powder is poured into a beaker containing ethanoic acid at room temperature.
- The initial temperature is indicated by a thermometer in the beaker.
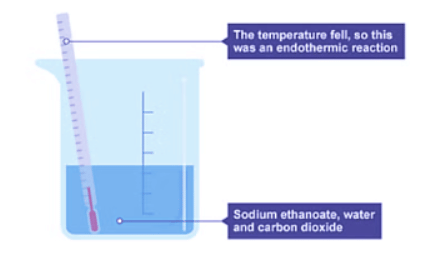
- The mixture in the beaker transforms into sodium ethanoate, water, and carbon dioxide.
- The thermometer registers a drop in temperature, signifying an endothermic reaction.
Related Searches
mock tests for examination, shortcuts and tricks, video lectures, Sample Paper, Exothermic and Endothermic Reactions, MCQs, Summary, Exothermic and Endothermic Reactions, pdf , Extra Questions, Semester Notes, Exam, Free, Objective type Questions, practice quizzes, Viva Questions, Exothermic and Endothermic Reactions, Previous Year Questions with Solutions, ppt, Important questions, past year papers, study material;


