Year 7 Exam > Year 7 Notes > Thermal Decomposition
Thermal Decomposition
Introduction
- Thermal refers to heat.
- Decomposing entails the process of breaking down.
- Thermal decomposition is a chemical reaction that occurs when a compound breaks down upon heating.
What is Thermal Decomposition?
- Definition: Thermal decomposition refers to a chemical process where a compound breaks down upon heating, resulting in the formation of new substances.
- Explanation: When a compound is subjected to heat, its chemical bonds are broken and reformed, leading to the creation of different substances, which can be either compounds or elements.
- Example: For instance, when calcium carbonate (CaCO3) is heated, it decomposes into calcium oxide (CaO) and carbon dioxide (CO2).
Characteristics of Thermal Decomposition
- High Temperatures: Thermal decomposition reactions typically occur at elevated temperatures where the reactants absorb significant amounts of energy.
- Product Formation: The reactants transform into products, which are the substances resulting from the chemical reaction.
- Energy Absorption: The process involves the absorption of energy by the reactants as they break down into new substances.
- Example: When mercuric oxide (HgO) is heated, it decomposes into mercury (Hg) and oxygen gas (O2).
Reactants and Products in Thermal Decomposition
- Reactant Definition: The starting compound in a thermal decomposition reaction, located on the left side of a chemical equation.
- Product Formation: The products are the new substances formed as a result of thermal decomposition, appearing on the right side of a chemical equation.
- Example: When potassium chlorate (KClO3) is heated, it decomposes into potassium chloride (KCl) and oxygen gas (O2).
Chemical Concepts
- Compound: A compound is a pure substance formed by chemically bonding two or more elements in a fixed ratio.
- Reactant: Reactants are chemicals present at the start of a reaction, listed on the left side of a chemical equation before the arrow.
- Element: An element is a pure substance composed of only one type of atom, as indicated on the periodic table.
Thermal Decomposition Reactions
- Thermal decomposition reactions involve absorbing energy from the surroundings.
- These reactions are categorized as endothermic, requiring energy input.
- They find applications in cooking and baking, such as in the preparation of cakes.
MULTIPLE CHOICE QUESTIONTry yourself: What is thermal decomposition?
Equations for Thermal Decomposition
- In a thermal decomposition reaction, there is typically one reactant and two or more products.
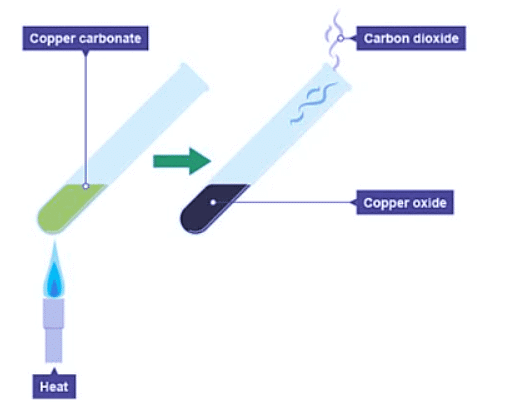
- For instance, heating copper carbonate results in the formation of copper oxide and carbon dioxide.
- Copper carbonate is initially green, while copper oxide is black, illustrating a color change during the reaction. The production of carbon dioxide can be observed using limewater, which turns cloudy.
- Word equation: copper carbonate → copper oxide + carbon dioxide
- Symbol equation: CuCO3(s) → CuO(s) + CO2(g)
Thermal Decomposition without Color Change
- Not all thermal decomposition reactions exhibit a color change. For example, heating magnesium carbonate leads to the production of magnesium oxide and carbon dioxide.
- There is no observable color change as both magnesium carbonate and magnesium oxide are white solids.
- Word equation: magnesium carbonate → magnesium oxide + carbon dioxide
- Symbol equation: MgCO3(s) → MgO(s) + CO2(g)
FAQs on Thermal Decomposition
| 1. What is thermal decomposition? |  |
Ans. Thermal decomposition is a chemical reaction where a compound is broken down into simpler substances by heating it.
| 2. What are some common examples of thermal decomposition? |  |
Ans. Some common examples of thermal decomposition include the breakdown of calcium carbonate into calcium oxide and carbon dioxide, and the decomposition of hydrogen peroxide into water and oxygen.
| 3. How does temperature affect thermal decomposition reactions? |  |
Ans. Increasing the temperature typically increases the rate of thermal decomposition reactions as it provides more energy for the bonds in the compound to break.
| 4. What is the importance of understanding thermal decomposition in industries? |  |
Ans. Understanding thermal decomposition is important in industries as it can help in designing efficient processes for the production of desired products and in the safe handling of chemicals during manufacturing.
| 5. How can thermal decomposition be used in waste management? |  |
Ans. Thermal decomposition can be used in waste management to break down organic waste materials into simpler compounds, reducing the volume of waste and potentially generating useful by-products such as energy.
Related Searches
Thermal Decomposition, mock tests for examination, Important questions, Sample Paper, past year papers, Thermal Decomposition, Previous Year Questions with Solutions, MCQs, Thermal Decomposition, pdf , Free, Exam, video lectures, Objective type Questions, ppt, Viva Questions, Semester Notes, shortcuts and tricks, Summary, practice quizzes, study material, Extra Questions;


