CBSE Class 10 > Class 10 Notes > Science > Mnemonics: Carbon and its Compounds
Mnemonics: Carbon and its Compounds

1. Homologous Series
Mnemonic: Monkeys Eat Peeled Bananas
Interpretations:
- M: Methane (1 Carbon)
- E: Ethane (2 Carbons)
- P: Propane (3 Carbons)
- B: Butane (4 Carbons)
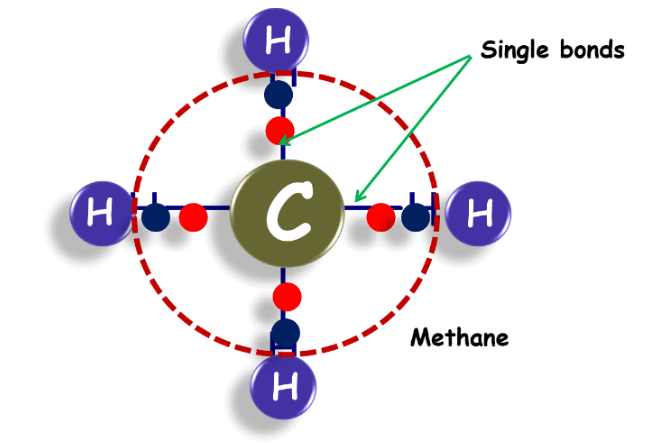 Methane
Methane
2. Properties of Ethanol and Ethanoic Acid
Mnemonic: SMELL
Interpretations:
- S: Solubility (both are soluble in water)
- M: Miscibility (ethanol is miscible with water)
- E: Esterification (ethanol reacts with acids to form esters)
- L: Litmus test (ethanoic acid turns blue litmus red)
- L: Low boiling point (ethanol has a low boiling point)
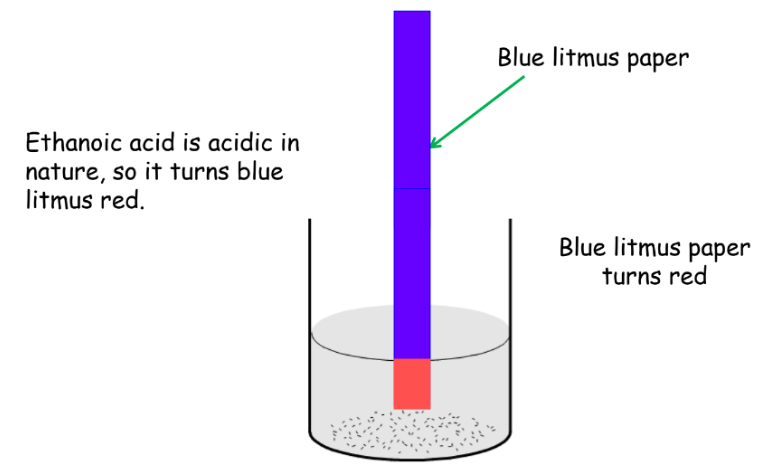 Ethanoic Acid Solution
Ethanoic Acid Solution
3. Versatile Nature of Carbon
Mnemonic: Cats Take Varying Forms
Interpretations:
- C: Catenation (Self-linking ability of carbon atoms)
- T: Tetravalency (Four valence electrons)
- V: Various bonds (single, double, triple)
- F: Forms (linear, branched, cyclic structures)
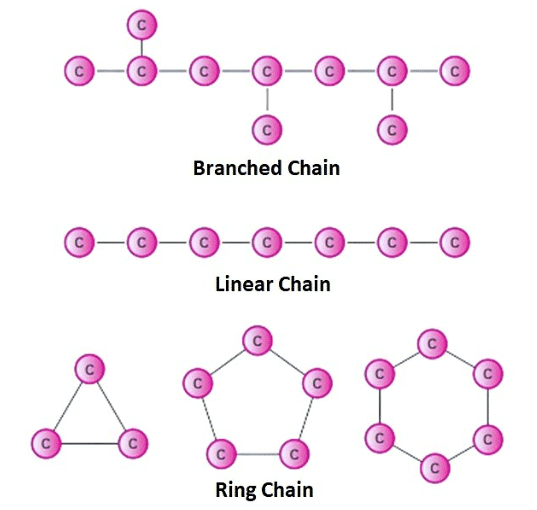 Catenation
Catenation
4. Chemical Properties of Carbon Compounds
Mnemonic: Curious Owls Always Search
Interpretations:
- C: Combustion (Burning of carbon compounds, releasing heat and light)
- O: Oxidation (Gain of oxygen or loss of hydrogen)
- A: Addition Reaction (Hydrogenation of unsaturated hydrocarbons)
- S: Substitution Reaction (Replacement of atoms, e.g., halogenation of alkanes)
The document Mnemonics: Carbon and its Compounds is a part of the Class 10 Course Science Class 10.
All you need of Class 10 at this link: Class 10
FAQs on Mnemonics: Carbon and its Compounds
| 1. What is a homologous series in organic chemistry? |  |
Ans. A homologous series is a group of organic compounds that have the same functional group and similar chemical properties, but differ by a constant unit, usually a -CH2- group. For instance, the alkane series (methane, ethane, propane, etc.) is a homologous series where each successive compound differs from the previous one by a -CH2- group.
| 2. What are the main properties of ethanol? |  |
Ans. Ethanol, or ethyl alcohol, is a colorless, flammable liquid with a characteristic odor. It is soluble in water due to its hydroxyl (-OH) functional group, which allows hydrogen bonding. Ethanol can be used as a solvent, in the manufacture of beverages, and as a fuel. It also has antiseptic properties and is used in medical applications.
| 3. How does ethanoic acid differ from ethanol in terms of properties? |  |
Ans. Ethanoic acid (acetic acid) is a colorless liquid with a pungent smell and is known for its sour taste. Unlike ethanol, it is a weak acid and can donate a proton (H+) in aqueous solution. Ethanoic acid is less volatile than ethanol and has a higher boiling point due to its ability to form hydrogen bonds more effectively, leading to stronger intermolecular forces.
| 4. Why is carbon considered a versatile element? |  |
Ans. Carbon is considered versatile because it can form stable covalent bonds with many elements, including itself, leading to a vast array of organic compounds. It can create single, double, and triple bonds, and can form chains, branched structures, and rings. This ability to bond in various ways allows for the complexity of organic chemistry and the diversity of life on Earth.
| 5. What are some common chemical properties of carbon compounds? |  |
Ans. Common chemical properties of carbon compounds include combustion, where they react with oxygen to produce carbon dioxide and water; addition reactions, particularly in unsaturated compounds; and substitution reactions, often seen in aromatic compounds. Additionally, many carbon compounds can undergo fermentation, esterification, and hydrolysis, showcasing their reactivity and versatility in different chemical environments.
Related Searches
Exam, Sample Paper, Viva Questions, mock tests for examination, MCQs, Semester Notes, study material, shortcuts and tricks, Important questions, pdf , past year papers, Mnemonics: Carbon and its Compounds, Previous Year Questions with Solutions, Summary, ppt, video lectures, Mnemonics: Carbon and its Compounds, Free, Objective type Questions, practice quizzes, Mnemonics: Carbon and its Compounds, Extra Questions;





