The Hydrogen Atom
Hydrogen Atomic Spectrum
Isaac Newton in 1666 realized that white light was composed of the colors of the rainbow. He transmitted sunlight sequentially through a narrow slit and a prism, to project a colored spectrum onto a wall. The same effect is noticed in the sky in the form of a rainbow. Rene Descartes and others had previously suggested that the white light became colored when refracted, and the color depended on the angle of refraction.
- Newton, however, could get back the white light with the help of a second prism, thus strengthening the idea that the white light was composed of separate colors. He further took a monochromatic component from the spectrum generated by one prism and then transmitted that through another prism, and demonstrated that no further colors were generated.
- Newton concluded that white light was made up of all the colors of the rainbow, and different color components was refracted through slightly different angles on transmitting through a prism. This separates the components of white light into the observed spectrum.
- William Wollaston discovered in 1802 that there were many small gaps in the solar spectrum. The rainbow of color had many thin dark lines. Beginning in 1814 Joseph von Fraunhofer investigated the solar spectrum more systematically, and he discovered nearly six hundred dark lines in the spectrum.
- Modern techniques can now detect many thousands of lines in sunlight. These lines are typical spectral absorption lines and are attributed to the absorption of light in the outer layers of the Sun. In general, an absorption spectrum is obtained when light passes through a gas. This spectrum appears as black lines only at specific wavelengths in the background of the continuous spectrum of white light. These missing wavelengths of the incoming radiation are absorbed by the gas.
- Bunsen, Kirchhoff, and others found that when gases are heated to incandescence, they emitted light with a series of sharp wavelengths. When analyzed by a simple prism or sophisticated spectrometer the spectrum appears as colorful lines on a black background.
- These lines are characteristic of the atomic composition of the gas. Each chemical element has its characteristic emission spectrum. The positions of the emission lines of an element are the same as the positions of its absorption lines. This indicates that atoms of a specific element absorb radiation only at specific wavelengths and the radiation emitted by atoms of each element has the same wavelengths as the radiation they absorb.
- Anders Ångström first observed the spectrum of hydrogen atoms in 1853. Ångström measured the four visible spectral lines of hydrogen with wavelengths 656.21, 486.07, 434.01, and 410.12 nm. Based on these numbers Balmer in 1885 proposed a simple formula for predicting the wavelength of any of the lines in atomic hydrogen:
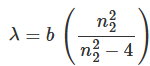
- Here b = 364.56 . The first four wavelengths of equation with n22 = 3 , 4 , 5 , 6 matched well with the experimental obtained spectral lines of hydrogen. The integer n22 extends theoretically to infinity and the series (known as Balmer series) represents a monotonically increasing energy and frequency of the absorption lines.
- Balmer's phenomenological formula was further generalized by Rydberg to determine the wavelengths of any of the lines in the hydrogen emission spectrum. Rydberg's phenomenological equation in terms of the inverse wavelength or the wavenumber
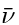 is expressed as:
is expressed as: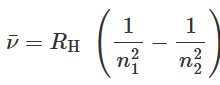
- Here RH = 1.097 37 × 10-7 m-1 (or 2.18 × 10-18 J) is the Rydberg constant and n1 and n2 are integers with n2 > n1.
The various combinations of n1 and n2 can be substituted into equation to calculate the wavelength of any lines in the hydrogen emission spectrum. - For the Balmer series n1 = 2, and n2 can be any whole number between three and infinity. The spectral lines for n1 = 1 are in the UV region, and these lines form the Lyman series. Thus, the spectral lines are grouped into series according to values of n1. Further series are Paschen series with n1 = 3, Brackett series with n1 = 4, Pfund series with n1 = 5, and Humphreys series with n1 = 6.
Quantum physics of hydrogen atom
The Rydberg phenomenological formula for hydrogen provided the exact positions of the spectral lines as they are observed experimentally in a laboratory. However, the explanation of the origin of the spectral lines had to wait for a complete understanding of the atomic structure.
- It was long known that matter is made of atoms, and according to nineteenth-century science, atoms were the smallest indivisible quantities of matter. This scientific view was however, negated by the subsequent discoveries of subatomic particles, such as electrons, protons, and neutrons.
- The electron was discovered by J J Thompson in 1897. Subsequently, in 1904 Thompson proposed the first model of atomic structure, in which an atom was comprised of negatively charged electrons embedded in an unknown positively charged matter like plums in a pudding. In 1909, Rutherford along with Ernest Marsden and Hans Geiger used α-particles in their famous scattering experiment (which is known as the Geiger-Marsden experiment) that disproved Thomson's 'plum-pudding' model.
- In the Geiger-Marsden experiment α-particles were incident on a thin gold foil and were scattered by gold atoms inside the foil. The outgoing α-particles were detected by a 360° scintillation screen surrounding the gold target. A small flash of light was observed on the screen when a scattered particle struck the screen.
- Rutherford and coworkers determined what fraction of the incident α-particles was scattered and what fraction were not deflected at all by counting the scintillations observed at the screen at various angles with respect to the direction of the incident beam. No back-scattered α-particles were expected if the plum-pudding model were correct. However, the results of the Rutherford experiment revealed that while a finite fraction of α-particles emerged from the foil unscattered as if the foil was not in their way a significant fraction of α-particles were back-scattered toward the source.
- Such a result is expected only when most of the mass and the entire positive charge of the gold atom were confined within a tiny space inside the atom. Based on the experimental observations, in 1911 Rutherford proposed a nuclear model of the atom with a positively charged nucleus of negligible size but containing almost the entire mass of the atom. The negatively charged electrons were located within the atom but at a relatively large distance from the nucleus.
- Around 1920 Rutherford coined the name proton for the nucleus of hydrogen. He also postulated the presence of a neutron, the hypothetical electrically neutral particle, to mediate the binding of positive protons in the nucleus of heavier elements. The neutron was experimentally discovered in 1932 by James Chadwick. Rutherford is credited with the discovery of the atomic structure, especially the idea of the atomic nucleus, but his model could not explain the Rydberg formula for the hydrogen emission lines.
- In 1913, Niels Bohr proposed a phenomenological theory of hydrogen atoms to explain the discrete wavelengths of the hydrogen spectrum. Bohr's model is a combination of the classical mechanics of planetary motion with the quantum concept of electromagnetic radiation. We recall here that Newton's universal law of gravitation has a similar formulation to Coulomb's law of electrostatics representing the attraction between two opposite charges in the sense that the gravitational force and the electrostatic force are both decreasing as 1/r2, where r is the separation distance between the bodies or charges.
Classically, if the electron moves around the nucleus in a planetary fashion, then it will then undergo centripetal acceleration and, as per Maxwell's laws of electromagnetism, an accelerating charge particle will radiate energy. This loss of energy will cause it to spiral down into the nucleus. Thus, a planetary configuration of hydrogen atoms as such will not be stable. Bohr intuitively proposed that the puzzle of hydrogen spectra may be solved if the following assumptions are made.
- In a hydrogen atom the electron revolves around a dense nucleus consisting of a proton in circular orbits.
- Electron orbit around the nucleus takes only particular values of radius. In such special orbits, an electron does not radiate energy in the form of an electromagnetic wave as per Maxwell's laws. Such orbits are known as stationary orbits.
- An electron is allowed to make transitions from one orbit with energy En to another orbit with energy Em. The electron moves to a higher-energy orbit when an atom absorbs electromagnetic radiation. On the other hand, when an electron transits to a lower-energy orbit, the atom emits electromagnetic radiation.
- These inter-orbit electron transitions with the simultaneous absorption or emission of electromagnetic radiation take place instantaneously. The allowed electron transitions need to satisfy the Einstein-Planck equation: hv = | En - Em|. Here hv is the energy of either an emitted or absorbed electromagnetic radiation with frequency v and h = 6.626 070 15 × 10-34 m2 kg s-1 is the Planck constant. Any change in the energy of an electron is quantized in the hydrogen atom.
- The angular momentum In of the electron in the nth orbit can take only discrete values ln = nh, where n = 1, 2, 3, .... Here h = h/2π. The angular momentum of electron is quantized. If we denote the radius of the nth orbit and the speed of electron in this orbit by rn and vn, respectively, then the first quantization condition can be expressed as me Un rn = nh. Here me is the mass of an electron.
The last assumption is known as Bohr's quantization rule, and the first three assumptions are called Bohr's postulates.
These three postulates along with the quantization rule of the early quantum theory of the hydrogen atom allow one to derive important properties of the hydrogen atom, such as its energy levels, its ionization energy, and the sizes of electron orbits, along with the Rydberg formula and the value of the Rydberg constant. Bohr's theory can also be applied to hydrogen-like ions with just one electron, such as He+, L++, Be+++, etc.
We shall now use Bohr's postulates to calculate the allowed energies of the hydrogen atom for different allowed orbits of the electron. Let us assume an electron with a negative charge e moves with a constant speed v along a circular orbit of radius r with the center at the hydrogen nucleus containing a proton with positive charge e. The force felt by the electron is the electrostatic Coulomb attraction due to the nucleus, which is expressed as: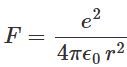
Here ϵ0 is the permittivity of the free space.
The electron feels a centripetal acceleration of magnitude v2/r towards the nucleus. If the mass of the electron is m, then from Newton's law we can express: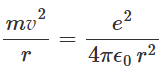 Using equation, the radius of the electron orbit r can be expressed as:
Using equation, the radius of the electron orbit r can be expressed as: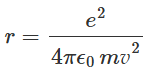
Now using Bohr's quantization rule, the angular momentum of an electron can be written as: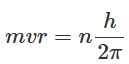
Here n is a positive integer.
Eliminating r from equations and we can get an expression for the velocity v of the electron as: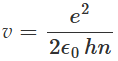
Substituting v from equation back to equation, we can get an expression for r as: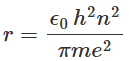
From equation we can see that there is an allowed orbit for each value of n and the radii of the allowed orbits are proportional to n2. The kinetic energy K of the electron in the nth orbit of the hydrogen atom can be expressed using equation as: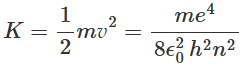 Assuming the potential energy of the electron-nucleus pair of the hydrogen atom to be zero when the electron and nucleus are widely separated, the potential energy V of the hydrogen atom can be expressed as:
Assuming the potential energy of the electron-nucleus pair of the hydrogen atom to be zero when the electron and nucleus are widely separated, the potential energy V of the hydrogen atom can be expressed as:
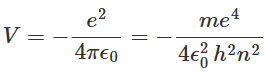 With the assumption that the kinetic energy of the hydrogen nucleus is negligible because of its relatively large mass, the total energy E of the hydrogen atom is expressed as:
With the assumption that the kinetic energy of the hydrogen nucleus is negligible because of its relatively large mass, the total energy E of the hydrogen atom is expressed as: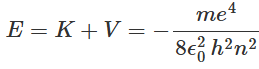 It is possible to obtain various parameters of the hydrogen atom when the electron is in the nth orbit. The hydrogen atom is said to be in the nth energy state in this situation.
It is possible to obtain various parameters of the hydrogen atom when the electron is in the nth orbit. The hydrogen atom is said to be in the nth energy state in this situation.
Radii of hydrogen atom orbits
The radius of the smallest circular orbit allowed to the electron in a hydrogen atom can be expressed as: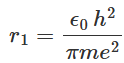 By substituting the values of e, m, h, and ϵ0 in equation (1.12), we obtain the radius r1 = 53 x 10-12 m or 0.053 nm. This length is generally represented by the term do and is called the Bohr radius. This is a suitable unit to measure lengths in atomic physics. In general, the radius of the nth orbit of an electron in a hydrogen atom is represented as:
By substituting the values of e, m, h, and ϵ0 in equation (1.12), we obtain the radius r1 = 53 x 10-12 m or 0.053 nm. This length is generally represented by the term do and is called the Bohr radius. This is a suitable unit to measure lengths in atomic physics. In general, the radius of the nth orbit of an electron in a hydrogen atom is represented as: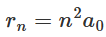
Ground and excited states of the hydrogen atom
The total energy of the hydrogen atom when the electron revolves in the smallest allowed orbit r1 = a0 can be calculated using equation as: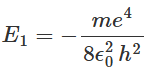 This is the lowest energy state of the hydrogen atom and is also known as the ground state. By putting the values of various constants in equation, one can obtain the value of the ground state energy as E1 = -13.6 eV. One can also see from equation that the energy of the hydrogen atom in the nth energy state is proportional to 1/n2. Thus, we can express En as:
This is the lowest energy state of the hydrogen atom and is also known as the ground state. By putting the values of various constants in equation, one can obtain the value of the ground state energy as E1 = -13.6 eV. One can also see from equation that the energy of the hydrogen atom in the nth energy state is proportional to 1/n2. Thus, we can express En as: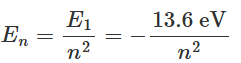
Figure shows schematically the allowed orbits, along with the corresponding energies of the hydrogen atom. The lowest energy state or the ground state corresponds to the lowest circular orbit. We note here that the energy expressed in equation is negative, and hence a larger magnitude means lower energy. The zero energy here corresponds to the energy state when the hydrogen nucleus and the electron are widely separated. The energy of the first excited state of a hydrogen atom is -3.4 eV.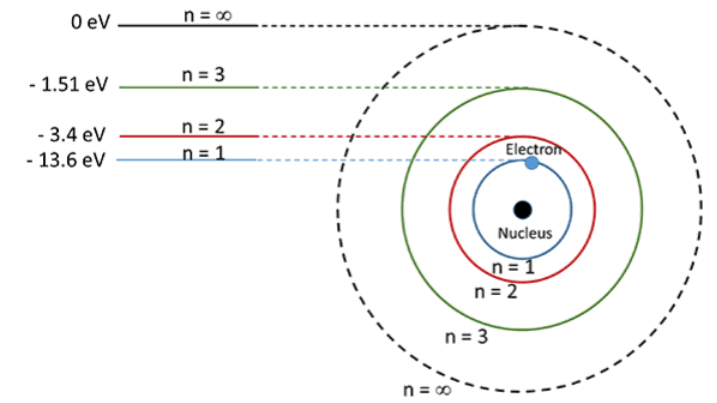
Schematic representation of the allowed orbits along with the corresponding energies of a hydrogen atom in Bohr's atomic model.
Spectra of the hydrogen atom
We shall now try to understand the experimentally obtained hydrogen spectra with the help of Bohr's model of atomic hydrogen. If an electron jumps from the n2th orbit to n1th orbit (where n2 > n1), the energy of the hydrogen atom changes from En2 to En1. The atom will radiate energy in the process. The corresponding inverse wavelength of the emitted radiation can be expressed as: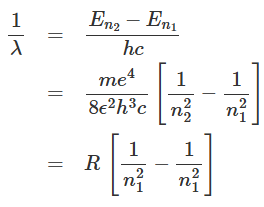 Here
Here 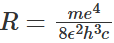 is known as Rydberg constant. With the insertion of the values of different constants, the Rydberg constant R turns out to be 1.0973 x 107 m-1. Equation (1.16) is in excellent agreement with Rydberg's phenomenological formula (1.2) obtained from the experimental results. The energy of the hydrogen atom can be expressed in terms of the Rydberg constant as:
is known as Rydberg constant. With the insertion of the values of different constants, the Rydberg constant R turns out to be 1.0973 x 107 m-1. Equation (1.16) is in excellent agreement with Rydberg's phenomenological formula (1.2) obtained from the experimental results. The energy of the hydrogen atom can be expressed in terms of the Rydberg constant as: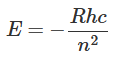 Often the energy of the atom is expressed in rydberg units, where the energy of 1 rydberg means -13.6 eV.
Often the energy of the atom is expressed in rydberg units, where the energy of 1 rydberg means -13.6 eV.
If a hydrogen atom undergoes a transition from the energy state n = 2 to the energy state n = 1, then the wavelength of the resultant emitted radiation is given by: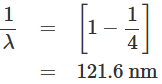 On the other hand, if a hydrogen atom undergoes a transition from the energy state n = ∞ to the energy state n = 1, then the wavelength of the resultant emitted radiation is given by:
On the other hand, if a hydrogen atom undergoes a transition from the energy state n = ∞ to the energy state n = 1, then the wavelength of the resultant emitted radiation is given by: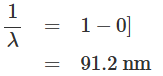
All the transitions ending at the n = 1 state, i.e., ground state, of the hydrogen atom correspond to the wavelengths grouped between 121.6 nm and 91.2 nm. These spectral lines belong to the Lyman series. Similarly, it can be shown that the transitions from the higher-energy states to n = 2 energy state generate radiation with wavelengths lying between 656.3 nm and 365 nm, which belong to the visible region and form the Balmer series. The transitions to the n = 3 energy state from the higher-energy state give rise to the Paschen series, where the wavelengths lie between 1875 nm and 822 nm. Thus, the grouping of wavelengths emitted generated from hydrogen gas can be explained with the help of Bohr's model of the hydrogen atom. The energy level diagram and spectral series of the hydrogen atom are presented in figure.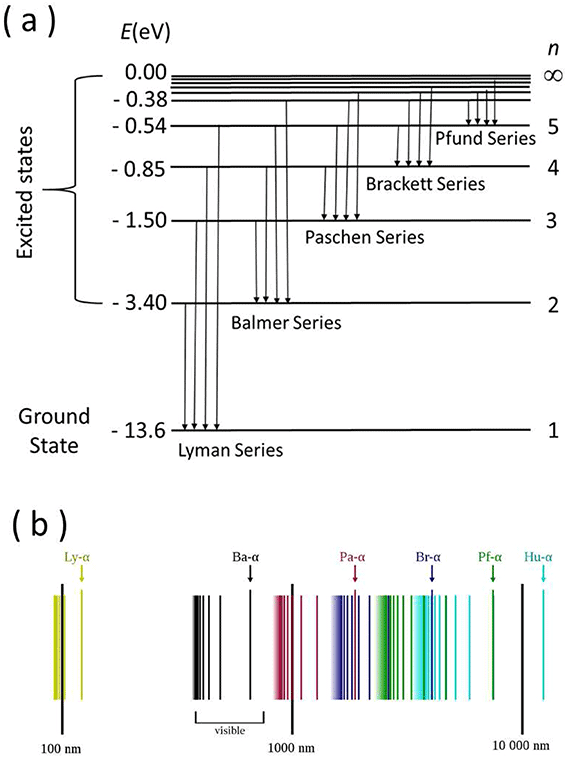
(a) Schematic representation of energy level diagram of hydrogen atom.
(b) Spectral series of hydrogen on a logarithmic scale. This Hydrogen spectrum image has been obtained by the author(s) from the Wikimedia website where it was made available under a CC BY-SA 3.0 licence. It is included within this book on that basis. It is attributed to OrangeDog.
Ionization potential, binding energy, and excitation potential
- If an energy excess of 13.6 eV is applied to a hydrogen atom in its ground state, then the total energy of the atom will be positive. In this situation, the electron is free to move anywhere and not bound to the nucleus anymore. The atom is now ionized with its electron being detached from the nucleus and moving independently with some kinetic energy.
- Ionization energy is the minimum energy needed to ionize an atom, and the ionization energy of a hydrogen atom in its ground state is 13.6 eV. The ionization potential is the potential difference through which an electron needs to be accelerated to acquire ionization energy. Thus, the ionization potential of a hydrogen atom in its ground state is 13.6 V.
- The energy released when the constituents of an atom are brought from infinity to form the atom is called the binding energy of the atom. It may also be defined in another way as the energy required to separate the constituents of an atom over large distances. Assuming that an electron and a proton are initially at rest and then brought together to form an atom, an energy of 13.6 eV will be released. Thus, the binding energy of a hydrogen atom is 13.6 eV, which is the same as its ionization energy.
- Excitation energy is the energy needed to take the atom from its ground state to an excited state. One can estimate from equation (1.17) that energy of 10.2 eV is required to take a hydrogen atom from the ground state to its first excited state. Thus, the excitation energy of a hydrogen atom in its first excited state is 10.2 eV. The excitation potential is the potential through which an electron needs to be accelerated to get the excitation energy. The excitation potential of a hydrogen atom in its first excited state is therefore 10.2 V.
Limitations of Bohr's model
- While Bohr's model of a hydrogen atom was an improvement over Rutherford's nuclear model, it failed to account for finer details of hydrogen spectra, such as multiple closely spaced spectral lines revealed by modern and more sophisticated spectroscopic techniques. At least seven components with slightly different wavelengths have been revealed in what was originally known as the 656.3 nm line. Moreover, Bohr's model was unable to explain the splitting of the spectral lines in the presence of an electric field (i.e., Stark effect) and magnetic field (i.e., Zeeman effect). It also cannot explain the formation of hydrogen molecules out of hydrogen atoms.
- The Bohr model of hydrogen is semiclassical. It combines the classical concept of electron orbits with the new concept of quantization. In Bohr's postulates, it is rather arbitrarily assumed that the orbits are quantized, and in those orbits the electron disobeys the classical law of electromagnetism and does not radiate energy. Accelerating electrons do not spiral into the nucleus by radiating energy as expected within the realm of classical electromagnetism.
Wave-particle duality of the electron and the Heisenberg uncertainty principle
The limitations of Bohr's model subsequently led to the formulation of a more appropriate and general quantum theory of atoms. Two important concepts are basic to the formulation of this new quantum theory:
- Wave-particle duality of matter.
- Heisenberg uncertainty principle
In 1924 Louis de Broglie introduced the concept that matter exhibited dual behavior-both particle and wavelike properties. In the same sense that the quantum of radiation photon has momentum as well as wavelength, electrons, and for that matter any material particle, should also have momentum and wavelength. In addition, de Broglie proposed the following relation between wavelength (λ) and momentum (p) of a material particle.:
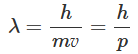
Here h is the Planck constant, m is the mass of the particle, p its velocity, and p its momentum.
In 1927, C J Davisson and L H Germer experimented to demonstrate the wave nature of the electron. This experiment, which is now popularly known as the Davisson-Germer experiment, showed that electron beams can undergo diffraction when passed through atomic crystals. This demonstrated that electrons behaving as waves could exhibit interference and diffraction. It may be noted here that according to de Broglie's theory, every object has a wavelike character associated with it. However, for macroscopic objects, due to the large value of mass m and inherently small value of Planck constant h, their wave properties are negligibly small and hence cannot be detected. On the other hand, for electrons and other subatomic particles with small masses, the associated wavelengths can be determined experimentally with an appreciable degree of accuracy.
In 1927 Wener Heisenberg introduced the uncertainty principle, which is a consequence of the wave-particle duality of matter and radiation. In the case of an electron, the uncertainty principle states that it is impossible to simultaneously determine the exact position and exact momentum (or velocity) of an electron in motion. This can be expressed mathematically as: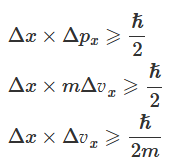
Here Δx denotes uncertainty in position, Δpx denotes uncertainty in momentum, and Δv2 denotes uncertainty in velocity. This indicates that if the position of the electron is known with a high degree of accuracy (i.e., Δx is small), then the velocity of the electron will be uncertain (i.e., Δvx is large), and vice versa.
Heisenberg's uncertainty principle rules out the existence of definite trajectories of electrons. According to Heisenberg's uncertainty principle for an electron (with mass 9.11 x 10-31 kg)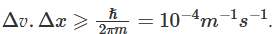 This means that if one can determine the exact location of an electron with an uncertainty of 10-8m, then the uncertainty in the velocity of the electron would be so large that the semiclassical picture of the electron moving in a fixed Bohr orbit will not hold good anymore. A Bohr orbit is a clearly defined path with the position and velocity of an electron being exactly known at the same time. Heisenberg's uncertainty principle thus indicates that the definite statement of position and momentum of electrons needs to be replaced with the concept of probability that the electron has a certain value of position and momentum at an instant of time. A proper model of the Hydrogen atom needs to take into account the wave-particle duality of electrons and be consistent with Heisenberg's uncertainty principle.
This means that if one can determine the exact location of an electron with an uncertainty of 10-8m, then the uncertainty in the velocity of the electron would be so large that the semiclassical picture of the electron moving in a fixed Bohr orbit will not hold good anymore. A Bohr orbit is a clearly defined path with the position and velocity of an electron being exactly known at the same time. Heisenberg's uncertainty principle thus indicates that the definite statement of position and momentum of electrons needs to be replaced with the concept of probability that the electron has a certain value of position and momentum at an instant of time. A proper model of the Hydrogen atom needs to take into account the wave-particle duality of electrons and be consistent with Heisenberg's uncertainty principle.
Schrödinger's wave equation of the hydrogen atom
- De Broglie's equation (equation) associates a wavelength λ with an electron. The particle property of an electron can be understood through this wave property of the electron, which is represented in terms of a wave function Ψ(r, t). This wave function varies continuously in space and at any instant it may be extended over a large part of space (r).
- This does not necessarily indicate that the electron is spread over that large part of space. If one puts an instrument to detect the electron at any point, either a whole electron will be detected or none. The question is then, where will the electron be found? The answer lies within the wave function Ψ(r, t). There is a probability to find the electron wherever |Ψ(r, t)2 ≠ 0.
- It may be noted that the wave function is a mathematical function whose value depends on the coordinates of the electron in the atom, and when an electron is in a particular energy state the wave function corresponding to that energy state contains all information about the electron.
- The wave function of a hydrogen atom or hydrogen-like ions with one electron is called atomic orbital. We shall see shortly that the wave functions are characterized by a set of quantum numbers. It should be made clear here that orbit and orbital are not synonymous. We have seen above that the Bohr semiclassical orbit is a circular path with the nucleus at the center, in which the electron is supposed to move.
- However, the Heisenberg uncertainty principle prohibits a precise description of such a path, and the existence of an orbit cannot be demonstrated experimentally. In contrast, an atomic orbital is a quantum mechanical concept, which corresponds to one-electron wave function ψ in an atom. While ψ as such has no straightforward physical meaning, the quantity |Ψ|2 termed as probability density provides very important information. We shall see below that wave function ψ provides not only information about the position of the electron but also many other properties, including of course the energy states.
- The wave function Ψ(r, t) of the electron and the allowed energies E of a hydrogen atom can be determined by solving the wave equation introduced by Erwin Schrödinger during 1925-26, which is a linear partial differential equation. A detailed formalism and the solution of Schrödinger's equation is provided in appendix A, and here will present only the essential details in the present context of the hydrogen atom. The Schrödinger equation can be written as:
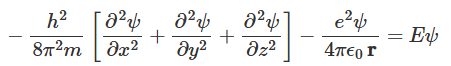
- Here (x, y, z) corresponds to a point at a distance r from the nucleus at the origin. The first term on the left-hand side represents the kinetic energy of the electron, and the second term represents the potential energy of the electron in the field of the nucleus consisting of a single proton. The term E on the right-hand side represents the energy of the atom. The discrete energy levels, spectral lines etc arise from the motion of the electron around the proton. However, the effects of the motion of the proton are ignored because the proton mass mp is much larger than the electron mass me.
- There can be an infinite number of solutions of equation. These wave functions Ψ(r), which satisfy equation, are characterized in terms of three parameters n, l, and ml , which are known as quantum numbers. The parameter n is known as the principal quantum number, l is the orbital angular quantum number, and ml is the magnetic quantum number. It can be shown that associated with each solution Ψnlml is a unique value of energy E of the hydrogen atom. The energy En corresponding to the wave function Ψnlml depends only on the quantum number n and can be expressed as:
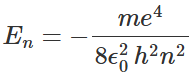
- We can see that these energies are identical to the allowed energies of hydrogen atoms calculated earlier within Bohr's model. This explains the success of Bohr's model in obtaining quantitatively the main spectral lines of hydrogen. For each principal quantum number n there are n values of orbital quantum number l, and for each l there are 2l+1 values of magnetic quantum number ml namely mI =- l,-l+1,-l+2, ... l-1,l.
- Figure presents the energy levels of the hydrogen atom, in which the heights of the horizontal levels represent the energy eigenvalues obtained by solving Schrödinger's wave equation and using equation for energy expression. The lowest possible energy or n = 1 ground state of a hydrogen atom is -Rhc = -13.6 eV. The corresponding radial wave function of the electron in this ground state is:
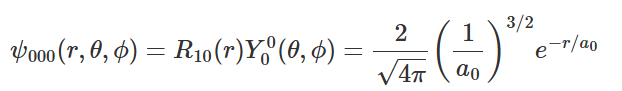
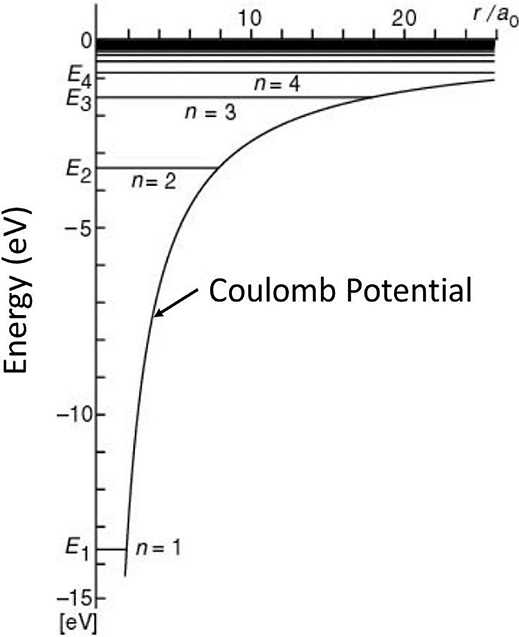 The energy eigenvalues of hydrogen atom obtained as a solution of Schrödinger's wave equation.
The energy eigenvalues of hydrogen atom obtained as a solution of Schrödinger's wave equation.
FAQs on The Hydrogen Atom
| 1. What is the significance of the hydrogen atomic spectrum in quantum physics? |  |
| 2. How do the radii of hydrogen atom orbits relate to the energy levels of the atom? |  |
| 3. What are the limitations of Bohr's model of the hydrogen atom? |  |
| 4. How does the wave-particle duality of the electron relate to the Schrödinger wave equation for the hydrogen atom? |  |
| 5. What is the ionization potential and binding energy of the hydrogen atom, and how are they related? |  |






