ICSE Class 10 > Class 10 Notes > Chemistry > Revision Notes: Periodic Table - Periodic Properties and Variations of Properties
Revision Notes: Periodic Table - Periodic Properties and Variations of Properties
Introduction
- It is a table which classifies all the known elements in accordance with their properties in such a way that elements with similar properties are grouped together in the same vertical column and dissimilar elements are separated.
- The 115 known elements are arranged in the Periodic Table in order of their increasing atomic number.
- The vertical columns are called groups.
- The horizontal rows are called periods.
Approaches to Periodic Classification of Elements
Dobereiner's Triads
In 1817, Dobereiner classified elements with similar chemical properties into groups of three called triads. He noted that the atomic mass of the middle element in a triad is the arithmetic mean of the other two.
This is called Dobereiner's Law of Triads.

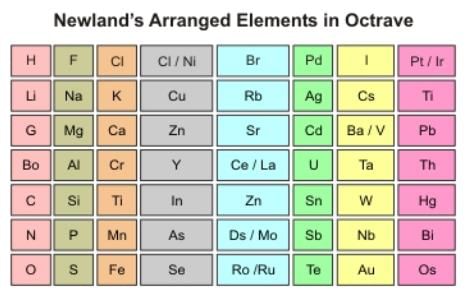
Newlands' Law of Octaves
In 1864, Newlands arranged elements in the increasing order of atomic mass and noticed that the eighth element was similar in physical and chemical properties to the first element, just like the eight nodes in music. This relationship is called the Law of Octaves.
Lother Meyer's Law
In 1869, Lother Meyer classified that atomic volume is the periodic function of atomic mass.
Mendeleev's Periodic Law
In 1869, Mendeleev stated the law of chemical periodicity.
The physical and chemical properties of elements are a periodic function of their atomic masses; i.e. when the elements are arranged in the increasing order of their atomic masses, the elements with similar properties are repeated after certain regular intervals.
This is called Mendeleev's Periodic Law.
Certain anomalies of Mendeleev's table were
(i) Position of hydrogen
(ii) Position of rare earths
(iii) Position of isotopes
(iv) Position of Noble gases
(v) Anomalous pairs of elements
(vi) Cause of periodicity
Modern Periodic Law
The physical and chemical properties of elements are a periodic function of their atomic numbers.Long Form of the Periodic Table
It is based upon the modern periodic law, i.e. elements are arranged according to the increasing order of their atomic number.
Periods
- The horizontal rows of elements in a periodic table are called periods. There are seven periods in the long form of the periodic table.
- The first period consists of 2 elements called the shortest period.
- The second and third periods consist of 8 elements called short periods.
- The fourth and fifth periods contain 18 elements each and are called long periods.
- The sixth period consists of 32 elements and it is the longest period.
- The seventh period is yet an incomplete period.
Groups
- The modern periodic table has eighteen vertical columns known as groups, arranged from left to right in the order: IA, IIA, IIIB, IVB, VB, VIB, VIIB, VIII (three columns), IB, IIB, IIIA, IVA, VA, VIA, VIIA and Zero.
- A group is determined by the number of electrons present in the outermost shell.
- Elements in groups 1, 2 and 13 to 17 are called normal elements.
- Elements in groups 3 to 12 are called transition elements.
- Group 18 at the extreme right contains noble or inert gases.
- Reactive metals are placed in groups 1 and 2.
- Transition elements [metals] are placed in the middle.
- Non-metals are placed in the upper right corner of the periodic table.
Periodicity
The properties which reappear at regular intervals, or in which there is gradual variation at regular intervals, are called periodic properties, and the phenomenon is known as the periodicity of elements.
Shells/Orbits
Electrons in an atom revolve around the nucleus in certain selected but fixed concentric circular paths called shells or orbits. These are associated with a definite amount of energy and are also called energy levels.
Valency
- It denotes the combining capacity of the atom of an element. It is equal to the number of electrons an atom can donate or accept or share.
- On moving from left to right in a period, the number of valence electrons increases from 1 to 8.
- Certain elements lose electrons in steps and hence show variable valency, e.g. Cu, Fe, Ag, Au etc.
- On moving down a group, the valence electrons and valency of all the elements in a group remain the same.
Periodic Properties of Elements
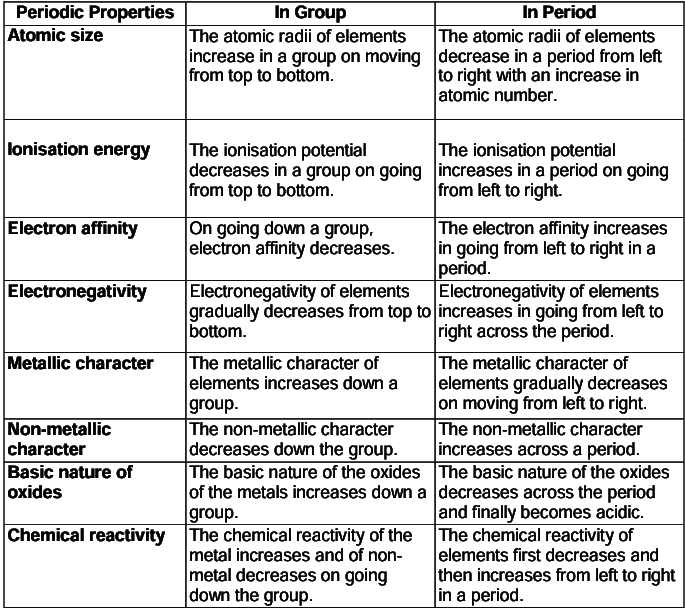
- Atomic size (atomic radii): It is the distance between the centre of the nucleus of an atom and its outermost shell.
- Metallic Character: Those elements which have a tendency to lose their valence electrons and form a positive ion are considered as metals.
Na - e- → Na+ - Non-metallic Character: Those elements which have a tendency to gain electrons in order to attain an octet in their outermost orbit are considered as non-metals.
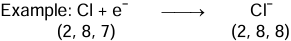
- Chemical Reactivity: In metals, greater the tendency to lose electrons, greater is the reactivity.
In non-metals, greater the tendency to gain electrons, greater is the reactivity. - Ionisation Energy: The energy required to remove an electron from a neutral isolated gaseous atom and convert it into a positively charged gaseous ion is called ionisation energy (IE) or first ionisation energy (IE1).
- Electron Affinity (EA) or Electron Gain Enthalpy: The amount of energy released while converting a neutral gaseous isolated atom into a negatively charged gaseous ion by the addition of electrons is called electron affinity.
- Electronegativity: The tendency of an atom in a molecule to attract the shared pair of electrons towards itself is called its electronegativity.
- Atomic Number (Z): The atomic number of an element is equal to the number of protons in the nucleus.
The atomic number is a unique property of an element, because no two elements have the same atomic number. - Mass Number (A): The mass number of an element is the sum of the number of protons and neutrons in the nucleus of the atom of the element.
Variation of Periodic Properties in the Periodic Table
The document Revision Notes: Periodic Table - Periodic Properties and Variations of Properties is a part of the Class 10 Course Chemistry Class 10 ICSE.
All you need of Class 10 at this link: Class 10
FAQs on Revision Notes: Periodic Table - Periodic Properties and Variations of Properties
| 1. Why does atomic radius decrease across a period in the periodic table? |  |
Ans. Atomic radius decreases across a period because the number of protons increases while electrons are added to the same electron shell. The increased nuclear charge pulls electrons closer, reducing overall atomic size. This trend is a key periodic property students must understand for Class 10 Chemistry exams.
| 2. How do ionization energy and electronegativity change down a group? |  |
Ans. Both ionization energy and electronegativity decrease down a group as atomic radius increases and valence electrons move farther from the nucleus. Weaker nuclear attraction makes it easier to remove electrons and attract them to other atoms. Understanding these periodic variations helps explain element behaviour and reactivity patterns.
| 3. What's the difference between metallic character and non-metallic character across the periodic table? |  |
Ans. Metallic character increases down a group and decreases across a period, while non-metallic character shows the opposite trend. Metals lose electrons easily due to low ionization energy; non-metals gain electrons readily with high electronegativity. This periodic property variation explains why elements on the left are metallic and those on the right are non-metallic.
| 4. Why do noble gases have the highest ionization energy in their periods? |  |
Ans. Noble gases possess completely filled valence shells with eight electrons (except helium with two), making them exceptionally stable and resistant to electron loss. The extremely high ionization energy reflects their reluctance to lose electrons. This periodic trend demonstrates why noble gases are chemically inert and rarely form compounds.
| 5. How does the size of ions relate to atomic number within a group or period? |  |
Ans. Ionic size increases down a group as electron shells are added, despite increasing nuclear charge. Across periods, cations are smaller than their neutral atoms, while anions are larger. These periodic variations in ionic radius are crucial for predicting compound formation and understanding chemical bonding behaviour in Class 10 Chemistry.
Related Searches
Semester Notes, Revision Notes: Periodic Table - Periodic Properties and Variations of Properties, MCQs, Exam, Sample Paper, ppt, shortcuts and tricks, pdf , Extra Questions, video lectures, mock tests for examination, practice quizzes, Viva Questions, Summary, past year papers, Important questions, study material, Objective type Questions, Previous Year Questions with Solutions, Revision Notes: Periodic Table - Periodic Properties and Variations of Properties, Revision Notes: Periodic Table - Periodic Properties and Variations of Properties, Free;






