Revision Notes: Study of Acids Bases and Salts
Acid
An acid is a compound which when dissolved in water yields hydronium ions (H3O+) as the only positively charged ions.
Examples: 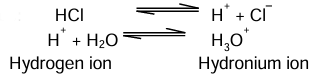
Classification of Acids
- Depending on sources
Organic acid: Acids which are usually obtained from plants are called organic acids. They contain carbon and hydrogen atoms.
Examples: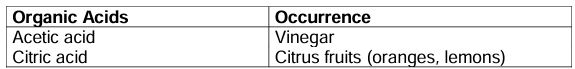 Inorganic (Mineral) acids: Acids which are obtained from minerals are known as inorganic acids.
Inorganic (Mineral) acids: Acids which are obtained from minerals are known as inorganic acids.
Examples: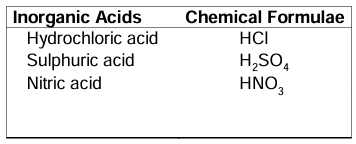
Depending on strength
(a) Strength of an acid: The strength of an acid depends on the concentration of the hydronium ions (H3O+) present in the aqueous solution of an acid.
i. Strong acids: A strong acid vigorously ionises in aqueous solution, thereby producing a high concentration of hydronium ions (H3O+).
Examples: HNO3, HCl, H2SO4
ii. Weak acids: Weak acids ionise only partially in aqueous solution to produce ions and molecules.
Examples: H2CO3, CH3COOH, HCOOHDepending on basicity
Basicity of an acid: The number of hydronium ions (H3O+) which can be produced by the ionisation of one molecule of that acid in aqueous solution.
(i) Monobasic acids: Acids which on ionisation in water produce one hydronium ion (H3O+) per molecule of the acid are known as monobasic acids.
Example: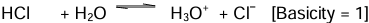
(ii) Dibasic acids: Acids which on ionisation in water produce two hydronium ions (H3O+) per molecule of the acid are known as dibasic acids.
Examples: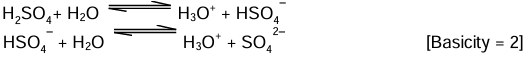
(iii) Tribasic acids: Acids which on ionisation in water produce three hydronium ions (H3O+) per molecule of the acid are known as tribasic acids.
Examples: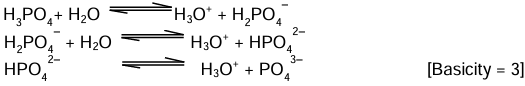
- Depending on concentration: The concentration of an acid means the amount of acid present in a definite amount of its aqueous solution.
i. Concentrated acid: An acid which contains a very small amount of water or no water is called a concentrated acid.
ii. Dilute acid: An acid which contains far more amount of water than its own mass is known as a dilute acid. - Depending on molecular composition
i. Hydracids: Acids which contain hydrogen, a non-metallic element and no oxygen are called hydracids.
Examples: HCl, H2S, HBr, HI
ii. Oxyacids: Acids which contain oxygen, hydrogen and a non-metallic element are called oxyacids.
Examples: H2SO4, HNO3, H2CO3
Preparation of Acids
- By synthesis
H2 + Cl2 → 2HCl - By the action of water on non-metallic or acidic oxides
SO3 + H2O → H2SO4
N2O5 + H2O → 2HNO3 - By oxidation of non-metals
S + 6HNO3 → H2SO4 + 2H2O + 6NO2
P + H3PO4 → H3PO4 + H2O + 5O2 - By displacement
NaCl + H2SO4 → NaHSO4 + HCl
NaNO3 + H2SO4 → NaHSO4 + HNO3
Properties of Acids
Physical properties
i. Sour in taste in aqueous solution.
ii. Turns blue litmus red.
iii. Some acids are solids and some are liquids at room temperature.
iv. All strong mineral acids have corrosive action on the skin and cause painful burns.
v. They are electrolytes, i.e. they conduct electricity in the aqueous state.
Chemical Properties
- Reaction with active metals
Mg + 2HCl → MgCl2 + H2 - Reaction with bases - Neutralisation
NaOH + H2SO4 → NaNO3 + H2O - Reaction with carbonates and bicarbonates
CaCO3 + 2HCl → CaCl2 + H2O + CO2 - Reaction with sulphites and bisulphites
CaSO3 + 2HCl → CaCl2 + H2O + SO2
NaHSO3 + HCl → NaCl + H2O + SO2 - Reaction with sulphides
ZnS + 2HCl → ZnCl2 + H2S - Reaction with chlorides

- Reaction with nitrates
Pb (NO3)2 + 2HCl → PbCl2 + 2HNO3
Uses of Some Acids
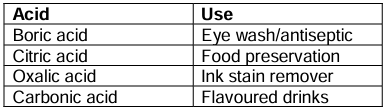
Bases
A base is either a metallic oxide or a metallic hydroxide or ammonium hydroxide which reacts with hydronium ions of an acid to form salt and water only.
Basic Oxide
A basic oxide is a metallic oxide which contains the ion O2- and reacts with an acid to form salt and water.
Alkalis
An alkali is a basic hydroxide which when dissolved in water produces hydroxyl (OH-) ions as the only negatively charged ions.
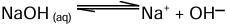
Note: All alkalis are bases, but all bases are not alkalis.
Classification of Bases
- On the basis of strength
i. Strong base: It undergoes almost complete ionisation in aqueous solution to produce a high concentration of OH- ions.
Example: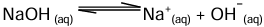
ii. Weak base: It undergoes only partial ionisation in aqueous solution to produce a low concentration of OH- in solution.
Example: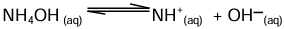
- On the basis of acidity
a. Acidity of a base: The number of hydroxyl ions (OH-) which can be produced per molecule of the base in aqueous solution.
i. Monoacidic base: Bases which dissociate in aqueous solution to produce one hydroxyl ion (OH-) per molecule of the base are called monoacidic bases.
Example: NaOH ⇌ Na+ + OH- [Acidity = 1]
ii. Diacidic base: Bases which dissociate in aqueous solution to produce two hydroxyl ions (OH ) per molecule of the base are called diacidic bases.
Example: Ca(OH)2 ⇌ Ca2+ + 2OH- [Acidity = 2]
iii. Triacidic base: Bases which dissociate in aqueous solution to produce three hydroxyl ions (OH ) per molecule of the base are called triacidic bases.
Example: Al (OH)3 ⇌ Al 3+ + 3OH- [Acidity = 3]
iv. By oxidation of non-metals
S + 6HNO3 H2SO4 + 2H2O + 6NO2
H2SO4 + 2H2O + 6NO2 - On the basis of composition
Concentrated alkali: It is an alkali with a relatively high percentage of alkali in its aqueous solution.
Dilute alkali: It is an alkali with a relatively low percentage of alkali in its aqueous solution.
Preparation of Bases
i. From Metals
2Mg + O2 → 2MgO
ii. By action of water or steam on reactive metals
2Na + 2H2O → 2NaOH + H2
iii. By the action of water on soluble metallic oxides
Na2O + H2O → 2NaOH
iv. By double decomposition
FeCl3 + 3NaOH → Fe (OH)3 + 3NaCl
v. By the action of oxygen on metal sulphides
2ZnS + 3O2 → 2ZnO + 2SO2
vi. By decomposition of salts
CaCO3 → CaO + CO2
Properties of Bases
Physical properties
- They have sharp and bitter taste.
- They change red litmus blue.
- Soapy substances, i.e. they are slippery to touch.
- They are strong electrolytes.
- They show mild corrosive action on the skin.
Chemical properties
- Reaction with carbon dioxide
2NaOH + CO2 → Na2CO3 + H2O - Reaction with acids - Neutralisation
Ca (OH)2 + 2HCl → CaCl2 + 2H2O - Reaction with metallic salts
CuSO4 + 2NH4OH → (NH4)2SO4 + Cu (OH)2
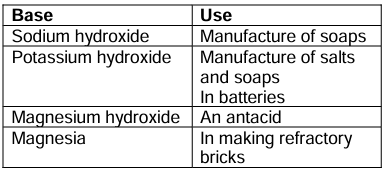
Uses of Some Bases
pH Value
It represents the strength of acids and alkalis expressed in terms of hydrogen ion concentration.
pH of Solution
pH of a solution is the negative logarithm to the base 10 of the hydrogen ion concentration expressed in mole per litre.
pH = -log10 (H+)
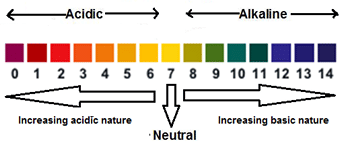
pH Scale
It is a scale showing the relative strength of acids and alkalis.
The normal pH scale ranges from 0 to 14 as shown below.
Indicators
They are complex substances which acquire separate colours in acidic and basic media.
Types of Indicators
a. Acid-base indicators: Common acid-base indicators such as litmus, methyl orange and phenolphthalein can distinguish between acid and basic solutions, but they cannot determine the strength of the solution.
b. Universal indicator: A universal indicator is a mixture of organic dyes which gives a definite colour change over a wide range of pH.
Salts
A salt is a compound formed by the partial or total replacement of the ionisable hydrogen atoms of an acid by a metallic ion or an ammonium ion.
Classification of Salts
- Normal salts: The salts formed by the complete replacement of the replaceable hydrogen ion of an acid molecule by a basic radical.
Example: HCl + NaOH → NaCl + H2O - Acid salts: The salts formed by partial replacement of the replaceable hydrogen ion of an acid molecule by a basic radical.
Example: NaOH + H2SO4 → NaHSO4 + H2O - Basic salts: The salts formed by the partial replacement of the hydroxyl group of a di- or tri-acidic base by an acidic radical.
Example: Mg (OH)2 + HCl → Mg (OH)Cl + H2O - Double salts: The salts formed by the union of two simple salts which dissolve in water and crystallise.
Example: Potash alum: K2SO4. Al2 (SO4)3. 24H2O - Mixed salts: Mixed salts are those salts which contain more than one basic or acidic radical.
Example: Sodium potassium carbonate NaKCO3 - Complex salts: Complex salts are those salts which on dissociation give one simple ion and one complex ion.
Example: Na [Ag (CN)2] ⇌ Na+ + [Ag (CN)2]-
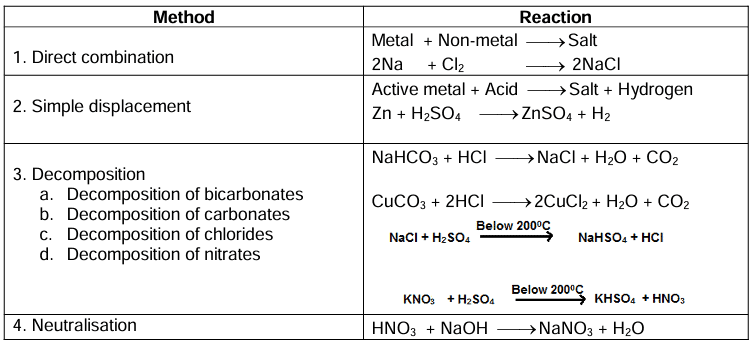
Preparation of Soluble Salts
Preparation of Insoluble Salts
- By direct combination
Reaction: Pb + S → PbS - By combination of an acidic oxide with a basic oxide
Reaction: SO2 + CaO → CaSO3 - Double decomposition
Reactions: BaCl2 + H2SO4 → BaSO4 + 2HCl
Laboratory Preparation of some Normal and Acid Salts
- Iron (III) chloride or anhydrous ferric chloride
It is prepared by passing dry chlorine gas over heated iron.
Fe + Cl2 → FeCl3 - Copper (II) sulphate
It is prepared by the reaction of copper oxide, copper hydroxides or copper carbonates with dilute sulphuric acid.
CuO + H2SO4 → CuSO4 + H2O
Cu (OH)2 + H2SO4 → CuSO4 + 2H2O
CuCO3 + H2SO4 → CuSO4 + H2O
CuSO4 + 5H2O → CuSO4.5H2O - Zinc sulphate and iron (II) sulphate
It is prepared by the reaction of metals with dilute sulphuric acid.
Zn + H2SO4 → ZnSO4 + H2O
ZnSO4 + 7H2O → FeSO4.7H2O - Lead chloride
It is prepared by adding either dilute hydrochloric acid or sodium chloride solution to a solution of lead nitrate.
Pb (NO3)2 + 2HCl → PbCl2 + 2HNO3 - Calcium carbonate
It is prepared by adding sodium carbonate solution to a hot solution of calcium chloride.
CaCl2 + Na2CO3 → CaCO3 + 2NaCl - Sodium bicarbonate
It is prepared by passing excess of carbon dioxide gas through a saturated solution of sodium carbonate.
Na2CO3 + CO2 + H2O → 2 NaHCO3 - Neutralisation
It is the process by which H+ ions of an acid react completely with the [OH-] ions of a base to give salt and water only.
Example: HCl (Acid) + NaOH (Base) → NaCl (Salt) + H2O (water)
Water of Crystallisation
It is the amount of water molecules which enter into loose chemical combination with one molecule of the substance on crystallisation from its aqueous solution.
Hydrated Salt
The salts which contain a definite number of water molecules as water of crystallisation are called hydrated salts.Examples: Na2CO3.10H2O (washing soda), CuSO4.5H2O (blue vitriol)
Anhydrous Salt
A salt which does not contain any water of crystallisation is called an anhydrous salt.Examples: NaCl, NaNO3, Pb(NO3)2
Deliquescence
Water soluble salts which on exposure to the atmosphere absorb moisture from the atmosphere, dissolve in the same and change into a solution. The phenomenon is called deliquescence and the salts deliquescent.
Examples: CaCl2, MgCl2, ZnCl2
Efflorescence
Crystalline hydrated salts which on exposure to the atmosphere lose their water of crystallisation partly or completely and change into a powder. This phenomenon is called efflorescent and the salts efflorescent.
Examples: CuSO4.5H2O, MgSO4.7H2O, Na2CO3.10H2O
FAQs on Revision Notes: Study of Acids Bases and Salts
| 1. What are the main classifications of acids? |  |
| 2. How are acids prepared in a laboratory setting? |  |
| 3. What are the physical and chemical properties of acids? |  |
| 4. What are some common uses of acids in everyday life? |  |
| 5. How are bases classified and what are their characteristics? |  |






