Mnemonics: Chemical Kinetics
Chemical Kinetics deals with the speed of chemical reactions and the factors controlling reaction rates. This chapter is crucial for competitive exams as it involves conceptual clarity, numerical problem-solving, and understanding of rate laws, order, molecularity, and temperature effects. Mastering mnemonics helps in quick recall of complex concepts during time-bound exams.
1. Rate of Chemical Reaction
1.1 Definition and Expression
- Rate of Reaction: Change in concentration of reactant or product per unit time.
- Formula: Rate = -(Δ[Reactant]/Δt) = +(Δ[Product]/Δt)
- Units: mol L-1 s-1 or M s-1
- Mnemonic - "RCP": Rate is Change in concentration Per time
- Sign Convention: Negative for reactants (decreasing), positive for products (increasing)
1.2 Average vs Instantaneous Rate
- Average Rate: Rate over a time interval; becomes less accurate for large intervals
- Instantaneous Rate: Rate at a specific instant; calculated using slope of tangent on concentration-time graph
- Mnemonic - "AI TAN": Average over Interval, Instantaneous uses TANgent
2. Factors Affecting Rate of Reaction
2.1 Four Major Factors
Mnemonic - "CTPC": Concentration, Temperature, Pressure, Catalyst
- Concentration: Higher concentration → more collisions → faster rate
- Temperature: Higher temperature → molecules have more kinetic energy → more effective collisions
- Pressure (for gases): Increased pressure → decreased volume → higher concentration → faster rate
- Catalyst: Provides alternate pathway with lower activation energy → increases rate without being consumed
2.2 Additional Factors
- Surface Area: Larger surface area (smaller particle size) → faster rate for heterogeneous reactions
- Nature of Reactants: Ionic reactions are faster than covalent bond-breaking reactions
- Mnemonic - "SN": Surface area and Nature also matter
3. Elementary and Complex Reactions
3.1 Elementary Reactions
- Definition: Single-step reactions occurring in one stage without intermediates
- Key Feature: Order equals molecularity for elementary reactions
- Example: H2 + I2 → 2HI (one step)
- Mnemonic - "ESOM": Elementary is Single step, Order = Molecularity
3.2 Complex Reactions
- Definition: Multi-step reactions with two or more elementary steps involving intermediates
- Key Feature: Order ≠ molecularity; overall rate determined by slowest step (rate-determining step)
- Example: 2NO + O2 → 2NO2 (occurs in multiple steps)
- Mnemonic - "CMS": Complex has Multiple Steps
4. Order and Molecularity
4.1 Order of Reaction
- Definition: Sum of powers of concentration terms in rate law equation
- Characteristics: Experimentally determined; can be zero, integer, or fractional; may differ from stoichiometric coefficient
- Formula: For aA + bB → products, if Rate = k[A]m[B]n, Order = m + n
- Mnemonic - "O-PEX": Order is sum of Powers, EXperimentally determined
4.2 Molecularity of Reaction
- Definition: Number of reacting species (molecules, atoms, ions) taking part in an elementary step
- Characteristics: Always a whole number; never zero; theoretical concept; applicable only to elementary reactions
- Types: Unimolecular (1), Bimolecular (2), Trimolecular (3)
- Mnemonic - "M-NET": Molecularity is Number of species, Elementary only, Theoretical
- Trap Alert: Molecularity is NEVER fractional or zero, but order can be
4.3 Order vs Molecularity - Comparison Table
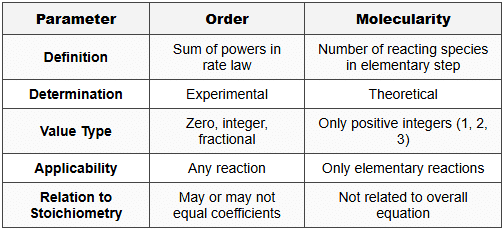
Mnemonic for Table - "OEDIT vs MTOPI": Order is Experimental, Decimal possible, any reaction; Molecularity is Theoretical, Only integers, Positive, elementary only
5. Rate Law and Rate Constant
5.1 Rate Law (Rate Equation)
- Definition: Expression relating rate of reaction to concentration of reactants raised to powers
- General Form: Rate = k[A]m[B]n
- k: Rate constant; m, n: Orders with respect to A and B
- Overall Order: m + n
- Mnemonic - "RLKP": Rate Law has K and Powers
- Trap Alert: Powers in rate law are NOT always equal to stoichiometric coefficients
5.2 Rate Constant (k)
- Definition: Proportionality constant in rate law; equals rate when all concentrations are unity
- Characteristics: Independent of concentration; depends on temperature; specific for each reaction
- Higher k value: Faster reaction
- Mnemonic - "K-TIN": K depends on Temperature, Independent of concentration, specific for each reactioN
5.3 Units of Rate Constant
- Formula: Units of k = (mol L-1)1-n s-1, where n = overall order
- Zero Order: mol L-1 s-1
- First Order: s-1 or time-1
- Second Order: L mol-1 s-1 or M-1 s-1
- Third Order: L2 mol-2 s-1
- Mnemonic - "Zero Most, First Simple, Second Less, Third Least": As order increases, concentration units in denominator increase
6. Zero Order Reactions
6.1 Differential Form
- Rate Law: Rate = k[A]0 = k
- Differential Form: -d[A]/dt = k
- Key Feature: Rate is independent of reactant concentration; remains constant throughout
- Mnemonic - "ZERO = Constant Rate": Zero order means rate doesn't change with concentration
6.2 Integrated Form
- Integrated Rate Law: [A] = [A]0 - kt
- [A]0: Initial concentration; [A]: Concentration at time t
- Graph: [A] vs t is a straight line with slope = -k, intercept = [A]0
- Mnemonic - "ZISL": Zero order, Integrated form, Straight Line ([A] vs t)
6.3 Half-Life (t1/2)
- Formula: t1/2 = [A]0/(2k)
- Key Feature: Half-life is directly proportional to initial concentration
- Characteristics: Half-life decreases as reaction proceeds; depends on [A]0
- Mnemonic - "ZHIP": Zero order Half-life is directly proportional to Initial concentration; not constant
- Trap Alert: Unlike first order, zero order half-life is NOT constant
6.4 Examples of Zero Order
- Photochemical Reactions: H2 + Cl2 → 2HCl (in presence of light)
- Surface Catalyzed Reactions: Decomposition of NH3 on platinum surface
- Enzyme Catalyzed Reactions: At high substrate concentration
7. First Order Reactions
7.1 Differential Form
- Rate Law: Rate = k[A]1 = k[A]
- Differential Form: -d[A]/dt = k[A]
- Key Feature: Rate is directly proportional to reactant concentration
- Mnemonic - "FIRD": First order rate is Directly proportional to concentration
7.2 Integrated Form
- Integrated Rate Law: ln[A] = ln[A]0 - kt or k = (2.303/t) log([A]0/[A])
- Alternative Form: log[A] = log[A]0 - (kt/2.303)
- Graph: ln[A] vs t (or log[A] vs t) is a straight line with slope = -k, intercept = ln[A]0
- Mnemonic - "FILOG": First order Integrated form uses LOGarithm
7.3 Half-Life (t1/2)
- Formula: t1/2 = 0.693/k = ln2/k
- Key Feature: Half-life is independent of initial concentration
- Characteristics: Half-life remains constant throughout the reaction; depends only on k
- Mnemonic - "FHIC": First order Half-life is Independent of initial Concentration
- Value to Remember: ln2 = 0.693 (frequently used in calculations)
7.4 Examples of First Order
- Radioactive Decay: All radioactive disintegrations follow first order
- Decomposition: N2O5 → 2NO2 + ½O2
- Isomerization: Cyclopropane → Propene
- Hydrolysis: Hydrolysis of methyl acetate in acidic medium
8. Zero vs First Order - Comparison Table
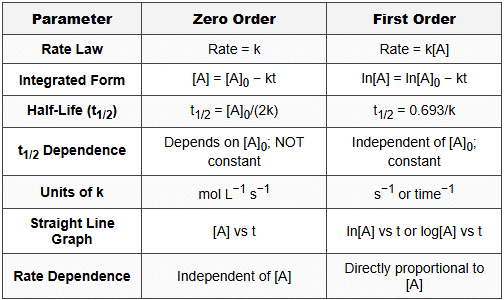
Mnemonic for Comparison - "Z-COIN vs F-LOIN": Zero is Constant rate, depends on Original concentration for half-life, Integrated simple, No log; First uses Log, half-life Only depends on k, Independent of [A]0, proportioNal to [A]
9. Effect of Temperature on Rate - Arrhenius Theory
9.1 Temperature Coefficient
- Rule of Thumb: Rate approximately doubles or triples for every 10°C rise in temperature
- Temperature Coefficient: Ratio of rate constants at two temperatures differing by 10°C
- Formula: Temperature coefficient = k(T+10)/kT ≈ 2 to 3
- Mnemonic - "TEN-DOUBLE": TEN degree rise makes rate roughly DOUBLE
9.2 Arrhenius Equation
- Formula: k = Ae-Ea/RT
- Parameters:
- k: Rate constant
- A: Arrhenius factor or pre-exponential factor or frequency factor (has same units as k)
- Ea: Activation energy (energy barrier for reaction; units: J mol-1 or kJ mol-1)
- R: Universal gas constant = 8.314 J K-1 mol-1
- T: Absolute temperature in Kelvin
- Mnemonic - "KAERT": K equals A times Exponential of -Ea over RT
9.3 Logarithmic Form of Arrhenius Equation
- Natural Log Form: ln k = ln A - (Ea/RT)
- Common Log Form: log k = log A - (Ea/(2.303RT))
- Graph: Plot of ln k vs 1/T is a straight line with slope = -Ea/R and intercept = ln A
- Mnemonic - "LOG-STRAIGHT": LOG form gives STRAIGHT line with 1/T
- Trap Alert: X-axis is 1/T, not T; slope is negative
9.4 Arrhenius Equation for Two Temperatures
- Formula: log(k2/k1) = (Ea/(2.303R)) × [(T2 - T1)/(T1T2)]
- Alternative Form: ln(k2/k1) = (Ea/R) × [(T2 - T1)/(T1T2)]
- Use: Calculate activation energy when rate constants at two temperatures are known
- Mnemonic - "LOG-RATIO-EA": LOG of RATIO of k values gives Ea
10. Activation Energy (Ea)
10.1 Definition and Concept
- Activation Energy: Minimum extra energy required by reactants to form an activated complex (transition state)
- Activated Complex: Unstable intermediate state with highest energy; has partial bonds
- Energy Profile: Ea (forward) = Energy of activated complex - Energy of reactants
- Mnemonic - "EAT-HUMP": Energy needed to cross the HUMP (energy barrier)
10.2 Characteristics of Activation Energy
- Higher Ea: Slower reaction (fewer molecules have sufficient energy)
- Lower Ea: Faster reaction (more molecules can react)
- Independent of: Temperature, concentration, pressure
- Depends on: Nature of reactants and reaction pathway
- Always Positive: Ea cannot be negative or zero for normal reactions
10.3 Calculation of Activation Energy
Method 1 - Using Rate Constants at Two Temperatures:
- Use formula: log(k2/k1) = (Ea/(2.303R)) × [(T2 - T1)/(T1T2)]
- Rearrange: Ea = (2.303R × log(k2/k1) × T1T2)/(T2 - T1)
Method 2 - Using Graphical Method:
- Plot ln k vs 1/T (Arrhenius plot)
- Slope = -Ea/R
- Ea = -Slope × R
Mnemonic - "TWO WAYS EA": Calculate Ea using TWO temperatures or graph
10.4 Effect of Catalyst on Activation Energy
- Catalyst Action: Provides alternate reaction pathway with lower activation energy
- Ea (catalyzed) <>a (uncatalyzed)
- Effect: More molecules have energy ≥ Ea; rate increases
- Important: Catalyst does NOT change ΔH of reaction; only lowers Ea
- Trap Alert: Catalyst lowers Ea for both forward and backward reactions equally
11. Collision Theory of Bimolecular Gaseous Reactions
11.1 Postulates of Collision Theory
- Postulate 1: Reactant molecules must collide for reaction to occur
- Postulate 2: Only a fraction of collisions are effective (lead to products)
- Postulate 3: For effective collision, molecules must have energy ≥ Ea
- Postulate 4: Molecules must have proper orientation during collision
- Mnemonic - "COEP": Collision needed, Orientation proper, Energy sufficient, only few are Productive
11.2 Collision Frequency (Z)
- Definition: Number of collisions per unit volume per unit time
- Proportionality: Z ∝ concentration of reactants
- Temperature Effect: Z increases slightly with temperature (due to increased velocity)
11.3 Fraction of Effective Collisions
- Energy Factor (e-Ea/RT): Fraction of molecules having energy ≥ Ea
- Orientation Factor (P or ρ): Steric factor; fraction of collisions with proper orientation
- Value: P ≤ 1; closer to 1 for simple molecules, much less than 1 for complex molecules
11.4 Rate According to Collision Theory
- Formula: Rate = PZ e-Ea/RT
- P: Steric factor (orientation factor)
- Z: Collision frequency
- e-Ea/RT: Fraction of molecules with sufficient energy
- Connection to Arrhenius: A (Arrhenius factor) = PZ
- Mnemonic - "PEZ": Rate formula has P, Exponential (e-Ea/RT), and Z
11.5 Why All Collisions Don't Lead to Reaction
- Reason 1 - Insufficient Energy: Most molecules don't have energy ≥ Ea
- Reason 2 - Improper Orientation: Molecules collide at wrong angle; reactive sites don't align
- Example: For reaction between NO and O3, only specific orientations lead to product formation
- Mnemonic - "EO-FAIL": Without proper Energy and Orientation, collision FAILS
11.6 Limitations of Collision Theory
- Steric Factor: Cannot predict P theoretically; must be determined experimentally
- Applicability: Works well for simple gas-phase reactions; less accurate for complex reactions
- Ignores: Quantum mechanical effects and molecular structure details
12. Master Mnemonics Summary
12.1 Core Concepts
- "CTPC": Factors affecting rate - Concentration, Temperature, Pressure, Catalyst
- "ESOM": Elementary reactions - Single step, Order = Molecularity
- "O-PEX": Order is sum of Powers, EXperimentally determined
- "M-NET": Molecularity is Number of species, Elementary only, Theoretical
12.2 Zero and First Order
- "ZERO = Constant Rate": Zero order rate independent of concentration
- "ZHIP": Zero order Half-life proportional to Initial concentration
- "FILOG": First order Integrated form uses LOGarithm
- "FHIC": First order Half-life Independent of initial Concentration
- "0.693": Remember this value for first-order half-life calculations (ln2)
12.3 Arrhenius and Activation Energy
- "TEN-DOUBLE": TEN degree rise makes rate roughly DOUBLE
- "KAERT": Arrhenius equation - K equals A times Exponential of -Ea over RT
- "LOG-STRAIGHT": LOG form of Arrhenius gives STRAIGHT line with 1/T
- "EAT-HUMP": Ea is energy to cross the energy HUMP
12.4 Collision Theory
- "COEP": Collision needed, Orientation proper, Energy sufficient, only few are Productive
- "PEZ": Rate formula has P (steric factor), Exponential (e-Ea/RT), and Z (collision frequency)
- "EO-FAIL": Without proper Energy and Orientation, collision FAILS
12.5 Units Quick Recall
- Zero Order k: "Zero has Most units" - mol L-1 s-1
- First Order k: "First is Simple" - s-1
- Second Order k: "Second has Less" - L mol-1 s-1
- Third Order k: "Third has Least concentration" - L2 mol-2 s-1
13. Common Traps and Mistakes
13.1 Order vs Molecularity Confusion
- Trap: Students often confuse order with stoichiometric coefficients
- Reality: Order must be determined experimentally; not related to balanced equation
- Trap: Molecularity can be zero or fractional
- Reality: Molecularity is ALWAYS a positive integer (1, 2, or 3 only)
13.2 Half-Life Dependencies
- Trap: Assuming all reactions have constant half-life
- Reality: Only first-order reactions have constant t1/2 independent of [A]0
- Trap: Zero order half-life is constant
- Reality: Zero order t1/2 depends on [A]0; decreases as reaction proceeds
13.3 Arrhenius Plot Mistakes
- Trap: Plotting ln k vs T
- Reality: Correct plot is ln k vs 1/T (reciprocal of temperature)
- Trap: Slope is +Ea/R
- Reality: Slope is -Ea/R (negative)
13.4 Catalyst Misconceptions
- Trap: Catalyst changes ΔH of reaction
- Reality: Catalyst does NOT change enthalpy change (ΔH); only lowers Ea
- Trap: Catalyst is consumed in reaction
- Reality: Catalyst is NOT consumed; regenerated at end of reaction
13.5 Unit Conversion Errors
- Trap: Using temperature in Celsius in Arrhenius equation
- Reality: Always convert temperature to Kelvin (K = °C + 273)
- Trap: Using Ea in kJ with R = 8.314 J K-1 mol-1
- Reality: Convert Ea to J (multiply by 1000) or use R = 0.008314 kJ K-1 mol-1
Mastering Chemical Kinetics requires understanding the distinction between experimental observations (order, rate law) and theoretical concepts (molecularity, collision theory). Focus on the mathematical relationships between rate, concentration, and temperature. Practice numerical problems involving half-life calculations, activation energy determination, and Arrhenius equation applications. Remember that mnemonics serve as memory anchors, but conceptual clarity is essential for solving complex multi-step problems in competitive exams. Regularly revise the comparison tables and trap alerts to avoid common mistakes during time-pressured exam conditions.
FAQs on Mnemonics: Chemical Kinetics
| 1. What is the importance of chemical kinetics in NEET examination? |  |
| 2. How can I memorize the factors affecting reaction rates for NEET? |  |
| 3. What are the common types of reaction mechanisms discussed in chemical kinetics? |  |
| 4. How do catalysts affect the rate of chemical reactions? |  |
| 5. What role do integrated rate laws play in solving kinetics problems for NEET? |  |






