IGCSE Class 5 > Class 5 Notes > Year 5 Science (Cambridge) > Chapter Notes: States and Properties of Matter
Chapter Notes: States and Properties of Matter
Gases
 Atmospheric Pollution
Atmospheric Pollution
Gases Around Us
- Gases are present in various places, such as in the air we breathe, in fizzy drinks, and inside bicycle and car tyres.
- Most gases are invisible, which makes it difficult for us to detect them using our senses.
- Additionally, many gases have no smell or taste, making them even less noticeable in our daily lives.
The Particle Model of Matter
The particle model helps us understand how solids, liquids, and gases are built and how they act based on how their particles are arranged and how they move.
Solids
- In solids, particles are very close together, with only tiny gaps between them, arranged in an orderly way.
- Particles in solids vibrate in place because of strong bonds, which stops them from moving around freely.
- This is why solids, like bricks, have a fixed shape and cannot be compressed or squashed.
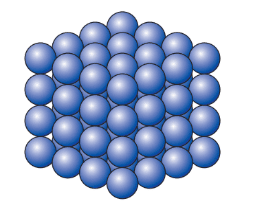 The particles in a solid
The particles in a solid
Liquids
- In liquids, particles are close but a bit more spread out than in solids, with small spaces between them.
- These particles are not in a fixed pattern and can move and vibrate faster, sliding past each other because the bonds are weaker.
- This allows liquids, like water, to flow and take the shape of their container.
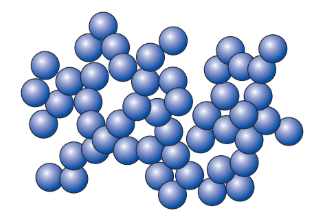 The particles in a liquid
The particles in a liquid
Gases and the particle model
- In gases, particles are far apart, with large spaces between them.
- These particles move freely in all directions at high speeds because of weak bonds.
- This is why gases, like air, have no fixed shape and can be compressed.
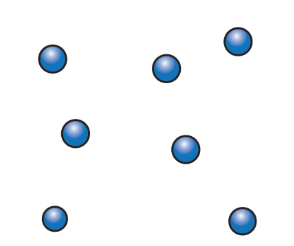 The particles in a gas
The particles in a gas
Air
- Air is composed of various gases, with nitrogen accounting for about four-fifths of its volume.
- Oxygen is crucial for human respiration; we inhale it, while we exhale carbon dioxide, a byproduct of metabolism.
- Air also contains water vapour and carbon dioxide.
- The atmosphere refers to the layer of air surrounding the Earth, consisting of these gases.
- Although gases like hydrogen and helium do exist, they are not commonly found in the Earth's atmosphere.
Air Pollution
- Air pollution is when harmful substances are found in the air we breathe.
- Air pollution happens when harmful things like smoke, dust, and gases from factories, power stations, cars, and planes make the air dirty and unsafe for living beings.
- Indoor air pollution can come from cigarette smoke, cooking fumes, and chemicals in spray cans and paints.
- Air pollution can lead to health issues like coughing, dizziness, headaches, and serious lung diseases, especially from cigarette smoke.
Properties of Water
Water Changes State
- Water can be found in three forms: solid (like ice), liquid (like water), and gas (like water vapour).
- Water changes its form when it is heated or cooled:
- When ice is heated, it melts into liquid water.
- When liquid water is heated, it boils and turns into steam (which is heated water vapour).
- When solids like ice are heated, their particles gain energy, move faster, and spread apart, causing the solid to change into a liquid (solid heat → liquid).
- When liquids like water are heated, their particles gain more energy, move quickly, and some escape as gas, forming steam above the liquid (liquid heat → gas).
Melting and Boiling Points
- The melting point is the temperature at which a solid turns into a liquid; for ice, this is 0°C.
- The boiling point is the temperature at which a liquid turns into a gas; for pure water, this is 100°C.
- Melting and boiling points are fixed characteristics of a substance and do not change unless the substance itself is changed.
- Temperature, which measures the heat in a substance, is recorded in degrees Celsius (°C) using a thermometer.
- Most water contains dissolved substances, which can cause it to boil at a temperature slightly different from 100°C.
- Water's boiling point decreases at higher altitudes above sea level.
Water expands
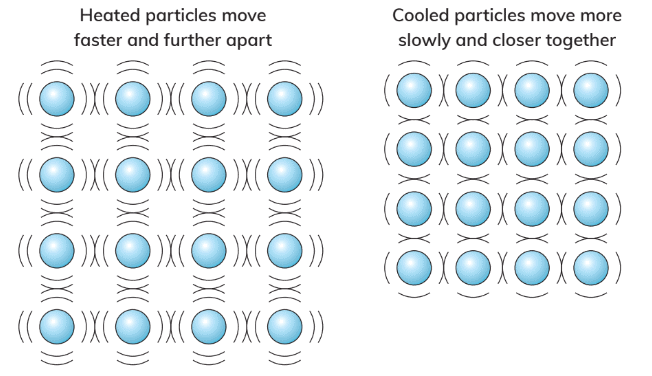
- When a substance is heated, its particles gain energy, move faster, and spread out, leading to expansion and an increase in volume.
- Conversely, when a substance is cooled, its particles lose energy, move slower, and come closer together, resulting in contraction and a decrease in volume.
- Water is unusual because it expands when it freezes into ice, rather than contracting.
- This unique property of water allows ice to float on liquid water.
Evaporation and Condensation
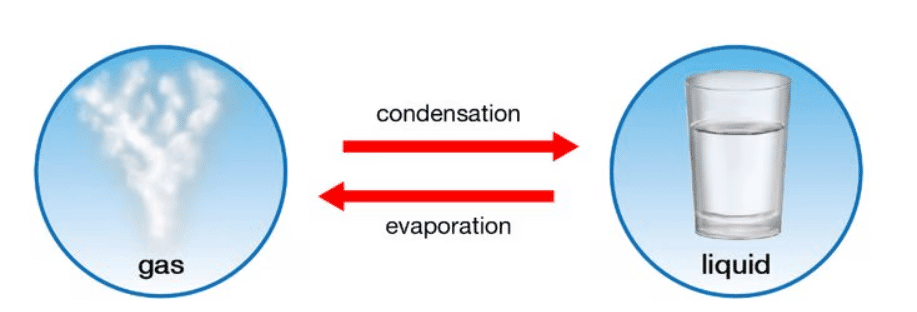
Evaporation
- Evaporation is the process where a liquid, such as water, transforms into a gas (water vapor) when it is heated sufficiently.
- This transformation occurs when the particles at the surface of the liquid gain enough heat energy to move more vigorously and escape into the air.
- Evaporation is a natural occurrence, evident in everyday situations like the drying of puddles or clothes left outside to dry.
Condensation
- Condensation is the process in which a gas, such as water vapor, cools down and changes into a liquid, forming droplets.
- This phenomenon happens when gas particles lose energy, slow down, and come closer together, resulting in the transition from gas to liquid state.
- A common example of condensation is observed when water droplets form on a cold surface, like a mirror, when warm, moist breath hits it.
- Condensation is the reverse of evaporation; it involves a gas becoming a liquid, while evaporation involves a liquid becoming a gas.
The Water Cycle
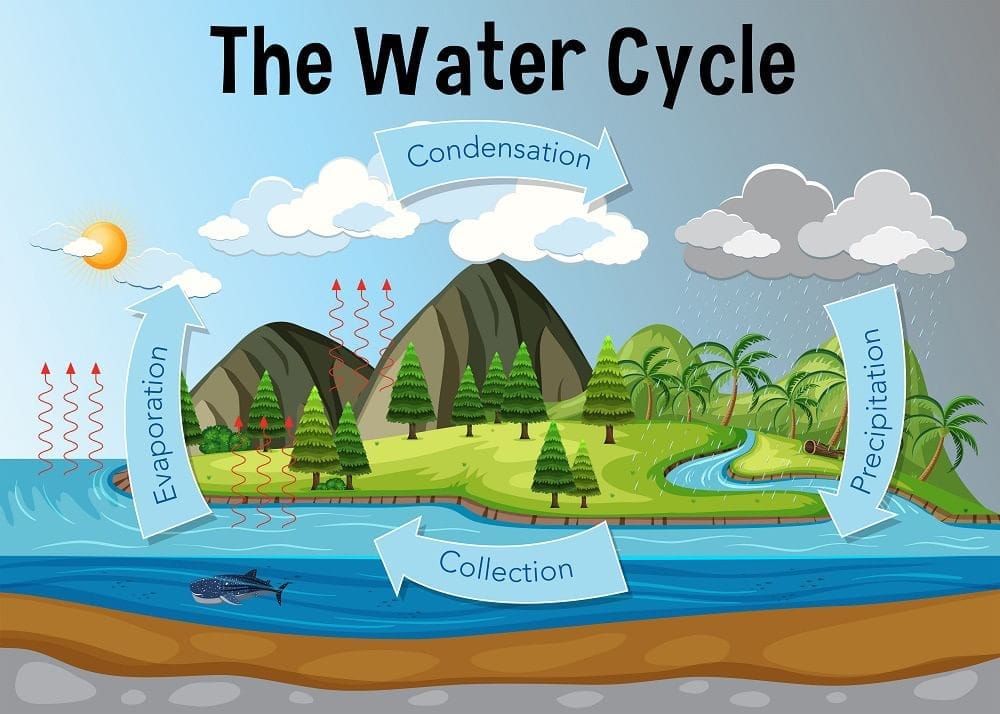
- The water cycle refers to the continuous movement of water among the land, sea, and air, primarily driven by the processes of evaporation and condensation.
- Key processes involved in the water cycle are:
- Evaporation: This is the process where water from oceans, lakes, and other surfaces transforms into water vapor and enters the atmosphere.
- Condensation: In this stage, water vapor cools down and changes back into liquid form, leading to the formation of clouds in the atmosphere.
- Precipitation: When the clouds become heavy with water, they release it in the form of rain, snow, or hail, which falls back to the Earth's surface.
- Freezing and Melting: Water can change between its solid (ice) and liquid (water) states depending on the temperature conditions.
- Precipitation, which includes rain, snow, and hail, plays a crucial role in returning water to the Earth's surface, thus continuing the water cycle.
Solutions
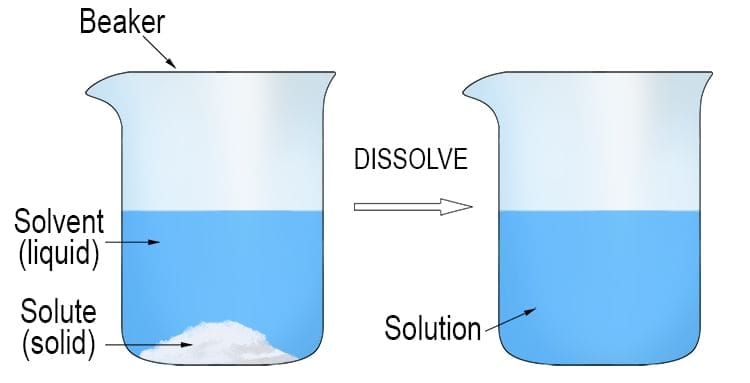
Dissolving and Solutions
- Dissolving occurs when a solid, like sugar, mixes with a liquid such as tea.
- The solid appears to disappear as its particles spread into the spaces between the liquid's particles.
- Solids that can dissolve in a liquid are called soluble solids, and solubility is a property of the solid.
- Solids that cannot dissolve in a liquid are insoluble solids.
- A solution is a type of mixture formed when a solid (solute) dissolves in a liquid (solvent), resulting in a uniform appearance where the solute is evenly distributed and not visible.
- In a solution:
- The solute is the solid that dissolves (e.g., sugar in tea, salt in seawater).
- The solvent is the liquid that dissolves the solid (e.g., tea, water).
- Dissolving is a property of both the solute and the solvent, as the liquid must be capable of dissolving the solid.
- Water is often called a universal solvent because it can dissolve a wide range of substances, although some materials, such as oils, do not dissolve in it.
- An example of a solution is seawater, where salt (solute) dissolves in water (solvent).
- When a solute dissolves, its particles spread evenly among the solvent particles, creating a uniform mixture that looks the same throughout.
Water Pollution
- Water pollution occurs when harmful substances contaminate water bodies, making the water unsafe for humans, animals, and plants.
- Common causes of water pollution include industrial discharge, agricultural runoff, sewage disposal, and plastic waste.
- Polluted water can harm aquatic life, disrupt ecosystems, and pose health risks to humans who consume or come into contact with the contaminated water.
Some solutions can be harmful
- Soluble substances can cause water pollution without being visible, making it difficult to detect contamination.
- Examples of soluble substances that lead to water pollution include:
- Chemical fertilizers used by farmers can wash into rivers and contaminate water sources.
- Pesticides sprayed on crops can dissolve in rainwater and pollute rivers and seas.
- Dissolved chemicals, such as acids from factories, can taint rivers and lakes.
- Insoluble substances contributing to water pollution include plastics and oils.
- Contaminated water poses serious environmental and health hazards, harming plants, animals, and humans.
Separating a Solution
- A solution consists of a solute and a solvent, and the particles are not chemically bonded, allowing for their separation.
- Evaporation is a common technique to separate a dissolved solute from a solution, such as extracting salt from seawater.
- Dissolving is a reversible process, meaning the solute can be recovered from the solution through methods like evaporation.
- For example, when a salt solution is evaporated, the water (solvent) vaporizes, leaving behind the solid salt (solute).
The document Chapter Notes: States and Properties of Matter is a part of the Class 5 Course Year 5 Science IGCSE (Cambridge).
All you need of Class 5 at this link: Class 5
FAQs on Chapter Notes: States and Properties of Matter
| 1. What are the main properties of gases? |  |
Ans. Gases have several key properties, including low density, high compressibility, and the ability to fill the shape and volume of their containers. Unlike solids and liquids, gas particles are far apart and move freely, which allows them to expand and occupy any available space.
| 2. How does evaporation occur? |  |
Ans. Evaporation is the process by which liquid molecules gain enough energy to escape into the air as gas. This occurs at the surface of the liquid and can happen at any temperature, although it occurs more rapidly at higher temperatures due to increased molecular energy.
| 3. What factors influence the rate of condensation? |  |
Ans. The rate of condensation is influenced by temperature, humidity, and air pressure. When warm, moist air comes into contact with a cooler surface, the temperature drops, causing water vapor to lose energy and condense into liquid water. Higher humidity levels can also increase condensation rates.
| 4. What is a solution, and how is it different from a mixture? |  |
Ans. A solution is a homogeneous mixture where one substance (the solute) is dissolved in another (the solvent). Unlike a regular mixture, where the components can often be separated and retain their individual properties, in a solution, the solute particles are evenly distributed at a molecular level and cannot be easily separated.
| 5. How does temperature affect the solubility of a substance in a solution? |  |
Ans. Temperature generally affects the solubility of solids and gases differently. For most solids, solubility increases with temperature; as the temperature rises, more solute can dissolve in the solvent. Conversely, for gases, solubility typically decreases with increasing temperature, as warmer temperatures provide gas molecules with more energy to escape the liquid.
Related Searches
shortcuts and tricks, past year papers, Exam, Previous Year Questions with Solutions, Free, Viva Questions, Sample Paper, Chapter Notes: States and Properties of Matter, Semester Notes, Important questions, practice quizzes, Summary, Extra Questions, ppt, Chapter Notes: States and Properties of Matter, study material, video lectures, Objective type Questions, pdf , MCQs, Chapter Notes: States and Properties of Matter, mock tests for examination;





