Chapter Notes: Changes Around Us - Physical and Chemical
Introduction
We observe many changes happening around us every day. Ice melts into water, flowers bloom from buds, fruits change colour and smell, and cold water becomes warm over time. These changes affect how things look, smell, feel or taste. By using our five senses - sight, smell, touch, hearing and taste - we can notice and understand these changes better.

In this chapter we will learn to distinguish between two broad kinds of changes: physical changes and chemical changes. We will study everyday examples, how to test for some chemical changes and why some changes are reversible while others are not.
Physical Change Vs Chemical Change
Physical Change
Definition: A physical change is a change in which the appearance, shape, size or state of a substance changes but its chemical composition remains the same. No new substance is formed in a physical change.
 Physical Changes
Physical ChangesKey features of physical changes
- Only physical properties such as shape, size or state change.
- The chemical composition of the substance remains unchanged.
- Physical changes are often reversible, though not always.
Examples of physical changes
- Paper folding: Folding paper into shapes changes its form; unfolding it returns the original sheet.

- Balloon inflation: Inflating a balloon stretches it; letting the air out returns it to the original shape. If a balloon is pricked and bursts, the rubber is still the same material but the original shape cannot be restored.
- Crushing chalk: Crushing chalk into powder changes its form, but the chalk remains chemically the same; the original piece cannot always be reformed.

- Change of states of water: Ice → water → steam are physical changes because the substance remains water.
Chemical Change
Definition: A chemical change (or chemical reaction) is a change in which one or more new substances with different properties are formed. Chemical changes are usually not easily reversible by simple physical methods.
 Chemical Changes
Chemical ChangesExamples and explanations
1. Blowing air into lime water
- When carbon dioxide (CO₂) from our breath is bubbled through lime water (a solution of calcium hydroxide), a chemical reaction takes place.
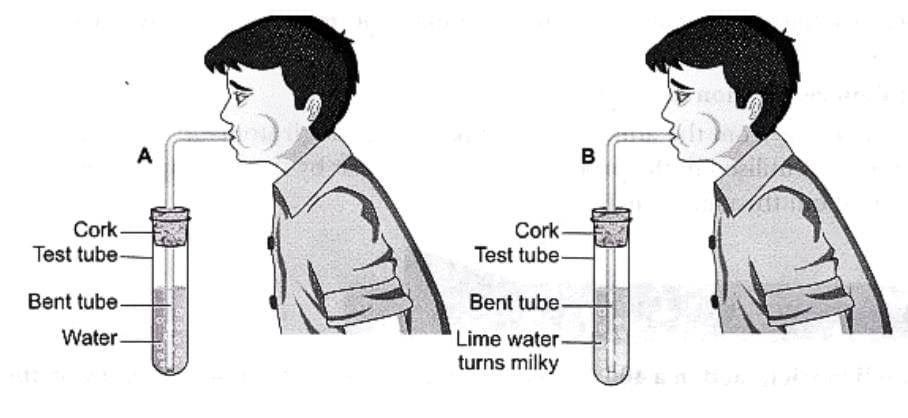
- The carbon dioxide reacts with calcium hydroxide to form calcium carbonate, a white insoluble solid that makes the solution look milky or cloudy.
- The white solid (calcium carbonate) eventually settles at the bottom as a precipitate.
- Water is also produced during this reaction.
Chemical equation (word form):
Calcium hydroxide (lime water) + Carbon dioxide → Calcium carbonate (insoluble) + Water
This experiment shows that a new substance is formed, so the process is a chemical change.
2. Vinegar and baking soda
- When vinegar (contains acetic acid) is mixed with baking soda (sodium bicarbonate), they react vigorously to produce carbon dioxide gas, seen as bubbling or fizzing.

- The carbon dioxide produced will turn lime water milky if bubbled through it, showing it is the same gas produced during respiration and many combustion processes.
Reaction (word form):
Vinegar + Baking soda → Carbon dioxide + Other substances
- This confirms the production of carbon dioxide gas by the chemical reaction.
Some Other Processes Involving Chemical Changes
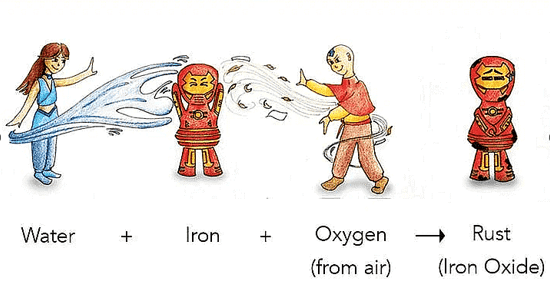
Rusting
- Rusting is a chemical change in which iron reacts with oxygen and moisture in the air to form iron oxide (rust), a new substance that is brown and flaky.
- Rust weakens iron objects such as nails, tools and bridges because the new substance has different properties from iron.
 Rusting of Iron
Rusting of IronCombustion
Combustion is a chemical reaction in which a substance reacts with oxygen to produce heat and often light. Materials that burn are called combustible substances (examples: wood, paper, cotton, kerosene).
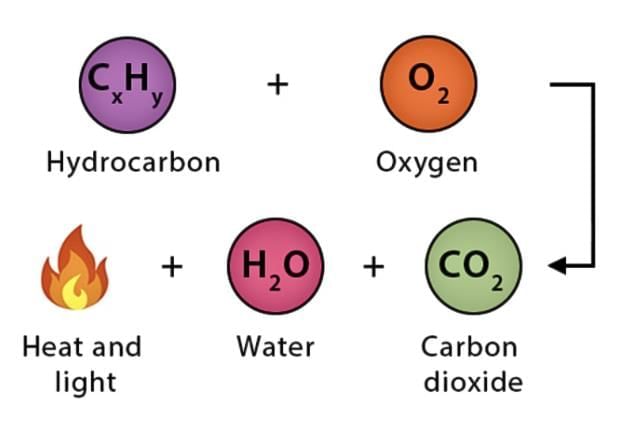
- For example, burning a magnesium ribbon in air gives a white powder called magnesium oxide, showing that new substances are produced during combustion.
- Combustion reactions release energy as heat and sometimes light.
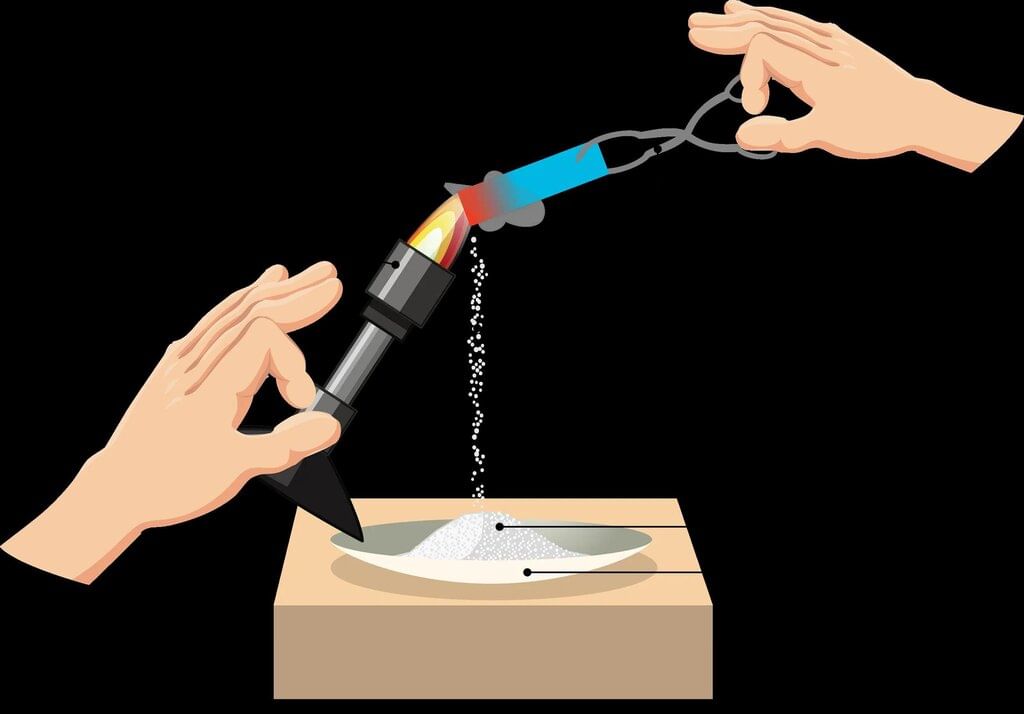
- The chemical reaction for burning magnesium is:
Magnesium + Oxygen → Magnesium oxide + Heat + Light
(Ribbon) (Air) (White powder)
Oxygen's role in combustion
- Oxygen from air is essential for most combustion reactions.
- If a burning candle is covered so it cannot get air, the flame goes out because oxygen is used up and cannot be replenished.
- During combustion, carbon in the fuel reacts with oxygen to form carbon dioxide, which can be detected by passing the gas through lime water (which turns milky).
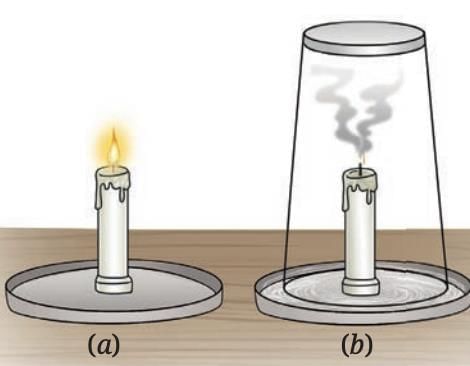
What else is needed to start combustion?
- Besides fuel and oxygen, heat is required to start combustion. The minimum temperature required for a substance to start burning is called its ignition temperature.

- For example, a lighted match supplies the heat needed to bring paper to its ignition temperature and it burns.
- Concentrated sunlight (using a magnifying glass) can also raise the temperature of paper to its ignition temperature and set it on fire.

Fascinating fact
Have you seen tiny glowing insects in gardens at night?

They are called fireflies. Their light is produced by a chemical reaction inside their bodies and is an example of bioluminescence. This light is produced with very little heat.
Science and society - safety
What to do if someone's clothes catch fire?
Wrap the person in a blanket or cloth to cut off the air supply and stop the flames. This is safer than trying to blow out the fire.
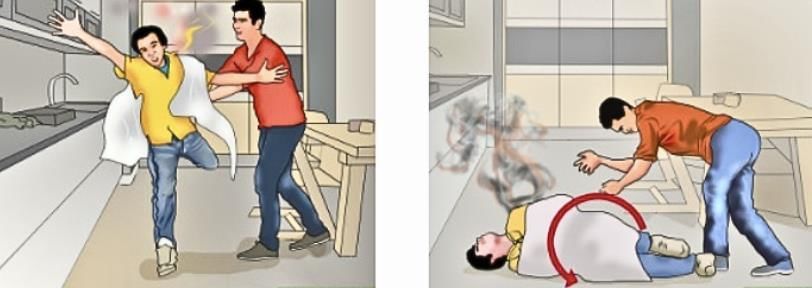
Important: Do not use synthetic blankets or clothes because they may melt and stick to the skin, causing more severe injuries.
Can Physical and Chemical Changes Occur in the Same Process?
1. Burning a candle: physical changes
- Melting of wax: Heat from the flame melts the solid wax to liquid. This is a physical change because the substance remains wax.
- Evaporation: Liquid wax near the wick vaporises and moves up the wick as vapour - a physical change.
- Solidification: Wax that drips and cools solidifies back into a solid - another physical change.
2. Burning a candle: chemical change
- Burning of wax vapour: The vapour burns in oxygen to produce carbon dioxide, water, heat and light. New substances are formed, so this is a chemical change.
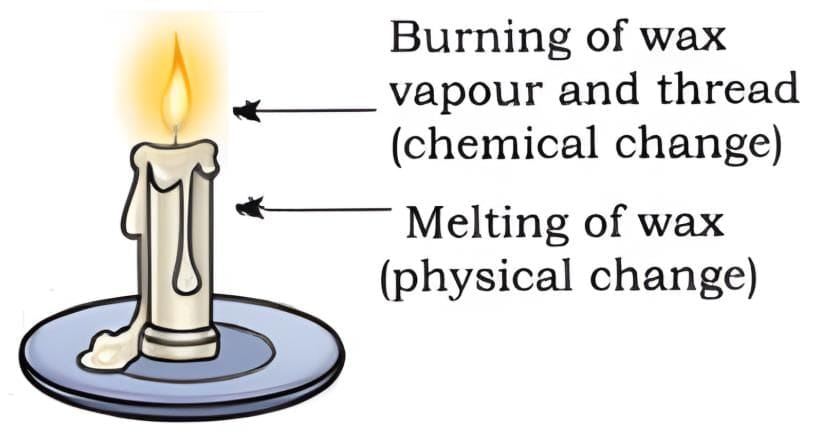
Conclusion: Burning a candle involves both physical and chemical changes occurring together.
Know a scientist: Michael Faraday

Michael Faraday, a 19th-century scientist, used candles to explain a number of physical and chemical processes. His popular lectures, titled "Chemical History of a Candle", describe melting, vapourisation and combustion in simple, clear language.
Are Changes Permanent?
Reversible changes
Definition: Reversible changes are those where the original substance or object can be restored by simple physical methods.

Examples:
- Melting ice: Ice melts to water; freezing the water reforms ice.
- Boiling water: Water becomes steam; condensing steam returns liquid water.
- Folding paper: Folding and then unfolding restores the original shape.
Irreversible changes
Definition: Irreversible changes are those where the original substance or object cannot be restored by simple physical means.

Examples:
- Chopping vegetables: Cut pieces cannot be joined back to form the whole vegetable.
- Making popcorn: Corn kernels transform into popcorn and cannot revert to kernels.
- Burning wood: Wood turns to ash; ash cannot become wood again.
Are All Changes Desirable?
Desirable changes
Desirable changes are useful or beneficial to us.
Examples:
- Milk to curd: Milk ferments to form curd which is edible and nutritious.
- Ripening fruits: Fruits change colour and become sweeter and edible.
- Cooking food: Raw ingredients change to cooked food which is easier to digest and safer to eat.
- Cutting fruits: Makes them ready to eat.
Undesirable changes
Undesirable changes are harmful or unwanted.
Examples:
- Rusting of iron: Damages tools and structures.
- Food decay: Spoils stored food, making it inedible.
Some changes may be good in one situation and bad in another. For example, decomposition of kitchen waste is undesirable when it causes foul smell and disease, but the same process is useful for making compost for plants.
Environmental impact
- Human activities such as burning fossil fuels in vehicles and industries release carbon dioxide and other pollutants into the air.
- These emissions increase atmospheric pollution and contribute to long-term changes like climate change.

Some Slow Natural Changes
Weathering of rocks
Weathering is the process by which rocks break down into smaller pieces (sediments) through physical and chemical processes. Over time these sediments contribute to soil formation.

Types of weathering
- Physical weathering: Caused by temperature changes (expansion and contraction), plant roots growing into cracks, or water freezing and expanding in crevices. These actions break rocks into smaller pieces like sand and gravel.
- Chemical weathering: Occurs when water or dissolved chemicals react with minerals in rocks, changing their composition. For example, iron-containing rocks may form a reddish layer of iron oxide (similar to rust) on exposure to water and air.
Both physical and chemical weathering over long periods help form soil, which is essential for plant growth and life on Earth.
Erosion
Erosion is the movement or removal of weathered rock, soil and sediments by natural forces such as flowing water, wind or glaciers.

Examples and effects
- Sand on riverbeds: Large rocks and stones are worn down and moved by rivers, producing fine sand and pebbles.
- Smoothing of river rocks: Flowing water rubs stones against each other, making them smooth over time.
- Landslides: Rapid erosion during landslides moves large amounts of soil and rock downhill. This is a fast physical change.
- When wind or water slows down, the carried sediments settle at the bottom; over thousands of years they can be compacted and hardened into new rocks.
These changes are usually slow, natural and often irreversible on human timescales.
Terms to Remember
- Physical changes: Only shape, size or state change; no new substances are formed. Examples: melting ice, boiling water, folding clothes.
- Chemical changes: New substances are formed. Examples: rusting, burning, lime water turning milky on passing carbon dioxide.
- Rusting: Iron reacts with air and water to form rust (iron oxide), a chemical change.
- Combustion: Burning in oxygen that produces heat and light; requires fuel, oxygen and sufficient heat (ignition temperature).
- Fireflies: Glow due to a chemical reaction called bioluminescence, which produces light with little heat.
- Burning a candle: Involves both physical changes (wax melting and evaporating) and a chemical change (vapour burning to form carbon dioxide and water).
- Reversible changes: Changes that can be reversed by simple means (e.g., melting and freezing of water).
- Irreversible changes: Changes that cannot be easily reversed (e.g., cutting, burning).
- Desirable changes: Useful or beneficial (e.g., cooking, making curd, ripening of fruits).
- Undesirable changes: Harmful or unwanted (e.g., rusting, food spoilage).
- Weathering: Breakdown of rocks into soil by physical and chemical processes.
- Erosion: Movement of soil and sediments by wind, water or ice, shaping the land.
- Michael Faraday: A scientist who explained physical and chemical processes using everyday examples such as candles in his lectures "Chemical History of a Candle".
You can practice questions from this chapter here: HOTS Questions: Changes Around Us: Physical and Chemical
FAQs on Chapter Notes: Changes Around Us - Physical and Chemical
| 1. What's the difference between physical and chemical changes with examples? |  |
| 2. How do I identify if a change around us is physical or chemical during experiments? |  |
| 3. Why does a candle burning count as a chemical change and not just melting? |  |
| 4. Can physical changes be reversed but chemical changes cannot-is this always true? |  |
| 5. What are real-life examples of physical and chemical changes I should memorise for exams? |  |







