Activity 2.1: Testing with Olfactory and Chemical Indicators
Procedure:
You have been provided with three test tubes. One contains distilled water, the second contains an acidic solution, and the third contains a basic solution.
You are provided only red litmus paper. Dip the red litmus paper into the solutions in each test tube and observe the color change.
How to identify the contents of each test tube:
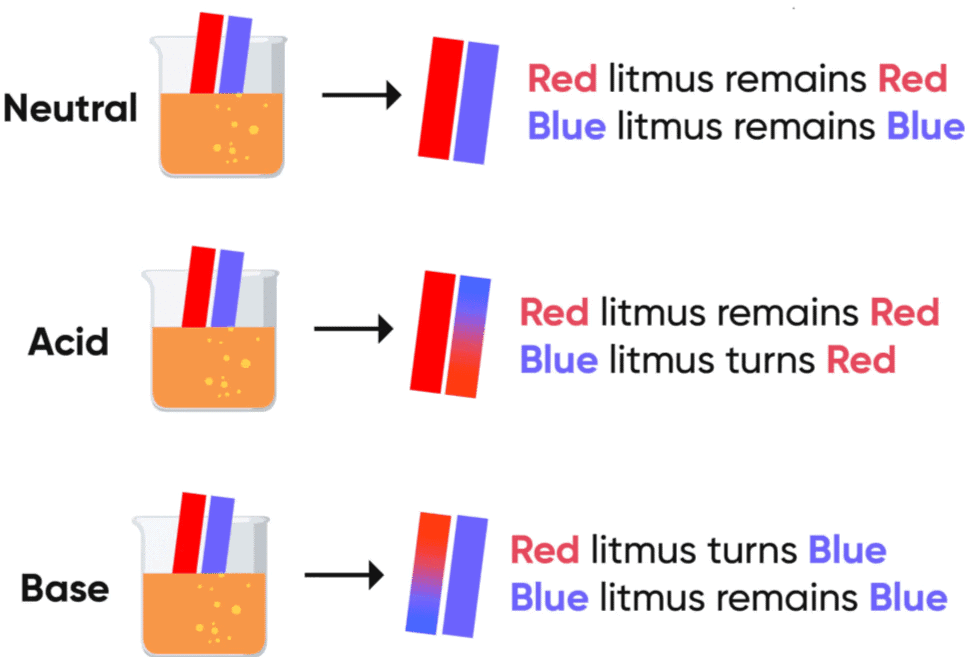
Test Tube 1 (Distilled Water): Dip the red litmus paper into this test tube. Since distilled water is neutral, the red litmus paper will remain red.
Test Tube 2 (Acidic Solution): Dip the red litmus paper into this test tube. Since the solution is acidic, the red litmus paper will remain red.
Test Tube 3 (Basic Solution): Dip the red litmus paper into this test tube. Since the solution is basic, the red litmus paper will turn blue.
Next, we will test the given solutions with different indicators. You will observe the changes in the colors of red litmus, blue litmus, phenolphthalein, and methyl orange when a drop of each solution is added to a watch-glass.
Solutions Provided:Hydrochloric acid (HCl), Sulfuric acid (H₂SO₄), Nitric acid (HNO₃), Acetic acid (CH₃COOH), Sodium hydroxide (NaOH), Calcium hydroxide (Ca(OH)₂), Potassium hydroxide (KOH), Magnesium hydroxide (Mg(OH)₂) and Ammonium hydroxide (NH₄OH)
Indicators Used:
Red Litmus Paper, Blue Litmus Paper, Phenolphthalein and Methyl Orange.
Observations:
| Solution | Red Litmus | Blue Litmus | Phenolphthalein | Methyl Orange |
|---|---|---|---|---|
| Hydrochloric Acid (HCl) | Remains Red | Remains Blue | Colourless | Red |
| Sulfuric Acid (H₂SO₄) | Remains Red | Remains Blue | Colourless | Red |
| Nitric Acid (HNO₃) | Remains Red | Remains Blue | Colourless | Red |
| Acetic Acid (CH₃COOH) | Remains Red | Remains Blue | Colourless | Red |
| Sodium Hydroxide (NaOH) | Turns Blue | Remains Blue | Pink | Yellow |
| Calcium Hydroxide (Ca(OH)₂) | Turns Blue | Remains Blue | Pink | Yellow |
| Potassium Hydroxide (KOH) | Turns Blue | Remains Blue | Pink | Yellow |
| Magnesium Hydroxide (Mg(OH)₂) | Turns Blue | Remains Blue | Pink | Yellow |
| Ammonium Hydroxide (NH₄OH) | Turns Blue | Remains Blue | Pink | Yellow |
Explanation:
Red Litmus Paper:
- In an acidic solution, the red litmus paper remains red.
- In a basic solution, the red litmus paper turns blue.
Blue Litmus Paper:
- In an acidic solution, the blue litmus paper turns red.
- In a basic solution, the blue litmus paper remains blue.
Phenolphthalein:
- In an acidic solution, phenolphthalein remains colourless.
- In a basic solution, phenolphthalein turns pink.
Methyl Orange:
- In an acidic solution, methyl orange turns red.
- In a basic solution, methyl orange turns yellow.
Conclusion:
From the above observations, we can identify whether a solution is acidic or basic by using various indicators, and the color change helps determine the pH nature of the solution.
Activity 2.2: Testing for Olfactory Indicators Using Onion, Vanilla, and Clove
Objective: To identify whether substances like onion, vanilla, and clove can be used as olfactory indicators by observing changes in their odour when exposed to acidic and basic solutions.
Procedure:
Take some finely chopped onions in a plastic bag along with some strips of clean cloth.
Tie up the bag tightly and leave it overnight in the fridge.
The cloth strips can now be used to test for acids and bases. Remove the cloth strips from the bag.
Check the odour of the cloth strips: Note the odour, which should be that of onions.
Place the strips on a clean surface.
Put a few drops of dilute HCl solution (acid) on one strip and a few drops of dilute NaOH solution (base) on the other strip.
Rinse both cloth strips with water and check the odour again.
Note your observations of any change in the odour after exposure to acid and base.
Observations:
After placing the dilute HCl solution on the strip, the odour might change due to the acidic reaction with the cloth strip.
After placing the dilute NaOH solution on the other strip, the odour might change due to the basic reaction with the cloth strip.
The odour change will help us identify whether onion can act as an olfactory indicator.

Next Steps:
Test with Vanilla Essence and Clove Oil:
Take some dilute vanilla essence and clove oil.
Check their odour initially.
Take dilute HCl solution in one test tube and dilute NaOH solution in another test tube.
Add a few drops of dilute vanilla essence to both test tubes and shake well.
Check the odour of both test tubes and record changes, if any.
Similarly, test the change in odour of clove oil with dilute HCl and dilute NaOH solutions and record your observations.
Observations with Vanilla Essence and Clove Oil:
Vanilla essence and clove oil may have distinct odours that could change when they come in contact with acids or bases.
The odour of vanilla essence and clove oil will be tested for their ability to act as olfactory indicators by noting any changes after being exposed to the acidic (HCl) or basic (NaOH) solutions.
Conclusion:
Based on the observations from the cloth strips and the changes in odour with vanilla essence and clove oil, you can conclude whether onion, vanilla, or clove can act as olfactory indicators by their ability to show a change in odour when exposed to acidic and basic solutions.
Onion may act as an olfactory indicator if there is a noticeable change in odour after exposure to acid and base.
Vanilla and clove oil may also act as olfactory indicators if their odour changes when tested with acidic or basic solutions.
Activity 2.3: Reaction of Zinc with Dilute Acids
Objective: To observe the reaction of zinc with different acids and test the properties of the gas evolved during the reaction.
Apparatus: Test tubes, Zinc granules, Dilute sulfuric acid (H₂SO₄), Soap solution, Burning candle, Other acids (HCl, HNO₃, CH₃COOH).
Procedure:
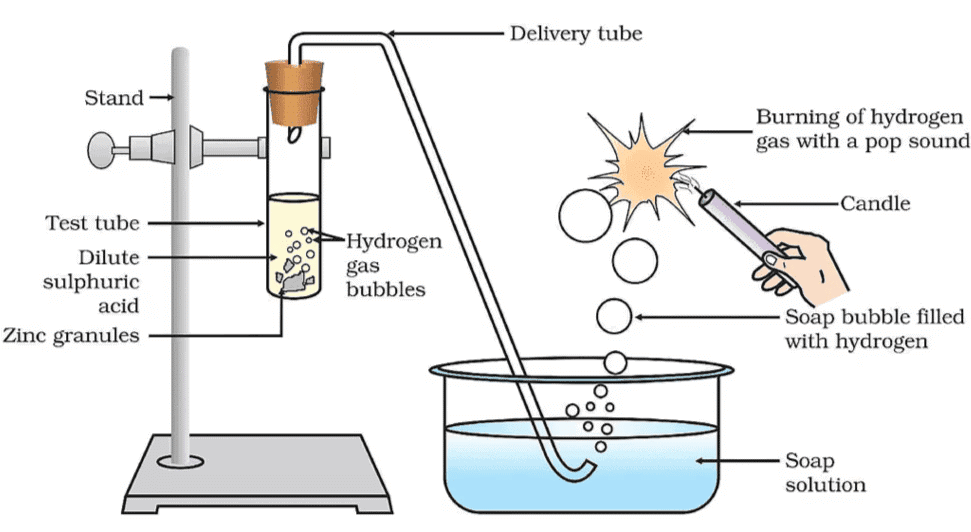
Set up the apparatus as shown in Fig. 2.1 (This usually involves a test tube, a delivery tube for the evolved gas, and a soap solution).
Take about 5 mL of dilute sulfuric acid in a test tube.
Add a few pieces of zinc granules to the dilute sulfuric acid.
Observe the surface of the zinc granules. You should see bubbles forming on the surface of the zinc.
Pass the gas being evolved through the soap solution.
Why are bubbles formed in the soap solution? The gas produced is hydrogen gas (H₂), and when it passes through the soap solution, it forms bubbles.
Take a burning candle near a gas-filled bubble.
Observe what happens. The hydrogen gas will burn with a pop sound, confirming the presence of flammable hydrogen gas.
Repeat the activity with other acids like hydrochloric acid (HCl), nitric acid (HNO₃), and acetic acid (CH₃COOH).
With each acid, the observations will be similar, but the rate of reaction and the intensity of the reaction might vary depending on the acid used.
Observations:
On the surface of zinc granules: You will observe bubbles forming, which is hydrogen gas being evolved.
Bubbles in soap solution: The bubbles form because hydrogen gas is passing through the soap solution, creating a foam of hydrogen bubbles.
When a burning candle is brought near a bubble: The hydrogen gas inside the bubble burns with a pop sound, confirming the presence of hydrogen.
- With other acids (HCl, HNO₃, CH₃COOH): You will still observe the evolution of hydrogen gas in all cases. The intensity of the reaction might be stronger with HCl due to its stronger acidic nature compared to acetic acid (CH₃COOH), which is weaker.
Reaction of Zinc with Hydrochloric Acid (HCl): Zn (s)+2HCl (aq)→ZnCl₂ (aq)+H₂ (g)
Reaction of Zinc with Sulfuric Acid (H₂SO₄): Zn (s)+H₂SO₄ (aq)→ZnSO₄ (aq)+H₂ (g)
Reaction of Zinc with Acetic Acid (CH₃COOH): Zn (s)+2CH₃COOH (aq)→Zn(CH₃COO)₂ (aq)+H₂ (g)
Conclusion:
In this activity, zinc reacts with various dilute acids to release hydrogen gas (H₂). The reaction can be confirmed by passing the gas through a soap solution to form bubbles and bringing a burning candle near the bubbles to observe the pop sound.
The observations in all cases are similar, as zinc reacts with acids to produce hydrogen gas, but the rate of reaction can vary with the strength of the acid.
Activity 2.4: Reaction of Zinc with NaOH Solution
Objective: To observe the reaction of zinc with sodium hydroxide solution and identify the gas evolved during the reaction.
Apparatus: Test tubes, Granulated zinc metal, Sodium hydroxide (NaOH) solution, Soap solution and Burning candle
Procedure:
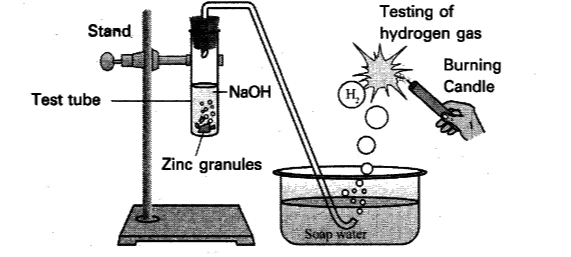
Take a few pieces of granulated zinc metal in a test tube.
Add 2 mL of sodium hydroxide (NaOH) solution to the test tube containing zinc.
Warm the contents of the test tube gently.
Repeat the steps from Activity 2.3:
Pass the gas being evolved through the soap solution.
Observe the formation of bubbles in the soap solution.
Take a burning candle near the gas-filled bubbles and observe what happens.
Observations:
When zinc reacts with sodium hydroxide solution, bubbles of gas will form.
The gas produced is hydrogen gas (H₂).
When the gas passes through the soap solution, bubbles will form because the hydrogen gas is trapped in the soap film.
When you bring a burning candle near the bubbles, the hydrogen gas burns with a pop sound, confirming the presence of hydrogen gas.
 Reaction of Zinc with Sodium Hydroxide SolutionChemical Reaction: Zn (s)+2NaOH (aq)→Na₂ZnO₂ (aq)+H₂ (g)
Reaction of Zinc with Sodium Hydroxide SolutionChemical Reaction: Zn (s)+2NaOH (aq)→Na₂ZnO₂ (aq)+H₂ (g)
Explanation:
- Zinc reacts with sodium hydroxide (NaOH) to form sodium zincate (Na₂ZnO₂) and hydrogen gas (H₂).
- The production of hydrogen gas is similar to the reaction with acids, and it can be tested by observing the bubbles and the pop sound when exposed to a burning candle.
Conclusion:
- The reaction between zinc and sodium hydroxide solution produces hydrogen gas (H₂), which can be confirmed by the formation of bubbles in the soap solution and the pop sound when brought near a burning candle.
- This is a redox reaction where zinc is oxidized and hydrogen ions from sodium hydroxide are reduced to hydrogen gas.
Activity 2.5: Reaction of Sodium Carbonate and Sodium Bicarbonate with HCl
Objective: To observe the reactions of sodium carbonate (Na₂CO₃) and sodium bicarbonate (NaHCO₃) with dilute hydrochloric acid (HCl) and test the gas evolved using lime water (calcium hydroxide solution).
Apparatus: Two test tubes, Sodium carbonate (Na₂CO₃) - 0.5 g, Sodium bicarbonate (NaHCO₃) - 0.5 g, Dilute hydrochloric acid (HCl), Lime water (calcium hydroxide solution), Delivery tube to pass gas through lime water and Cork or stopper for sealing the test tube.
Procedure:
Label two test tubes as A and B.
In Test Tube A, take about 0.5 g of sodium carbonate (Na₂CO₃).
In Test Tube B, take about 0.5 g of sodium bicarbonate (NaHCO₃).
Add 2 mL of dilute HCl to both test tubes (A and B).
Observe the reactions in both test tubes. Bubbles should form in both cases due to the production of gas.
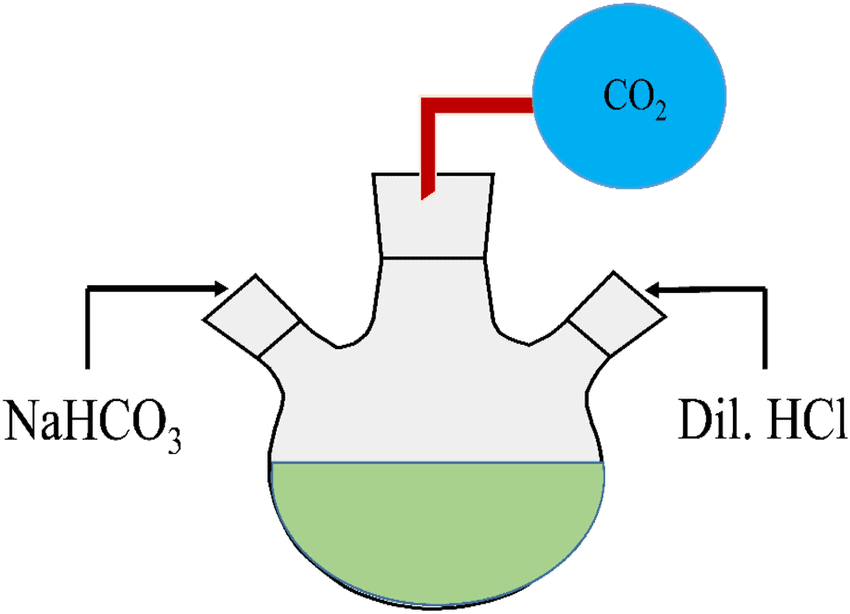
Pass the gas produced in each case through lime water (calcium hydroxide solution) as shown in Fig. 2.2 and record your observations.
Observations:
1. Test Tube A (Sodium Carbonate + HCl):
- When sodium carbonate (Na₂CO₃) reacts with hydrochloric acid (HCl), it produces carbon dioxide (CO₂) gas.
- You will observe bubbling in both test tubes due to the evolution of gas.
- Passing the gas through lime water: The gas reacts with lime water and forms a milky white precipitate of calcium carbonate (CaCO₃), indicating the presence of carbon dioxide (CO₂) gas.
CERT
Reaction:
Na₂CO₃ (aq)+2HCl (aq)→2NaCl (aq)+H₂O (l)+CO₂ (g)
CO₂ (g)+Ca(OH)₂ (aq)→CaCO₃ (s)+H₂O (l)
2. Test Tube B (Sodium Bicarbonate + HCl):
- When sodium bicarbonate (NaHCO₃) reacts with hydrochloric acid (HCl), it also produces carbon dioxide (CO₂) gas.
- You will observe bubbling in this test tube as well due to gas production.
- Passing the gas through lime water: The gas reacts with lime water and forms a milky white precipitate of calcium carbonate (CaCO₃), again confirming the presence of carbon dioxide (CO₂) gas.
Reaction:
NaHCO₃ (aq)+HCl (aq)→NaCl (aq)+H₂O (l)+CO₂ (g)CO₂ (g)+Ca(OH)₂ (aq)→CaCO₃ (s)+H₂O (l)
Conclusion:
- Both sodium carbonate (Na₂CO₃) and sodium bicarbonate (NaHCO₃) react with dilute hydrochloric acid (HCl) to produce carbon dioxide (CO₂) gas.
- The formation of a milky precipitate in lime water confirms that carbon dioxide (CO₂) gas is produced in both reactions.
- The key difference between the reactions of sodium carbonate and sodium bicarbonate is that sodium bicarbonate reacts more readily with HCl and produces CO₂ at a lower temperature.