Entropy Change & Gibb's Phase Rule
What is Entropy?
Entropy is a state function that measures the degree of randomness or disorder in a macroscopic system. While a decrease in enthalpy can contribute to a process's spontaneity, it doesn't fully explain it, especially for spontaneous endothermic reactions. For instance, in the isothermal reversible expansion of an ideal gas, the enthalpy change (ΔH) is zero, yet the process occurs spontaneously. Entropy provides a way to understand this spontaneity.
Internal interactions between various subsystems give multiple entropy changes.
What is Entropy Change?
As a state function, entropy change (ΔS) depends only on the initial and final states of a system, not on the path taken or whether the process is reversible or irreversible. Greater disorder in an isolated system corresponds to higher entropy. In chemical reactions, entropy change reflects the rearrangement of atoms or ions. If the products are more disordered than the reactants, entropy increases. For example, crystalline solids typically have lower entropy compared to other solids due to their ordered structure.
Adding heat to a system increases molecular motion, enhancing randomness and thus entropy. A system at a higher temperature generally has greater randomness than at a lower temperature. The entropy change is inversely proportional to the system's temperature, meaning heat added at a lower temperature causes a larger increase in randomness compared to the same heat added at a higher temperature.
Expression for entropy change
The general expression for entropy change can be given by: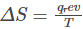
Where,
q = heat
T = temperature
For a spontaneous process, entropy change for the system and the surrounding must be greater than zero, that is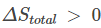
The general expression can be given as: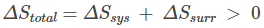
Gibbs Phase Rule
Josiah Willard Gibbs, a prominent American scientist, made significant contributions to thermodynamics, developing key rules and formulas widely used in science. One of his most notable achievements is the Gibbs Phase Rule, a fundamental principle derived from thermodynamic concepts. This rule establishes a relationship between the number of chemical components, their phases, and variables like temperature and pressure to determine the equilibrium characteristics of a chemical system.
What is the Gibbs Phase Rule?
Proposed by Josiah Willard Gibbs in the 1870s, the Gibbs Phase Rule is a critical concept in physics, chemistry, and mathematics. It describes how chemical components in different phases-solid, liquid, or gas-interact under the influence of temperature and pressure to define the equilibrium state of a system. The rule is essential in applications such as geothermal barometry and assessing the acidity or oxygen state of chemicals. A key principle of the Gibbs Phase Rule is that an increase in the number of components leads to greater variance (degrees of freedom), while an increase in the number of phases reduces the variance.
Understanding the Gibbs Phase Rule
The Gibbs Phase Rule is vital for studying the geological history and behavior of naturally occurring minerals. Minerals exhibit varied reactions under changing pressure, temperature, acidity, or basicity. The rule laid the groundwork for advancing research into these geological processes, enabling scientists to characterize chemical reactions, determine equilibrium conditions, and analyze the effects of temperature and pressure changes on mineral systems.
Example of the Gibbs Phase Rule
Consider water (H₂O) as a single component existing in all three phases (solid, liquid, gas). For pure liquid water at a specific pressure (e.g., 1 atm), the system has one component (C = 1) and one phase (P = 1). Using the Gibbs Phase Rule formula, P + F = C + 2, the degrees of freedom (F) are calculated as F = 1 + 2 - 1 = 2. This means temperature and pressure can be independently varied, illustrating the system's flexibility.
Key Terminology and Applications
The Gibbs Phase Rule is widely applied in geothermal barometry, physicochemical studies, and phase diagram construction to represent relationships between variables. Its formula is:
P + F = C + 2
Where:
- P represents the number of phases (solid, liquid, or gas) in the system.
- C indicates the number of chemical components (e.g., SiO₂, CaO), which may consist of single or multiple constituents.
- F denotes the degrees of freedom or variance, reflecting the number of independent variables (e.g., temperature, pressure) that can be adjusted without altering the system's equilibrium.
The rule facilitates the observation of how temperature and pressure affect chemical behavior, equilibrium determination, and the mapping of phase diagrams to visualize these interconnections.
Gibbs Phase Rule Derivation
Gibbs Phase rule for the General System is determined by the derivation of the fundamental thermodynamics principle. The Gibbs-Duhem equation represents a relationship between temperature, pressure, and the chemical potential of the components.
dG=Vdp-Sdt+Nidi
To simplify the equation, the composition of each phase is expressed as P(C-1), and the number of variances is expressed as P(C-1)+2. Thus, the number of equilibria is expressed as E=C(P-1). The Gibbs Phase rule derivation is determined as:
F={P(C-)+2}-{C(P-1)}
F={CP-P+2-CP+C}
F=C-P+2
Whereas, The Gibbs Phase rule for General System is written as,
P+F=C+2
Conclusion
Gibbs Phase rule is an important aspect of the geological and scientific field. It represents a relationship between the number of the chemical components that are present in the respective phase, under the influence of the variables, that is, temperature and pressure, to determine the equilibrium of the system. Gibbs Phase rule has been used in geothermal barometry to estimate the pressure and temperature, and in Physicochemical conditions, i.e., determining the acidity and the oxidation state of the chemical reactions, etc. Gibbs Phase Rule derivation is determined from the relationship of thermodynamics, so to understand the concept of the Gibbs phase rule formula, it is required to have a basic knowledge of thermodynamics.
FAQs on Entropy Change & Gibb's Phase Rule
| 1. What is Entropy in thermodynamics? |  |
| 2. What is Entropy Change and how is it calculated? |  |
| 3. What is the Gibbs Phase Rule? |  |
| 4. How is the Gibbs Phase Rule derived? |  |
| 5. Why is understanding Entropy Change and the Gibbs Phase Rule important for UPSC aspirants? |  |





