NCERT Based Activity: A Journey through States of Water
Activity 8.1: Let us observe
- Put an ice cube in a cup, leave it on the table and observe.
- Ice gets converted into water.
Q: What can you conclude from the observations?
Ans: Ice gets converted into water.
Q: Does this mean that ice and water are the same substance?
Ans: Yes, ice and water are the two forms of the same substance.
These forms are also called states. These different states of water show many differences in their behaviour. Water flows but ice does not. Water splashes but ice does not.
Activity 8.2: Let us investigate
- Take a tablespoon of water on a steel plate as shown in Fig. 8.1.
- Observe whether water seeps through to the other side of the plate or not.
- Keep observing this at regular intervals until the water completely disappears.

Q: What do you infer?
Ans: This water gets converted into gaseous state called water vapour.
Q: Is this activity enough to come to the conclusion that water does not seep through a steel plate?
Ans: This activity is enough to come to the conclusion that water does not seep through a steel plate.
Q: If water does not seep through the steel plate. Then, where has the water gone?
Ans: This water gets converted into gaseous state called water vapour.
Activity 8.3: Let us experiment
- Take cold water in a glass tumbler.
- Add a few ice cubes into it as shown in Fig. 8.2.

- Leave it undisturbed for five minutes and observe it.
- Record your observations and the questions that arise in your mind in Table 8.1. You can also touch the outer surface of the glass tumbler to feel if there is any change.
- You may have many observations and questions here.

- One observation that arises in Aavi's mind is, "There are some water droplets (tiny drops) appearing on the outer surface of the glass tumbler." Initially, water droplets are deposited and these droplets combine together to form bigger drops. You can also try the above process with a metal container. You may be curious about where the water droplets come from.
Q: Record your observations and the questions that arise in your mind in Table 8.1.
Ans:
Q: Suggest possible reasons explaining the appearance of water droplets on the outer surface of the glass tumbler.
Ans: When the water vapour present in the air comes in contact with a cold surface, it forms water droplets.
Activity 8.4: Let us measure
- Take a glass tumbler half-filled with water containing a few ice cubes. Cover it with a small steel plate. Weigh it on a digital weighing balance.
- Observe the reading on the balance and record the weight after every five minutes.
- Continue observing for 30 minutes. Record your observations in Table 8.2.

- You may observe some water droplets on the glass tumbler. Water vapour from the air comes in contact with the cold surface of the glass tumbler and gets converted into water droplets on the glass tumbler through condensation. There is an increase in the reading on the digital weighing balance.
Q: Predict what will happen to the mass of cold water kept on the digital weighing balance. Will it increase or decrease or remain the same?
Ans: There is an increase in the reading on the digital weighing balance.
Q: Record your observations in Table 8.2.
Ans: There is an increase in the reading on the digital weighing balance.
Q: Do your findings match with your predictions? Explain your observations.
Ans: You may observe some water droplets on the glass tumbler. Water vapour from the air comes in contact with the cold surface of the glass tumbler and gets converted into water droplets on the glass tumbler through condensation. There is an increase in the reading on the digital weighing balance.
Q: Can we conclude that water is not seeping through the wall of the tumbler? Can we also conclude that the water collected outside the tumbler is only due to condensation?
Ans: No, we cannot say that conclusively from Activity 8.4.
Q: What more can you do to show that water is not seeping from the glass tumbler? How would you modify Activity 8.4 to find the answer?
Ans: Repeat Activity 8.4 with the following modifications- Mark the water level on the glass tumbler with a permanent marker or a visible tape.
Q: What do you observe? What can you conclude from this?
Ans: Water level in the glass tumbler does not go down but the extra water gets collected on the outer surface of the glass tumbler. This activity shows that water is not seeping from the glass tumbler and the extra water is getting collected because of condensation.
Activity 8.5: Let us identify
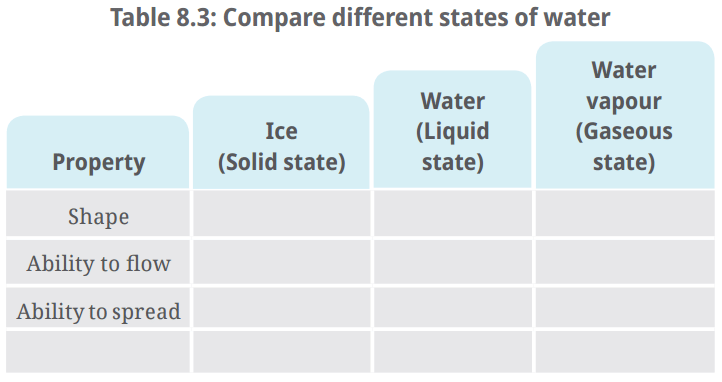
- Put an ice cube in one container and transfer it to another container of different shape. Record your observations in Table 8.3.
- Pour water from one container to another container of a different shape.
Q: What changes do you notice in the shape of the ice cube?
Ans: Ice (solid state) retains its shape irrespective of the container in which it is placed.
Q: Observe how water behaves compared to the ice cube and make a record. Did you notice how water flows from one container to the other? What happens to its shape?
Ans: Water (liquid state) flows and changes its shape. Water does not have a fixed shape. It takes up the shape of the container in which it is kept.
Q: Pour water on a clean surface and observe how it spreads.
Ans: Water also has the property to spread while keeping the volume constant.
Q: When water gets converted into water vapour, how does this water vapour spread? Compare this with the spreading behaviour of water.
Ans: Water vapour (gaseous state) exhibits a property of spreading out in the entire available space.
Q: Record your observations in Table 8.3.
Ans:
Activity 8.6: Let us complete the diagram
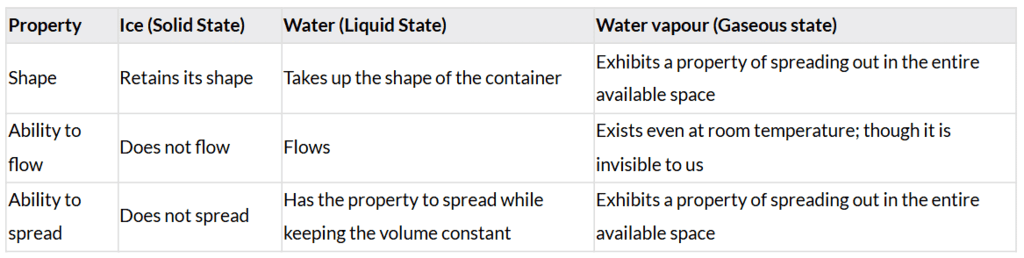
Q: Fill up the blank boxes in Fig. 8.5 marked as A, B, C and 1, 2, 3, 4 for conversion of different states of water using the words given in the box. Two words have been filled for you.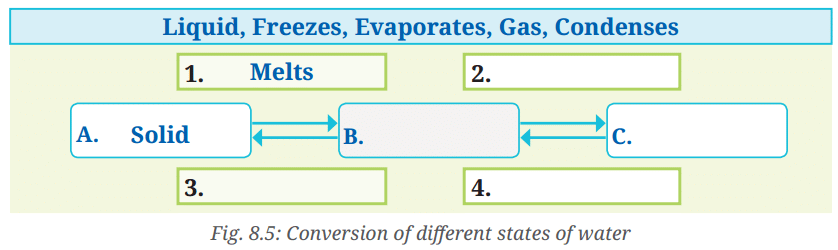
Ans:
A. Solid
B. Liquid
C. Gas
1. Melts
2. Evaporates
3. Condenses
4. Freezes
Activity 8.7: Let us investigate
- Take water in a small cap of a bottle (you may use sanitiser in place of water).
- Take the same amount of water in a plate. The exposed area of water in the bottle cap and the plate are different.
- Keep both of them near each other.

- Record the time taken for the water to completely evaporate in each case in Table 8.4.

Q: What conditions did we keep the same?
Ans: The amount of water, the environmental conditions (temperature, humidity, air movement), and the proximity of the bottle cap and plate were kept the same.
Q: What did we change in this investigation?
Ans: The exposed surface area of water was changed (smaller in the bottle cap, larger in the plate).
Q: What did we measure?
Ans: The time taken for the water to completely evaporate in each case.
Q: Record the time taken for the water to completely evaporate in each case in Table 8.4.
Ans: If you spread out water on a plate, its area exposed to air is larger. Therefore, evaporation is faster.
Q: What can you conclude from this investigation?
Ans: If you spread out water on a plate, its area exposed to air is larger. Therefore, evaporation is faster.
Activity 8.8: Let us explore
- Take identical caps of two bottles.
- Pour equal amount of water in each of the cap.
- Place one of the cap in sunlight and keep the other in shade as shown in Fig. 8.6.
- Observe the two caps of bottles after every 15 minutes.
- Record the time taken for the water to completely evaporate in each case.
- You can also repeat this activity on a windy or a rainy day, and record your observations.
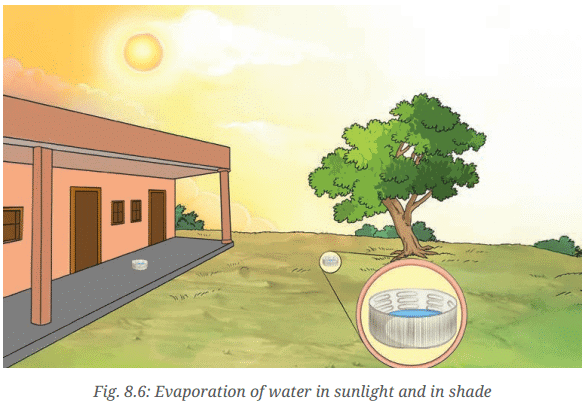
Q: Record the time taken for the water to completely evaporate in each case.
Ans: Water evaporates faster from the cap kept in sunlight compared to the cap kept in shade.
Q: What conclusions can you draw from Activity 8.8 and other similar experiences?
Ans: Water evaporates faster from the cap kept in sunlight compared to the cap kept in shade. It is a common observation that clothes dry faster on a hot sunny day. It is once again a common observation that clothes dry faster on a windy day. With the increase in the movement of air, water evaporates faster.
Q: Do clothes dry faster or slower on a windy day?
Ans: It is once again a common observation that clothes dry faster on a windy day.
Activity 8.9: Let us make a model
- Take two earthen pots of different sizes.
- Fill the bottom of the larger pot with a layer of sand.
- Place the smaller pot into the centre of the larger one as shown in Fig. 8.7.
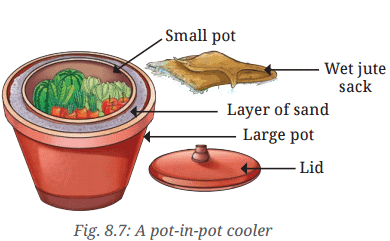
- Fill the gap between the pots with more sand.
- Pour water in the sand area.
- Place a lid or wet jute sack to cover the top of the smaller pot.
- Allow 4-5 hours for the mini pot-in-pot cooler to cool down. The time range can be influenced by many conditions.
- Keep some vegetables and fruits in it and observe for a week on a daily basis to check for the freshness of the vegetable Compatibilty Mode vegetables and fruits kept inside the cooler. You will have to add water regularly to keep the sand moist.
Q: Observe and discuss how it creates a cooling effect inside the pots.
Ans: Water seeps through the surface of the earthen pot and evaporates, which imparts a cooling effect on the water.
Q: For how many days can the vegetables and fruits be kept fresh in it?
Ans: The time range can be influenced by many conditions.
Q: What are the conditions which can affect the number of these days?
Ans: The time range can be influenced by many conditions.
Q: What else can be used in place of sand for better cooling?
Ans: Other porous substances that can hold water and facilitate evaporation could be used in place of sand for better cooling.
Activity 8.10: Let us engage in a group activity
- Take an empty discarded one litre plastic bottle. Pour about one cup of water into it.
- Handle burning paper carefully.
- Close the lid tightly. Now quickly squeeze and release the bottle continuously for about 2-3 minutes. Observe the space above the water in the bottle.
- Repeat the same activity after adding a small burnt piece of newspaper into the water.
Q: What will you observe?
Ans: In this case, you will observe some haziness (clouds) above the water in the bottle.
- The burnt newspaper provides very small invisible dust particles, around which water vapour condenses and forms clouds.
Activity 8.11: Let us understand the process
- Label Fig. 8.9 using arrows shown and the words given in the box to show where water is stored, how water changes its state and where it moves.
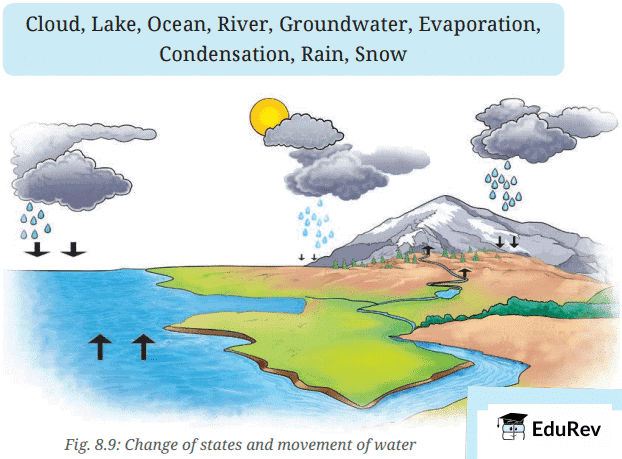
Ans: The labeling is as follows: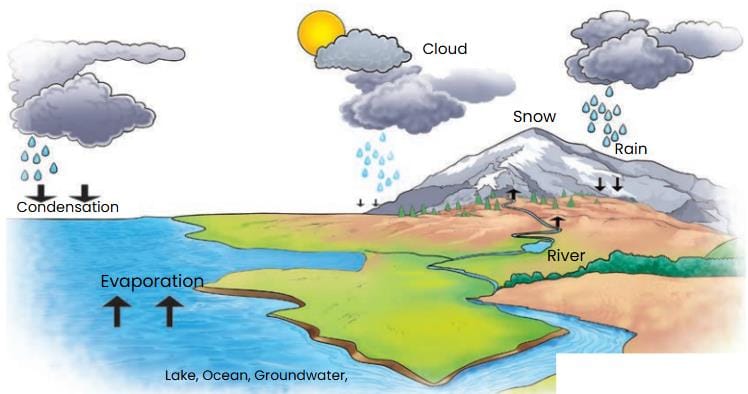
- The water from the ocean and the Earth's surface evaporates into the atmosphere as vapour and returns as rain, hail or snow, ultimately flowing back to the oceans. This circulation of water is known as the water cycle.
FAQs on NCERT Based Activity: A Journey through States of Water
| 1. What are the three states of water? |  |
| 2. How does water change from one state to another? |  |
| 3. What is the importance of the water cycle? |  |
| 4. Why is it essential to understand the states of water for daily life? |  |
| 5. How does temperature affect the state of water? |  |







