Chapter Notes: Miscellaneous Topics of Pathology
Tumors of Salivary Glands
Salivary gland tumors are most commonly found in the parotid gland (65-80%), followed by the submandibular gland (10%), and minor salivary glands like the sublingual glands (10-15%). The likelihood of a tumor being cancerous is lower in larger glands, making tumors in minor salivary glands more likely to be malignant. In contrast, tumors in the parotid gland are mostly benign.
These tumors primarily affect adults and are slightly more common in females, although Warthin tumors are more frequently found in males.
1. Pleomorphic Adenoma or Mixed Tumors
- These are the most prevalent benign tumors, originating from a combination of ductal (epithelial) and myoepithelial cells, exhibiting characteristics of both epithelial and mesenchymal tissues.
- Around 60% of tumors in the parotid gland are mixed tumors.
- Risk factors include exposure to radiation.
- Pleomorphic adenomas typically present as rounded, well-defined masses, usually not exceeding 6 cm in size.
- The epithelial components resemble ductal or myoepithelial cells and are often situated within a background of loose myxoid tissue, which may contain cartilage and, rarely, bone.
- These tumors usually manifest as painless, slowly growing, and mobile lumps.
- When cancer develops within a pleomorphic adenoma, it is referred to as carcinoma ex pleomorphic adenoma or a malignant mixed tumor, with the risk of malignancy increasing over time.
2. Warthin's Tumor (Papillary Cystadenoma Lymphomatosum)
- Warthin's tumor is the second most common benign salivary gland tumor, primarily found in the parotid gland.
- This tumor is more prevalent in males and typically occurs in individuals aged 50 to 70.
- Approximately 10% of cases are multifocal, and 10% are bilateral.
- There is a higher incidence of Warthin's tumor among smokers.
- These tumors are generally round to oval, encapsulated masses ranging from 2 to 5 cm in diameter.
- Histologically, Warthin's tumors exhibit cystic spaces lined with neoplastic epithelial cells supported by a dense lymphoid stroma.
- The distinctive two-layer lining consists of columnar cells with abundant, finely granular, eosinophilic cytoplasm, characteristic of an oncocytic appearance, resting on a layer of cuboidal to polygonal cells.
3. Mucoepidermoid Carcinoma
- Mucoepidermoid carcinomas primarily occur in the parotid glands (60 to 70%) and are the most common type of malignant tumor in salivary glands.
- These tumors can grow up to 8 cm in size, lack well-defined capsules, and often invade surrounding tissues.
- They are associated with a specific genetic alteration, t(11;19), leading to the formation of a fusion gene involving MECT1 and MAML2.
- Histologically, mucoepidermoid carcinomas are characterized by cords, sheets, or cystic structures composed of squamous, mucous, or intermediate cells.
- These hybrid cell types exhibit squamous features, with varying sizes of mucus-filled vacuoles, which are best visualized using mucin stains.
- Other salivary gland tumors include adenoid cystic carcinoma and acinic cell tumor.
- Adenoid cystic carcinoma, although relatively rare, is found in 50% of cases in minor salivary glands, particularly in the palate.
- These tumors have the ability to invade around nerves, possess a high recurrence rate, and over 50% have the potential to metastasize to distant organs such as bone, liver, and brain.
- The acinic cell tumor consists of cells resembling normal serous acinar cells of salivary glands, primarily arising in the parotid glands, with few cases occurring in submandibular glands.
- These tumors typically feature clear cytoplasm, and the cells may be solid or vacuolated, forming sheets or various patterns.
- Histologically, acinic cell tumors display small cells with dark, compact nuclei and scant cytoplasm, arranged in tubular, solid, or cribriform patterns, with spaces filled with a hyaline material suggesting excess basement membrane.
Other Important Tumors
A. Neuroblastoma
- Neuroblastoma is the most common solid tumor in children outside the brain, often diagnosed in infants.
- Most cases occur sporadically, but familial cases (1-2%) are linked to mutations in the anaplastic lymphoma kinase ( ALK. gene.
- The primary sites of origin include:
- Adrenal medulla (40%)
- Para-vertebral sympathetic chain in the abdomen (25%)
- Posterior mediastinum (15%)
- Clinical Types:
- Pepper type. Seen in stillborns and young infants, spreads to the liver and lymph nodes, then to the lungs and bones.
- Hutchinson type. Secondary growth in the orbit, meninges, skull, and long bones, affecting children up to 15 years old.
- Histologically, neuroblastoma is a small round blue cell tumor characterized by a central area of neuropil surrounded by tumor cells forming Homer-Wright pseudorosettes.
- Tumor Cell Markers:
- Tumor cells exhibit positivity for neuron-specific enolase and contain dense core granules.
- Some cells can mature into ganglion cells, leading to ganglioneuroblastoma, while better-differentiated tumors with fewer neuroblasts are called ganglioneuromas.
- The presence of Schwannian stroma, mature Schwann cells, and fibroblasts is necessary for diagnosing ganglioneuroblastoma and ganglioneuromas.
- Metastasis: Metastasis occurs early and widely, with common sites including the liver, lungs, bone marrow, and bones. Approximately 60-80% of children present with stage 3 or 4 tumors.
- Stage 4S: This special stage indicates a local primary tumor with limited spread to the skin, liver, and/or bone marrow, applicable to infants under 1 year.
Prognostic Factors
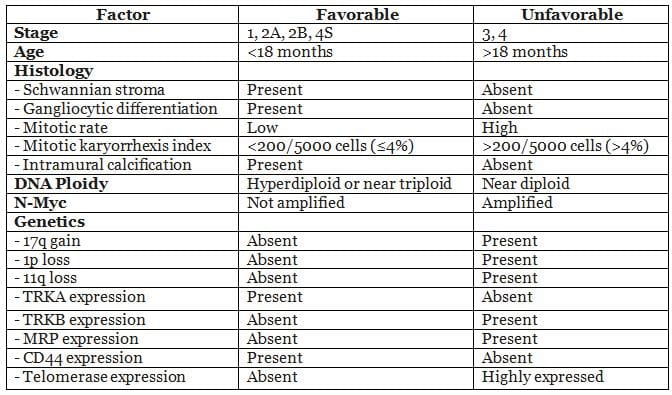
B. Retinoblastoma
- Retinoblastoma is the most common malignant eye tumor in children, with 90% of cases diagnosed before the age of 7.
- Hereditary retinoblastoma affecting both eyes is known as bilateral retinoblastoma.
- When bilateral retinoblastoma is accompanied by a pineal gland tumor, it is referred to as Trilateral retinoblastoma.
- These tumors originate from the neuroepithelium of the retina and are characterized by the presence of Flexner-Wintersteiner rosettes, which consist of clusters of cuboidal or short columnar cells surrounding a central lumen, with nuclei displaced away from the lumen, resembling photoreceptor-like elements.
- Retinoblastoma also exhibits Homer-Wright rosettes, where cells are arranged radially around a central tangle of fibrils, and Fleurettes, indicating the photoreceptor differentiation of tumor cells.
C. Wilms' Tumor
Wilms' tumor is the most prevalent primary kidney tumor in children in the United States. It is associated with three distinct congenital malformation syndromes:
- WAGR Syndrome: Characterized by aniridia, genital anomalies, and intellectual disability, with a 33% risk of developing Wilms' tumor. Patients have germline deletions of the WT1 and PAX6 genes, both located on chromosome 11p13.
- Denys-Drash Syndrome: Features gonadal dysgenesis (male pseudohermaphroditism) and early-onset kidney disease leading to renal failure, with diffuse mesangial sclerosis as the hallmark glomerular lesion. Germline WT1 mutations are present, and affected individuals are also at higher risk for gonadoblastomas.
- Beckwith-Wiedemann Syndrome: Marked by organomegaly, macroglossia, hemihypertrophy, omphalocele, and adrenal cytomegaly. The genetic locus, termed "WT2," is on chromosome 11p15.5. Patients face increased risks for hepatoblastoma, adrenocortical tumors, rhabdomyosarcomas, and pancreatic tumors in addition to Wilms' tumors.
Morphology:
Grossly, Wilms' tumors typically appear as large, solitary, well-defined masses. On sectioning, they are soft, uniform, and tan to gray, with occasional hemorrhage, cysts, or necrosis. Microscopically, they exhibit a triphasic pattern of blastemal, stromal, and epithelial cells in most cases.
D. Langerhans Cell Histiocytosis (LCH)
LCH encompasses several conditions:
- Letterer-Siwe Syndrome: A multifocal, multisystem form of LCH.
- Pulmonary Langerhans' Cell Histiocytosis: Commonly seen in adult smokers, often regressing after smoking cessation.
- Eosinophilic Granuloma: Includes the Hand-Schüller-Christian triad, defined by calvarial bone defects, diabetes insipidus, and exophthalmos.
Tumor cells express HLA-DR, S-100, and CD1a, with characteristic Birbeck granules in the cytoplasm, resembling tennis rackets under electron microscopy.
E. Mediastinal Tumors
The mediastinum, located between the pleural sacs, is divided into three compartments:
- Anterior Mediastinum: Extends from the sternum to the pericardium and brachiocephalic vessels, containing the thymus, anterior mediastinal lymph nodes, and internal mammary arteries and veins.
- Middle Mediastinum: Lies between the anterior and posterior mediastinum, housing the heart, ascending and transverse aorta, venae cavae, brachiocephalic arteries and veins, phrenic nerves, trachea, main bronchi, adjacent lymph nodes, and pulmonary arteries and veins.
- Posterior Mediastinum: Bounded by the pericardium and trachea anteriorly and the vertebral column posteriorly, containing the descending thoracic aorta, esophagus, thoracic duct, azygos and hemiazygos veins, and posterior mediastinal lymph nodes.
Common Lesions:
- Anterior Mediastinum: Thymomas, lymphomas, teratomas, and thyroid masses are most frequent.
- Middle Mediastinum: Vascular masses, lymph node enlargement (from metastases or granulomatous disease), and pleuropericardial or bronchogenic cysts are common.
- Posterior Mediastinum: Neurogenic tumors, meningoceles, meningomyeloceles, gastroenteric cysts, and esophageal diverticula are typical.
Thymoma
The thymus, which develops from the third and fourth pharyngeal pouches, is located in the anterior mediastinum. It is made up of epithelial and stromal cells, as well as lymphoid precursors.
Thymoma occurs when the epithelial cells of the thymus become cancerous. Thymoma is the most common cause of an anterior mediastinal mass in adults. These tumors are usually found in people during their fifth and sixth decades of life. About 90% of thymomas are located in the anterior mediastinum. Thymomas are primarily benign tumors, but they have the potential to become malignant. The epithelial part of the tumor may consist mainly of round or oval cells from the cortex, spindle-shaped cells from the medulla, or a combination of both.
Clinical Presentation and Associated Conditions
- Thymomas can cause symptoms in 40% of patients due to pressure on mediastinal structures or their association with myasthenia gravis.
- Other related syndromes that may occur alongside myasthenia gravis include:
- Acquired hypogammaglobulinemia
- Pure red cell aplasia
- Graves' disease
- Pernicious anemia
- Dermatomyositis-polymyositis
- Cushing syndrome
- Thymomas may be linked to the Epstein-Barr virus.
WHO Histologic Classification of Thymus Tumours
- Type A: Medullary thymoma
- Type AB: Mixed thymoma
- Type B1: Predominantly cortical thymoma
- Type B2: Cortical thymoma
- Type B3: Well-differentiated thymic carcinoma
- Type C: Thymic carcinoma
The genetic changes in thymomas are not well understood. Some information indicates that the Epstein-Barr virus may be associated with these tumours.
Scleroderma
Scleroderma can be classified into three main types: diffuse, limited (also known as CREST syndrome), and morphea/linear. While diffuse and limited scleroderma are systemic diseases affecting the whole body, linear/morphea scleroderma primarily impacts the skin.
Diffuse Scleroderma
Diffuse scleroderma, or progressive systemic sclerosis, is the most severe form of the disease. It has a rapid onset and leads to widespread hardening of the skin. This type often causes significant damage to internal organs, especially the lungs and gastrointestinal tract, and poses a greater threat to life.
Limited Scleroderma/CREST Syndrome
Limited scleroderma, also known as CREST syndrome, is a milder form of the disease with a slower onset and progression. Skin hardening is usually restricted to the hands and face, and there is less severe involvement of internal organs, resulting in a better prognosis. Often, Raynaud's phenomenon occurs years before the onset of scleroderma, and skin thickening may be limited to the fingers, a condition known as sclerodactyly.
CREST syndrome is characterized by:
- Calcinosis
- Raynaud's syndrome
- Esophageal dysmotility
- Sclerodactyly
- Telangiectasia
This form is associated with antibodies against centromeres and typically does not affect the lungs or kidneys.
Morphea/Linear Scleroderma
- Morphea/linear scleroderma is characterized by localized patches of hardened skin, with minimal or no involvement of internal organs.
- Diagnosis of scleroderma is based on clinical evaluation and the presence of specific autoantibodies, particularly anti-centromere and anti-scl70/anti-topoisomerase antibodies. In some cases, a biopsy may be performed. Approximately 90% of individuals tested for scleroderma have a detectable anti-nuclear antibody. The anti-centromere antibody is more commonly found in the limited form of scleroderma (80-90%) compared to the diffuse form (10%). Conversely, the anti-scl70 antibody is more prevalent in the diffuse form (30-40%).
Deficiencies and Toxicities of Metals
1. Boron:
- Deficiency: No known biological function.
- Toxicity: Can lead to developmental defects, male sterility, and testicular atrophy.
2. Calcium:
- Deficiency: Results in reduced bone mass and osteoporosis.
- Toxicity: Can cause renal insufficiency (milk-alkali syndrome), nephrolithiasis, and impaired iron absorption.
3. Copper:
- Deficiency: Leads to anemia, growth retardation, defective keratinization and pigmentation of hair, hypothermia, degenerative changes in aortic elastin, osteopenia, and mental deterioration.
- Toxicity: Causes nausea, vomiting, diarrhea, hepatic failure, tremors, mental deterioration, hemolytic anemia, and renal dysfunction.
4. Chromium:
- Deficiency: Impairs glucose tolerance.
- Toxicity: Occupational exposure can lead to renal failure, dermatitis, and pulmonary cancer.
5. Fluoride:
- Deficiency: Results in dental caries.
- Toxicity: Can cause dental and skeletal fluorosis, and osteosclerosis.
6. Iodine:
- Deficiency: Leads to thyroid enlargement and cretinism.
- Toxicity: Can cause thyroid dysfunction and acne-like eruptions.
7. Iron:
- Deficiency: Causes muscle abnormalities, koilonychia, pica, anemia, reduced work performance, impaired cognitive development, premature labor, and increased perinatal and maternal mortality.
- Toxicity: Leads to gastrointestinal effects (nausea, vomiting, diarrhea, constipation), iron overload with organ damage, and acute systemic toxicity.
8. Manganese:
- Deficiency: Results in impaired growth and skeletal development, reproduction, lipid and carbohydrate metabolism, and an upper body rash.
- Toxicity: Causes neurotoxicity and Parkinson-like symptoms. Occupational exposure can lead to encephalitis-like syndrome, Parkinson-like syndrome, psychosis, and pneumoconiosis.
9. Molybdenum:
- Deficiency: Leads to severe neurologic abnormalities.
- Toxicity: Can cause reproductive and fetal abnormalities.
10. Selenium:
- Deficiency: Results in cardiomyopathy, heart failure, and striated muscle degeneration.
- Toxicity: Causes general symptoms like alopecia, nausea, vomiting, abnormal nails, emotional lability, peripheral neuropathy, lassitude, garlic odor to breath, and dermatitis. Occupational exposure can lead to lung and nasal carcinomas, liver necrosis, and pulmonary inflammation.
11. Phosphorus:
- Deficiency: Causes rickets (osteomalacia), proximal muscle weakness, rhabdomyolysis, paresthesia, ataxia, seizures, confusion, heart failure, hemolysis, and acidosis.
- Toxicity: Leads to hyperphosphatemia.
12. Zinc:
- Deficiency: Causes growth retardation, altered taste and smell, alopecia, dermatitis, diarrhea, immune dysfunction, failure to thrive, gonadal atrophy, and congenital malformations.
- Toxicity: Leads to reduced copper absorption, gastritis, sweating, fever, nausea, and vomiting. Occupational exposure can cause respiratory distress and pulmonary fibrosis.
FAQs on Chapter Notes: Miscellaneous Topics of Pathology
| 1. What is a pleomorphic adenoma and where is it commonly found? |  |
| 2. What are the key characteristics of Warthin's tumor? |  |
| 3. How does mucoepidermoid carcinoma differ from other salivary gland tumors? |  |
| 4. What is the significance of neuroblastoma in pediatric oncology? |  |
| 5. What are the common types of mediastinal tumors and their implications? |  |





