Chapter Notes: Chemical Changes and Reactions
Introduction
In this chapter, we will learn about chemical changes and reactions, which are important processes in chemistry. A chemical reaction happens when substances (called reactants) change to form new substances (called products). We will explore how these reactions occur, the types of reactions like combination and decomposition, and the conditions needed for them to happen, such as heat, light, or electricity. We will also study how to identify reactions through changes like color, gas formation, or precipitates. This chapter helps us understand the behavior of substances and how they transform in different situations.
Chemical Reaction
- A chemical reaction is a process where reactants (starting substances) change to form new products.
- During a reaction, the bonds in reactants break, and new bonds form to create products.
- A chemical bond is the force that holds atoms together in a molecule or compound.
- A chemical change or reaction happens when particles come close together (collide).
- Collisions happen by supplying energy, like heat, light, or electricity.
- In some cases, a catalyst is needed for a chemical change to occur.
Mixing (Alone) Is Not Enough: Just mixing two substances does not always cause a reaction.
- Example 1: Iodine and phosphorus react when brought into close contact.
- Example 2: Lead nitrate (white) and potassium iodide (yellow) react to make lead iodide (yellow) and potassium nitrate (white).
Pb(NO₃)₂ (aq) + 2KI (aq) → 2KNO₃ (aq) + PbI₂ (s). - In some cases, a chemical reaction happens when substances are mixed, such as an acid and a salt.
- Example 3: Oxalic acid crystals and sodium carbonate react in water.
H₂C₂O₄ (aq) + Na₂CO₃ (aq) → Na₂C₂O₄ (aq) + H₂O (l) + CO₂ (g). - Sodium chloride and silver nitrate also react in a solution to form a precipitate of silver chloride.
NaCl (aq) + AgNO₃ (aq) → AgCl (s) + NaNO₃ (aq).
Heat
- Some chemical reactions happen only when heat is provided.
- Example 1: Copper carbonate decomposes on heating into copper oxide and carbon dioxide.
CuCO₃ (s) → CuO (s) + CO₂ (g). - Example 2: Lead nitrate decomposes on heating, leaving brown gas nitrogen dioxide and colorless oxygen gas.
2Pb(NO₃)₂ (s) → 2PbO (s) + 4NO₂ (g) + O₂ (g).
Light
- Some chemical reactions take place by the action of light; these are called photochemical reactions.
- Examples include reactions in plants where they form glucose from carbon dioxide and water using sunlight.
6CO₂ (g) + 6H₂O (l) → C₆H₁₂O₆ (aq) + 6O₂ (g). - Another example: Hydrogen and chlorine react in the presence of sunlight.
H₂ (g) + Cl₂ (g) → 2HCl (g).
Electricity
- Certain chemical reactions, like the decomposition of certain compounds, occur only when electricity is passed through the substance.
Example: Electrolysis of acidulated water occurs only in the presence of electricity, to give hydrogen and oxygen.
Pressure
- Mercuric chloride and potassium iodide when rubbed in a mortar, give a scarlet- coloured substance called mercuric iodide.
- Nitrogen and hydrogen, when subjected to high pressure, produce ammonia (in the presence of iron as catalyst)
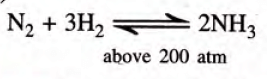
Catalyst
- Some reactions need a special substance to make them go faster or slower.
- This special substance is called a catalyst.
- The catalyst does not take part in the reaction, so it does not get used up.
- It only helps to change the speed of the reaction.
Example: Decomposition of Potassium Chlorate
- Potassium chlorate breaks down into potassium chloride and oxygen.
- This happens only at a very high temperature of 700°C.
- At this temperature, the oxygen is released very slowly.
- If we add manganese dioxide to potassium chlorate, it breaks down at a much lower temperature, 300°C.
- In this reaction, manganese dioxide acts as a catalyst because it lowers the temperature needed and speeds up the reaction.
- The equation for this reaction is: 2KClO₃ MnO₂ → 2KCl + 3O₂
(a) Positive Catalyst
- A positive catalyst makes a chemical reaction happen faster.
- It increases the speed of the reaction without being part of it.
- For example, manganese dioxide helps break down hydrogen peroxide faster.
Negative Catalyst
- A negative catalyst slows down a chemical reaction.
- It is used to reduce the speed of the reaction.
- Example 1: Phosphoric acid slows down the breaking down of hydrogen peroxide.
- Example 2: Alcohol slows down the oxidation of chloroform.
Characteristics of Chemical Reactions
Chemical reactions have certain characteristics that we can observe.
(i) Evolution of Gas
- In many chemical reactions, one of the products is a gas.
- Example: When zinc reacts with dilute sulfuric acid, hydrogen gas comes out with fizzing (effervescence).
- Equation: Zn (s) + H₂SO₄ (aq) → ZnSO₄ (aq) + H₂ (g).
(ii) Change of Color
- Some reactions are identified by a change in the color of the reactants.
- Example: When a few pieces of iron are dropped in a blue copper sulfate solution, the blue color fades, and eventually, the solution turns light green due to the formation of ferrous sulfate.
Fe (s) + CuSO₄ (aq) → FeSO₄ (aq) + Cu (s). - Color change: Blue (copper sulfate) to green (ferrous sulfate).
(iii) Formation of Precipitates
- Chemical reactions where insoluble solids are formed are called precipitation reactions.
- Example: When a solution of silver nitrate is added to sodium chloride, a white precipitate of silver chloride is formed.
AgNO₃ (aq) + NaCl (aq) → AgCl (s) + NaNO₃ (aq). - Example 2: When ferrous sulfate solution is added to sodium hydroxide, a dirty green precipitate of ferrous hydroxide is formed.
FeSO₄ (aq) + 2NaOH (aq) → Fe(OH)₂ (s) + Na₂SO₄ (aq).
(iv) Change of State
- In many chemical reactions, the state of reactants and products changes, such as from solid to gas or liquid.
- Example: Ammonia gas reacts with hydrogen chloride gas to produce solid ammonium chloride.
NH₃ (g) + HCl (g) → NH₄Cl (s).
Types of Chemical Change or Reaction
Chemical reactions can be classified into different types based on how reactants change into products.
1. Direct Combination (or Synthesis)
- In this reaction, two or more substances combine to form a single product (A + B → AB).
- Substances A and B take place to form a new molecule AB.
In Combination Reactions
- The reaction is also called synthesis.
- Carbon burns in oxygen to form a gaseous compound, carbon dioxide.
C (s) + O₂ (g) → CO₂ (g). - When magnesium is burnt, it combines directly with the oxygen of air to form magnesium oxide.
2Mg (s) + O₂ (g) → 2MgO (s).
2. Decomposition
- It is the breaking up of a compound into simpler compounds or elements (AB → A + B).
- Decomposition may occur in the presence of heat, light, or by the passage of an electric current.
- In a decomposition reaction, a compound breaks up into two or more elements or compounds.
- Example: The compound mercuric oxide, when heated, decomposes to form two elements: mercury and oxygen.
2HgO (s) → 2Hg (l) + O₂ (g). - The symbol (Δ) (delta) is used to signify that heat has caused the reaction.
- A Decomposition Reaction That Is Brought About by Electric Current Is Known as Electrolysis Decomposition
- When an electric current is passed through acidulated water, the latter decomposes into hydrogen and oxygen.
2H₂O (l) → 2H₂ (g) + O₂ (g). - A Compound Can Break Up to Form Two or More Compounds
- Example: The compound potassium nitrate, on heating, decomposes to produce a compound, potassium nitrite, an element, oxygen.
2KNO₃ (s) → 2KNO₂ (s) + O₂ (g).
Thermal Decomposition of Metal Compound
(i) Metal hydroxide
(ii) Metal carbonates

(iii) Metal bicarbonate (metal hydrogen carbonate)
Metal bicarbonates or metal hydrogen carbonates decomposes to give metal carbonate, water vapour and carbon dioxide.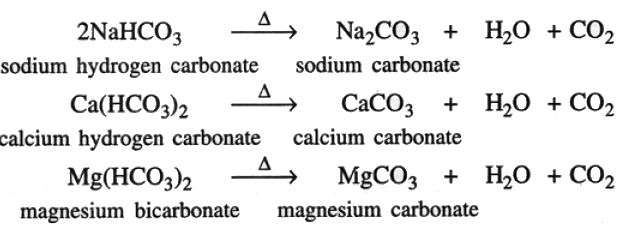 (iv) Metal nitrates
(iv) Metal nitrates
Digestion of Food in Our Body
- The starch present in the food we eat decomposes into glucose and sugar.
- Proteins undergo decomposition to form amino acids.
- Fats decompose into fatty acids.
- These products are finally oxidized by respiration into carbon dioxide and water.
- Starch → Glucose → CO₂ + H₂O.
3. Displacement
- It is a chemical change in which a more active element displaces a less active element from its salt solution.
- Example: When a solution of copper sulfate is kept in a glass beaker, and a strip of zinc is added, the blue color of the solution gradually fades, and reddish-brown copper settles down in the beaker.
Zn (s) + CuSO₄ (aq) → ZnSO₄ (aq) + Cu (s).
4. Double Decomposition
- In this type of reaction, two compounds react to form two new compounds by the mutual exchange of radicals (AB + CD → AD + CB).
- Precipitation Reaction: A chemical reaction in which two compounds in their aqueous state react to form an insoluble salt (a precipitate) as one of the products.
- Example: When a solution of silver nitrate is added to a test tube containing dilute hydrochloric acid, a white precipitate of silver chloride is formed.
AgNO₃ (aq) + HCl (aq) → AgCl (s) + HNO₃ (aq).
(a) Precipitation Reaction
- A chemical reaction in which two compounds in their aqueous state react to form an insoluble salt (a precipitate) as one of the products.
- Example: Barium chloride (aq) + sodium sulfate (aq) → barium sulfate (s) + sodium chloride (aq).
- Equation: BaCl₂ (aq) + Na₂SO₄ (aq) → BaSO₄ (s) + 2NaCl (aq).
(b) Neutralization Reaction
- A chemical reaction between an acid and a base that forms salt and water only is referred to as a reaction of neutralization.
- The reaction takes place because the hydrogen ion (H⁺) from the acid combines with the hydroxyl ion (OH⁻) from the base to form water.
- Equation: NaOH + HCl → NaCl + H₂O.
As a Chemical Process, Neutralization Has Many Uses
- When someone is stung by a bee, formic acid enters the skin and gives pain, which can be relieved by rubbing the spot with slaked lime or baking soda, both of which are bases.
- If our stomach glands secrete excess HCl, we may suffer from acidity, which can be relieved by taking magnesium hydroxide or sodium carbonate, both of which are bases.
- In agriculture, if the soil is too acidic, it is taken in dilute form to make up for it.
- Acid That Is Accidentally Spilled on Our Clothes or Body Can Be Neutralized with Ammonia Solution.
- If the soil is somewhat acidic and thus crops do not grow well, the soil is neutralized with slaked lime or calcium oxide.
- If the Soil Is Somewhat Acidic, and Thus Crops Do Not Grow Well, the Soil Is Neutralized with Slaked Lime.
- It is the process in which a reaction of strong acid or a strong base and weak acid and strong base occurs.
- This happens because a salt is formed due to a basic solution.
- A basic solution turns red litmus blue.
Energy, Change in Chemical Reactions
- Every chemical reaction involves a change in energy.
- The energy can be in the form of heat, sound, or electricity.
- The substances that have a fixed amount of stored energy are called potential energy.
- During a chemical reaction, this stored energy is either released or absorbed.
1. Exothermic Reaction
- An exothermic reaction is a chemical reaction where heat energy is given out to the surroundings.
- This causes an increase in the temperature of the surroundings.
Example:
- When carbon burns in oxygen, it forms carbon dioxide and releases heat.
C + O₂ → CO₂ + Heat - When water is added to quicklime, a lot of heat energy is produced along with alkaline calcium hydroxide (also called slaked lime).
CaO + H₂O → Ca(OH)₂ + Heat - When hydrogen is burnt in oxygen, water is formed, and heat is released.
2H₂ + O₂ → 2H₂O + Heat - Formation of ammonia: Nitrogen reacts with hydrogen in the presence of a catalyst (finely divided iron) at 450°C to 500°C, and above 200 atmospheres of pressure to form ammonia.
N₂ + 3H₂ ⇌ 2NH₃ + Heat
2. Endothermic Reaction
- An endothermic reaction is a chemical reaction where heat energy is absorbed from the surroundings.
- This causes a fall in the temperature of the surroundings.
- These reactions cannot happen without a constant supply of energy from an external source.
Examples:
- Formation of carbon disulphide: When carbon is heated with sulphur at a high temperature, liquid carbon disulphide is formed.
C + 2S → CS₂ - When nitrogen and oxygen are heated together to a temperature of about 3000°C, nitric oxide gas is formed. This is an endothermic reaction.
N₂ + O₂ + Heat (3000°C) → 2NO - Calcium carbonate decomposes into carbon dioxide and calcium oxide when it is heated to a temperature of about 1000°C. This is also an endothermic reaction.
CaCO₃ + Heat (1000°C) → CaO + CO₂
Photochemical Reaction
A photochemical reaction is a reaction that happens with the absorption of light energy.
Example: Photosynthesis: Plants use sunlight to combine carbon dioxide and water to form glucose and oxygen.
6CO₂ + 12H₂O + Chlorophyll + Sunlight → C₆H₁₂O₆ + 6H₂O + 6O₂
Electrochemical Reaction
- An electrochemical reaction is a reaction that happens with the absorption of electrical energy.
- Example: Acidulated water breaks into hydrogen and oxygen when an electric current passes through it.
2H₂O + Electric Current → 2H₂ + O₂
Points To Remember
- When energy is applied to matter, changes happen. These changes are of two types: physical and chemical.
- A physical change is when only the physical properties of a substance get changed, not its chemical composition. For example, breaking of glass.
- A physical change is a temporary change and is reversible in nature.
- In a physical change, only the physical properties, such as state, color, shape, etc., undergo a change.
- A chemical change is when the chemical composition and properties of reacting substances undergo a change. For example, burning of paper.
- A chemical change is a permanent change and is irreversible.
- Chemical changes occur by (i) mixing the substances in solution state, (ii) by heat, (iii) light, (iv) electricity, (v) pressure, (vi) in the presence of a catalyst.
- A chemical reaction is the process of breaking the chemical bonds of reactants and formation of new bonds to form new products.
- A chemical reaction is confirmed by (i) evolution of gas, (ii) change of color, (iii) formation of precipitate, (iv) change of state.
- In a combination reaction, two or more substances combine to form a new substance. For example, Fe + S → FeS.
- A decomposition reaction is when a substance breaks down into two or more substances. For example, 2HgO → 2Hg + O₂.
- In a displacement reaction, one part of a molecule is replaced by another. For example, CuSO₄ + Fe → FeSO₄ + Cu.
- A double displacement reaction is when two reacting molecules exchange their ions. For example, NaCl + AgNO₃ → AgCl + NaNO₃.
- A reaction in which one of the products is formed as an insoluble substance that is thrown out of the solution as solid (precipitate) is called a precipitation reaction. For example, Na₂SO₄ + BaCl₂ → BaSO₄ + 2NaCl.
- A reaction in which heat is liberated (or given out) is called an exothermic reaction.
- A reaction in which heat is absorbed is called an endothermic reaction.
FAQs on Chapter Notes: Chemical Changes and Reactions
| 1. What is a chemical reaction? |  |
| 2. What are the characteristics of chemical reactions? |  |
| 3. What are the different types of chemical reactions? |  |
| 4. How can we identify a chemical change? |  |
| 5. Why is the law of conservation of mass important in chemical reactions? |  |





