Chapter Notes: General Radiology
Introduction
Radiation is energy emitted from a source that travels through materials or space. It is classified into two main types:
- Ionizing Radiation
- Non-Ionizing Radiation
Types of Electromagnetic Radiation:
Ionizing:
- X-rays
- Gamma (γ) rays
- Cosmic rays
Non-Ionizing:
- Ultraviolet (UV) rays
- Visible light
- Infrared (IR) rays
- Microwaves
- Radiowaves
Types of Particulate Radiation:
- Alpha (α) particles
- Beta (β) particles
- Protons
- Neutrons
Diagnostic Modalities and Associated Radiations:
Ionizing Radiation:
- X-rays: Used in CT scans, radiography, DEXA, IVP/IVU, HSG, bronchography, ERCP, fluoroscopy
- Gamma (γ) rays: Used in PET, bone scans, radionuclide scans, SPECT
Non-Ionizing Radiation:
- MRI
- Ultrasound (USG)
- Thermography
- Doppler
- MRCP
X-Ray Tube
- Wilhelm Conrad Roentgen, a German physicist, discovered X-rays on November 8, 1895, and was awarded the first Nobel Prize for Physics in 1901.
- Roentgen is considered the Father of Radiology, and International Radiology Day is celebrated on November 8th.
Parts of X-Ray Tube:
- Glass Envelope: Provides a protected and vacuumed environment for the tube.
- Target Window: A thinned area in the lower part of the glass envelope that allows X-rays to exit the tube.
- Cathode: Negatively charged electrode consisting of a filament, supporting wires, and focusing cups.
- Filament: Heated by an electric current to approximately 2200°C, causing the metal to release free electrons despite the attractive pull of the positive ion lattice.
- Anode: Positively charged electrode, typically made of tungsten, where fast-moving electrons from the cathode strike the target, producing X-rays (1% of energy) and heat (99% of energy).
- X-rays are produced primarily through two processes:
- Photoelectric Effect: Interaction with the inner shell (K-shell) electron, producing characteristic radiations.
- Bremsstrahlung Reaction: Interaction with the nucleus, accounting for 80% of X-ray production in most tubes, except in mammography where the photoelectric effect predominates.
Two Types of Anode
- Stationary Anode: Used in dental radiography and portable/mobile radiography due to its simpler design.
- Rotating Anode: Used in most cases due to its better heat tolerance capacity.
Filter
- Filters block low-energy radiation to reduce the radiation dose to the patient without affecting image quality.
- Typically made of aluminum.
- The target window also acts as an inherent filter in the X-ray tube.
Collimator
- A beam-restricting device that limits scattered radiation and directs the X-ray beam.
Grid
- Made up of lead, consisting of parallel-arranged lead strips placed between the patient and the cassette.
- Blocks scatter radiation to improve image quality on the film.
- Slightly increases radiation dose to the patient as it blocks some useful radiation.
Cassette
- In conventional radiography, it is a screen-film system.
- Screen: Made of calcium tungstate, converts X-rays into light.
- Film: Made of photosensitive material, typically silver bromide.
- Single-coated film: Silver bromide present on one surface, most commonly used.
- Double-coated film: Used in screen-film systems and dental radiography.
Contrast in Radiography
- Contrast refers to the ability to distinguish an object from its background in an image.
- It is influenced by KVP (kilovolt peak) and MAS (milliampere-seconds).
- Film Focal Distance. This is the distance between the X-ray tube and the cassette. The standard distance for most radiography is 100 cm. However, for chest X-rays, a distance of 180 cm is used to reduce cardiac magnification.
Penetration in Radiography
- To achieve proper penetration in radiography, the KVP setting needs to be adjusted.
Contrast Adjustment in Radiography
- Both KVP and MAS are important for adjusting contrast in radiography.
- Increasing KVP results in decreased contrast, while increasing MAS leads to increased contrast.
- KVP is a significant factor affecting contrast, along with MAS.
KVP, Kontrast, Voltage, and Penetration in Radiography
- Kilo volt peak (KVP) is related to voltage in radiography.
- Milli ampere second (MAS) is calculated by multiplying current with exposure time and is directly proportional to the energy of the beam.
- KVP does not affect energy but is directly proportional to penetration.
- MAS is inversely proportional to contrast and determines the number of photons in a specified area, influencing background blackening.
- MAS is directly proportional to contrast.
Radiation Units
Exposure Dose
- Definition: Measures the amount of X-rays or gamma rays that ionize air.
- Conventional Unit: Roentgen (R), defined as the radiation liberating a charge of 2.58 × 10⁻⁴ Coulombs per kilogram of air under standard conditions.
- SI Unit: Coulombs per kilogram (C/kg), where 1 Roentgen equals 2.58 × 10⁻⁴ C/kg.
Absorbed Dose
- Definition: Quantifies the energy deposited by radiation in a material.
- Conventional Unit: Rad, equivalent to 100 ergs of energy absorbed per gram of material.
- SI Unit: Gray (Gy), defined as 1 joule of energy absorbed per kilogram of material.
- Conversion: 1 Gray = 100 rads.
Equivalent Dose
- Definition: Reflects the biological impact of different radiation types.
- Formula: HT = WR × D, where HT is the equivalent dose, WR is the radiation weighting factor (quality factor), and D is the absorbed dose (in rads or Gray).
- Conventional Unit: Rem (now largely replaced).
- SI Unit: Sievert (Sv), where 1 Sievert = 100 rems.
Effective Dose
- Definition: Accounts for the varying sensitivity of tissues/organs to radiation's stochastic effects, compared to uniform whole-body exposure.
- Formula: HE = WT × HT, where HE is the effective dose, HT is the equivalent dose, and WT is the tissue weighting factor (based on tissue susceptibility).
- Unit: Expressed in Sieverts (Sv) or millisieverts (mSv).
- Calculation: Equivalent dose × Tissue weighting factor = Effective dose.
Harmful Effects of Radiation
- Ionization of molecules leads to free radical generation, causing DNA damage.
- Most common radiation side effect: Skin erythema.
Deterministic Effects:
- Directly proportional to dose, with a dose threshold.
- Severity is dose-related.
- Examples: Skin effects, epilation, lens opacities.
Stochastic Effects:
- Not directly proportional to dose, random events without a threshold.
- Probability increases with dose, follows an "all or none" phenomenon.
- Severity may not be dose-related.
- Examples: Genetic side effects, teratogenicity, mutations.
Radiation Protection for Patient
- 10-Day Rule: In females of reproductive age, modalities using ionizing radiation should be performed within the first 10 days of the menstrual cycle to avoid radiation exposure to an undiagnosed pregnancy, as ovulation does not occur during this period.
- Radiation protection aims to eliminate deterministic side effects and reduce the probability of stochastic side effects.
Pregnant Occupational Worker
- Should not be exposed to more than 2 mSv of radiation applied to the surface of the lower abdomen during the declared term of pregnancy, equivalent to 1 mSv to the fetus.
- Cerebrovascular Syndrome: At doses of 20-100 Gy to the total body, death occurs within 24-48 hours due to neurologic and cardiovascular failure, preventing failure of other systems.
- Gastrointestinal Syndrome: At doses of 5-12 Gy, death occurs in days due to nausea, vomiting, prolonged diarrhea, dehydration, sepsis, and death.
ALARA Approach
Stands for "As Low As Reasonably Achievable" radiation dose for patients.
- Radiation exposure limits:
- ICRP (International Commission on Radiological Protection): 20 mSv/year over 5 years for occupational workers, 1 mSv/year over 5 years for the general public.
- NCRP (National Council on Radiation Protection): Cumulative dose = Age in years × 10 mSv for occupational workers, 5 mSv for 5-year period for the general public.
- AERB (Atomic Energy Regulatory Board): 100 mSv for 5-year period for occupational workers, 1 mSv/year for 5 years for the general public.
- Radiation Worker Protection:
- Aprons: Made of lead with a thickness of 0.5 mm (minimum lead equivalence of 0.25 mm per AERB). Increasing thickness increases weight.
- Lead-free Aprons: Made of tin, antimony, and bismuth.
- TLD (Thermoluminescent Dosimeter): A badge worn by radiation workers to monitor radiation exposure, checked every 3 months.
- Radiation exposure in CT scans varies by body part: CT head = 2.3 mSv, CT abdomen = 10 mSv.
- A CT chest has radiation exposure equivalent to approximately 400 chest radiographs.
Digital Radiography
- Involves digitizing conventional film.
- Computed Radiography (CR): Uses a phosphor plate to capture a latent image, which is processed to produce a digital image.
- Direct Radiography (DR): Uses a flat panel detector to convert X-rays directly into an electrical signal for digital imaging.
Computed Radiography
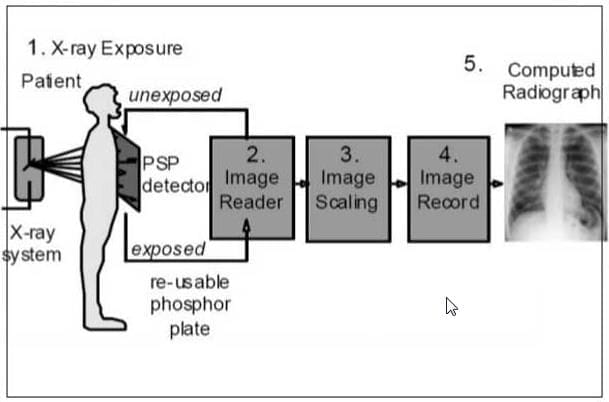
Process:
- Conventional X-ray exposes a phosphor plate (PSP).
- Latent image formed on the plate.
- Laser beam scans the plate, causing emission of light.
- Ultra-sensitive photomultiplier tube (PMT) converts light into an electronic signal.
- Signal is digitized for display on a CRT or hard copy.
Steps:
- Load cassette.
- Position patient and tube.
- Perform exposure.
- Transport and process cassette.
- Assess image quality to end examination.
Advantages:
- Allows manipulation and positioning of image receptor for cross-table projections, useful in trauma cases.
Disadvantages:
- Slower process with more steps.
- Potential for repetitive motion injuries due to long-term cassette handling.
Direct Radiography
Process:
- X-rays pass through the patient to a flat panel detector.
- Detector converts X-rays into electrical energy.
- Electrical energy is processed into a digital image.
Mammography
- Uses lower kVp for higher image contrast and higher mA for shorter exposure times compared to chest and abdominal examinations.
- Technical Features:
- Small focal spot size (0.2-0.5 mm).
- Beryllium window.
- Target-filter combinations: Molybdenum (preferred), Rhodium, Tungsten.
- Typically performed after age 40 due to dense glandular parenchyma in younger breasts, which makes lesions harder to detect and increases radiation sensitivity.
- Ultrasound (USG) is used for palpable lesions to differentiate solid from cystic lesions and to rule out breast abscess.
- Two Views:
- Mediolateral: More important due to greater coverage area.
- Craniocaudal: Provides additional perspective.
- Mammography has 10-40 times more radiation exposure than a chest X-ray (CXR).
- BIRADS Classification for Mammography Lesions:
- Grade 0: Incomplete evaluation, requires completion.
- Grade 1: Normal, continue routine screening (CRS).
- Grade 2: Benign calcified fibro, continue routine screening.
- Grade 3: Probably benign (<2% malignancy risk), short follow-up.
- Grade 4: Suspicious, indeterminate, requires further evaluation.
- Grade 5: Highly suspicious (>15% malignancy risk), biopsy recommended.
- Grade 6: Biopsy-proven malignancy, assess for contralateral breast and multicentric disease in the same breast.
Ultrasound
- Advantages:
- Real-time imaging.
- No radiation exposure.
- Portable and widely available.
- Cost-effective.
- Principle: Works on the piezoelectric effect, where electricity passed through a crystal (commonly Lead Zirconate Titanate, PZT) generates sound waves.
- Frequency Range: Medical ultrasound uses frequencies between 2-20 MHz.
- Transducer Operation: A handheld transducer sends ultrasound waves into the body and receives reflected waves, which are processed to generate images on a monitor.
- Real-time B-scans: Allow investigation of moving body structures.
- Note: Posterior structures are better visualized on transesophageal echocardiography; 5 layers of the gastrointestinal tract are seen on endoscopic ultrasound.
- Clinical Transducer Frequencies:
- 3.5-5 MHz for thick, deep body parts (e.g., abdomen, obstetrics).
- 7.5-10 MHz for small body parts (e.g., orbit, thyroid, breast).
- 7.5-20 MHz for intracavitary transducers (e.g., endovaginal for pelvis, endorectal for prostate, transesophageal for heart, intravascular for blood vessels, endoscopic for pancreatic lesions, esophageal/rectal cancer staging).
- Methods of Display:
- A-Mode (Amplitude): Plots amplitude of returning signals against depth, used for orbital biometry.
- B-Mode (Brightness): Most common mode, assigns grey scale values to signal amplitude to form an image of the scan plane.
- M-Mode (Motion): Detects rhythmic motion in the scan plane without amplitude considerations, used for valvular morphology and motion.
- Nomenclature of Ultrasound Images:
- Echogenic/Hyperechoic: Regions reflecting a lot of sound, appear bright/white (e.g., fat, calculus, bones, stones).
- Hypoechoic: Regions reflecting fewer sound waves, appear dark/black.
- Isoechoic: Regions with similar echo patterns to normal viscera or soft tissue.
- Anechoic: Fluid appears black, reflecting no sound waves.
- Posterior Acoustic Enhancement: Increased brightness beyond objects transmitting many sound waves (e.g., cysts).
- Posterior Acoustic Shadowing: Decreased brightness beyond objects reflecting a lot of sound (e.g., stones).
- Ultrasound as Investigation of Choice for:
- Hydrocephalus in infants.
- Retinal detachment.
- Minimal pleural effusion.
- Minimal pericardial effusion.
- CHPS (Congenital Hypertrophic Pyloric Stenosis).
- Gallstones.
- Acute cholecystitis (HIDA scan is theoretically best).
- Screening for rotator cuff injuries (initial investigation).
- Renal colic in pregnancy.
- Minimal ascites.
- Pararenal fluid collection post-renal transplant.
- Obstetrics indications.
- Scrotal pathologies.
- Varicose veins.
- Developmental dysplasia of the hip (DDH).
Advance Applications of USG
Doppler
Used for assessing moving structures, primarily blood flow, with applications in:
- Presence/absence of flow.
- Direction of flow (toward probe = red, away from probe = blue, turbulent flow = green due to mixing).
- Velocity of flow.
- Pattern of flow.
- Velocity assessment is critical for detecting stenosis, where velocity increases at the point of stenosis.
- Note: The vessel-transducer angle should be less than 60 degrees for accurate velocity calculation.
Flow Patterns:
- Peripheral Arteries: High resistance due to peripheral vascular resistance, typically triphasic pattern.
- Arterial Vasoconstriction: Increased resistance, flow becomes biphasic.
- Peripheral Vasodilation: Decreased resistance, flow becomes monophasic with pulsatility.
- Arterial Obstruction: Significant pressure loss, flow distal to obstruction becomes monophasic (tardus parvus).
- Visceral Arteries (hepatic, renal, umbilical): Low resistance, monophasic pattern with pulsation.
- Intrauterine Growth Restriction (IUGR): Increased placental resistance leads to decreased diastolic flow, increasing the systolic/diastolic (S/D) ratio (>3.0 in IUGR, normal <2.5).
- Absent Diastolic Flow: Occurs when diastolic force equals placental resistance, a sign of severe IUGR.
- Reversal of Diastolic Flow: Indicates impending fetal death.
- Veins: Continuous flow without pulsations, monophasic pattern.
Elastography
- A method that applies a distorting force (stress) to tissue, moving it by a few millimeters, and creates an image based on the tissue's response (strain).
- Ultrasound elastography uses the transducer to apply stress manually, detecting strain by tracking speckle pattern changes.
- More sensitive than clinical palpation, especially for deeper structures.
- Applications:
- Assessment of liver fibrosis degree.
- Differentiation of benign from malignant lesions in liver, breasts, and prostate.
CT Scan
- Developed by Sir Godfrey Hounsfield, the first clinical CT machine was installed in the early 1970s at Atkinson Morley Hospital, London.
- 1980s: Slip-ring technology enabled continuous X-ray unit rotation, reducing acquisition time to 1 second and allowing helical data acquisition.
- Late 1990s: Multidetector CT (MDCT) introduced multiple thin detector rows, enabling simultaneous acquisition of multiple sections, further reducing acquisition time and enabling thin sections.
- 2000s: Cardiac CT introduced with ECG-synchronization to freeze cardiac motion, with detector rows increasing to 64-320 and rapid tube rotation (<0.3 s).
- Basic Principle: Linear attenuation of X-rays.
- CT Number/Hounsfield Unit (HU):Numerical value assigned to a tissue based on its X-ray attenuation:
- Air: -1000 HU
- Fat: -50 to -100 HU
- Water: 0 HU
- Muscle: 10-40 HU
- Blood: ~60 HU
- Contrast: 130 HU
- Bone: >400 HU
- Slice Thickness: Thickness of tissue scanned in one rotation.
- Pitch: Ratio of table movement per rotation to beam collimation or slice thickness.
- Scan Length: Table movement during the entire scan, calculated as Pitch × beam collimation × (total scan duration / time per rotation).
- Pitch Effects:
- Increased pitch: Decreases scan duration and radiation dose but reduces scan quality.
- Decreased pitch: Increases scan duration and radiation dose but improves scan quality.
- Multislice/Multidetector CT Advantages:
- Faster acquisition.
- Coverage of larger areas.
- Reduced movement artifacts.
- Isotropic multi-planar reformats.
- Improved vascular and cardiac imaging.
- Faster patient throughput.
Dual Source CT
Uses two X-ray sources operating at different potentials (kV), allowing differentiation of body tissues and fluids by comparing image differences.
Applications:
- Pulmonary nodule characterization: Limited data.
- Fatty liver quantification for liver donors: Useful, more validation needed.
- Characterization of liver lesions: Useful results from initial studies.
- Characterization of adrenal mass: Further data needed.
- Coronary in-stent stenosis: Few data available.
- Bone subtraction for CTA post-processing: Useful.
- Kidney-stone differentiation: More data needed.
- Iron deposition in liver: MRI is superior.
- Bone densitometry: Better tests available.
- Advantages:
- Ability to scan at any heart rate without beta-blockers.
- Better temporal resolution (83 ms).
- Suitable for trauma, oncology, and obese patients.
High Resolution CT Scan (HRCT)
A specialized CT technique for specific diseases, including:
- Interstitial lung disease.
- Bronchiectasis.
- Miliary tuberculosis.
- Temporal bone evaluation.
- CSF leaks.
Principle:
- Thin slice thickness or collimation.
- Narrow field of view.
- Bone algorithm for image reconstruction.
Radiation Dose in a CT Scan
CTDI (CT Dose Index): A standardized measure of radiation dose output, representing the average absorbed dose along the z-axis from contiguous irradiations in one axial CT scan.
- CTDI estimates the average dose within the central region of a scan volume for multiple contiguous scans (Multiple Scan Average Dose, MSAD).
Dose Length Product (DLP): DLP (mGy·cm) = CTDIvol (mGy) × scan length (cm).
- DLP reflects the total energy absorbed and potential biological effect of the scan, proportional to the scan volume's z-extent.
MRI
- Magnetic Resonance Imaging (MRI) is a non-invasive method for mapping structure and function within the body using the gyromagnetic property of protons, without ionizing radiation.
- Principle: Nuclear magnetic resonance (discovered by Purcell and Bloch), with MRI invented by Lauterbur and Mansfield, and functional MRI by Ogawa and Rosen.
- Mechanism: Nuclei with odd numbers of protons/neutrons (e.g., hydrogen) absorb and emit energy at a resonant frequency in a magnetic field.
- Parts of MRI:
- Maxwell Coil: Gradient coil creating magnetic field gradients along the main magnetic field.
- Radio Frequency Coils: Generate a stronger magnetic field in the scan field.
- Faraday Cage/Shield/Hoffman Box: Blocks external static electric fields, made of copper.
- Superconductors: Electromagnets determining magnet strength, measured in Tesla.
- Image Acquisition:
- In the absence of a magnetic field, protons move randomly.
- Under a magnetic field, protons align.
- Radiofrequency pulses cause realignment, and when turned off, protons return to their original position, releasing energy as an electrical voltage signal.
- Spin-Lattice Relaxation (T1): Protons release energy to the surrounding lattice.
- Spin-Spin Relaxation (T2): Protons release energy to surrounding spins.
- The electrical signal is digitized and analyzed to produce MR images.
Contraindications of MRI
MRI was once thought to mean "metal results in injury," but many metallic devices, especially those made of titanium, are now MRI-compatible.
Contraindicated Devices/Conditions:
- Cardiac pacemakers (some are compatible at 1.5 Tesla, check compatibility).
- Cochlear implants.
- Intraocular metallic foreign body.
- Electronically, magnetically, or mechanically activated implants (e.g., defibrillators).
- Aneurysmal clips.
- Prosthetic heart valves.
- Insulin pumps and nerve stimulators.
- Stapedial implants.
- Claustrophobia.
- First trimester of pregnancy (safety not established).
Safe for MRI:
- Orthopedic implants (usually titanium; stainless steel implants may cause heating, requiring shorter scans and lower magnet strength).
- Pregnancy after the first trimester.
- Cholecystectomy clips, sternal sutures, IUCDs, non-metallic foreign bodies, breast implants.
- Pediatric patients (preferred due to lack of ionizing radiation, often under sedation).
- Coronary stents (drug-eluting stents safe after 3 months).
T1-Weighted MRI (Bright): Fat, hemorrhage, proteinaceous substances, melanin, paramagnetic agents (gadolinium). Dark on T1 and T2: Air, flowing blood (on SE/FSE images), cortical bone, ligaments, tendons, dense fibrous tissues.
Special Sequences of MRI
FLAIR (Fluid Attenuated Inversion Recovery):
- T2-weighted sequence suppressing water to enhance pathology visibility.
- Used in brain imaging where water is a confounding factor.
- Uses:
- Better visualization of brain pathologies (most appear white).
- Differentiates arachnoid cysts (CSF-filled, black on FLAIR) from epidermoid cysts.
STIR (Short Tau Inversion Recovery):
- T2-weighted sequence suppressing fat, making pathology white and fat black.
- Used in musculoskeletal and body imaging where fat reduces contrast.
- Uses:
- Identifies bone marrow edema in stress fractures, avascular necrosis, Perthes disease.
- Enhances disease visualization in areas with high fat content.
Contrast-Enhanced MRI (CEMRI):
- Uses gadolinium-containing compounds to reduce T1 and T2 relaxation times, increasing signal on T1-weighted images and decreasing signal on T2-weighted images.
- Images are acquired as T1-weighted since human eyes better appreciate white.
MRCP (Magnetic Resonance Cholangiopancreatography):
- Heavily T2-weighted sequence showing fluid-filled structures (biliary and pancreatic ducts) as high signal intensity.
- Non-invasive, no external contrast required, preferred over diagnostic ERCP for biliary and pancreatic duct pathologies.
MR Angiography:
- Visualizes vessels without external contrast using the Time of Flight (TOF) technique.
- Preferred for assessing renal artery stenosis, intracerebral aneurysms, and carotid artery stenosis.
Diffusion-Weighted MRI (DWI):
- Assesses random (Brownian) motion of water molecules.
- Restricted diffusion appears white on DWI.
- Common Causes of Restricted Diffusion:
- Acute ischemic infarct (most common).
- Abscess.
- High cellular tumors.
- Epidermoid cyst.
Gradient Echo Images:
- Used to detect calcification and hemorrhage on MRI.
Spectroscopy:
Assesses metabolites in tissues for early disease diagnosis, as metabolic changes precede structural changes.
Common Metabolites:
- N-Acetyl Aspartate (NAA): Neuronal marker, assesses neural integrity, decreased in most brain disorders except Canavan's disease.
- Choline: Marker of cell membrane turnover, increased in malignancies.
- Creatine: Marker of energy stores, stable reference marker.
- Lipid Peak: Useful to differentiate tuberculoma (rich in caseous necrosis) from neurocysticercosis.
MRI is Usually a Preferred Investigation for
- All brain tumors (contrast-enhanced MRI).
- Extent of parotid tumors (due to neural involvement).
- Ventricular function (most accurate).
- Posterior mediastinal mass (neurogenic origin).
- Bony metastasis in the spine (bone scan preferred elsewhere).
- Chronic subarachnoid hemorrhage.
- Traumatic paraplegia (due to neural involvement).
- Extent of Pott's spine (biopsy best for diagnosis).
- Stress fracture, Perthes disease, avascular necrosis, early osteomyelitis (bone marrow edema).
- Pregnancy after the first trimester (for non-obstetric abdominal pathologies; USG preferred for antenatal scanning).
- Spinal cord pathologies.
- Gynecological malignancies.
- Uterine anomalies.
Comparison of MRI, CT Scan, and USG
Ionizing Radiation:
- MRI: None.
- CT Scan: Moderate to high.
- USG: None.
Principle:
- MRI: Magnetic moment of protons (gyromagnetism).
- CT Scan: Linear attenuation of X-rays.
- USG: Piezoelectric effect.
Time for Scanning:
- MRI: 10 minutes to 1 hour.
- CT Scan: Less than a minute.
- USG: Variable.
Bony Details:
- MRI: Cortical bone not well seen, bone marrow very well seen.
- CT Scan: Cortical bone very well seen.
- USG: Not well seen; structures posterior to bone not visible due to acoustic shadowing.
Soft Tissue Structure:
- MRI: Very well seen and differentiated.
- CT Scan: Seen but not well characterized.
- USG: Very well seen.
Multiplanar Capability:
- MRI: Images acquired in any plane.
- CT Scan: Acquired in axial planes, reconstructible in any plane on MDCT.
- USG: Yes, depending on probe position.
Real-Time Imaging:
- MRI: Mostly not.
- CT Scan: No.
- USG: Yes.
Portability:
- MRI: Usually not.
- CT Scan: Usually not.
- USG: Yes.
Cost:
- MRI: Very costly.
- CT Scan: Costly.
- USG: Cheap.
Uses:
- MRI: Neural tissues, bone marrow, cartilage, ligaments, tendons, soft tissues.
- CT Scan: Cortical bone, acute hematoma in brain, calculi, calcifications, air, guiding lung biopsies.
- USG: Fluid-containing structures, guiding biopsies.
Best MRI Sequence to Differentiate Epidermoid Cyst/Arachnoid Cyst: DWI > FLAIR.
Note: Gadolinium does not cross the blood-brain barrier (BBB).
Risk of Nephrogenic Systemic Fibrosis (NSF): Seen only in patients with chronic kidney disease due to gadolinium.
Contrast Agents in Radiology
- Gadolinium-containing compounds are more nephrotoxic than iodinated compounds in equimolar concentrations, but less gadolinium is used, reducing nephrotoxic side effects.
MRI Contrast Agents
Gadolinium Chelates:
- Most commonly used MRI contrast agents, safe due to lack of iodine, with minimal risk of anaphylaxis or allergic reactions.
- Paramagnetic, reducing T1 and T2 relaxation times.
- Common side effects: Mild headache, metallic taste.
- Contraindicated in pregnant patients due to potential crossing of the blood-placental barrier, with unknown fetal effects.
- Nephrogenic Systemic Fibrosis (NSF): A scleroderma-like dermatopathy in patients with compromised renal function.
Risk Factors for NSF:
- Increased or repeated doses of gadolinium.
- Use of linear gadolinium chelates, particularly gadodiamide (Omniscan).
European Guidelines for NSF Prevention:
- High-Risk Patients: CKD 4 and 5 (GFR <30 mL/min), including dialysis patients and those awaiting renal transplant.
- Low-Risk Patients: CKD 3 (GFR 30-59 mL/min), children <1 year old due to immature renal function.
- Not at Risk: Normal renal function.
- Highest-Risk Agents: Gadodiamide, Gadopentetate dimeglumine, Gadoversetamide (contraindicated in CKD 4/5, use with caution in CKD 3 and children <1 year, require eGFR measurement).
- Intermediate-Risk Agents: Gadobenate dimeglumine, Gadofosveset trisodium, Gadoxetate disodium (eGFR measurement not mandatory).
- Low-Risk Agents: Gadobutrol, Gadoterate meglumine, Gadoteridol (eGFR measurement not mandatory).
USG Contrast Agents
- Gas-filled microbubbles (0.1-10 μm diameter) with a phospholipidic, albumin, or polymer shell containing inert gas.
- Mechanism of Action: Increases backscattering echo intensity due to acoustic impedance differences between blood and gas, enhancing color, power, and spectral Doppler signals or grayscale image intensity.
- Examples: Albumex, Levovist, Echogen.
- Uses:
- In the liver: Characterization of focal liver lesions, detection of portal vein/collateral flow in cirrhosis, evaluation of TIPSS stent patency/stenosis, visualization of small intrahepatic collaterals in Budd-Chiari syndrome.
- Improved vessel visualization in Doppler.
- Note: Air embolism requires at least 100 mL of air, while USG contrast uses only 0.5 mL.
Iodinated Water Soluble Contrast Media
Types:
- Ionic Monomers: Iodine particle ratio 3:2, high osmolality (HOCM), e.g., Diatrizoate (Urografin, Gastrografin), Iothalamate. Preferred for non-vascular use.
- Ionic Dimers: Iodine particle ratio 6:2, low osmolality (LOCM), e.g., Ioxaglate.
- Non-Ionic Monomers: Iodine particle ratio 3:1, low osmolality (LOCM), e.g., Iohexol, Iomeron, Iopamidol, Ipromide, Ioversol, Xenetix. Most preferred for vascular administration.
- Non-Ionic Dimers: Iodine particle ratio 6:1, iso-osmolality (IOCM), e.g., Iodixanol (Visipaque). Best for patients with associated renal disease.
Contrast Reactions
- Idiosyncratic Anaphylactoid Reactions: Treated with adrenaline.
- Non-Idiosyncratic Reactions: Other adverse effects not related to allergic responses.
Contrast-Induced Nephropathy
- Definition: Sudden deterioration in renal function following recent intravascular administration of iodinated contrast medium without another nephrotoxic event.
- Diagnosis: Commonly defined as an absolute increase in serum creatinine of 0.5 mg/dL or a 25-50% increase from baseline.
- Pathogenesis: Likely due to decreased renal perfusion from renal vasoconstriction caused by high osmolality of contrast media.
- Symptoms: Usually asymptomatic, with creatinine peaking in 3-5 days (5-7 days in severe oliguric patients).
Risk Factors:
- Pre-existing renal impairment (serum creatinine >1.3 mg/dL, GFR <60 mL/min).
- Dehydration.
- Congestive heart failure (CHF).
- Use of nephrotoxic drugs (NSAIDs, aminoglycosides).
- Hypersensitivity diseases (e.g., multiple myeloma).
- Hypertension.
- Hyperuricemia (e.g., active gout).
- Proteinuria (>0.5 g/dL).
- Diabetes mellitus, age >70 years.
Prevention in High-Risk Patients:
- Avoidance of Iodinated Contrast: Substitute IOCM/LOCM for HOCM to reduce osmotic load.
- Hydration: Most effective prevention, using 0.9% saline at 100 mL/hr, 6-12 hours before and 4-12 hours after contrast administration. Oral hydration is less effective.
- Sodium Bicarbonate: Found useful in some studies for CIN prevention.
- N-Acetylcysteine: Controversial; may lower serum creatinine without preventing renal injury, not a substitute for screening and hydration.
- Diuretics (Mannitol, Furosemide): No benefit for CIN risk reduction.
- Other Agents (Theophylline, Endothelin-1, Fenoldopam): No convincing evidence for CIN prevention.
- Hemodialysis/Hemofiltration: Hemodialysis not effective; hemofiltration has some benefit.
Patient on Metformin
Risk: Metformin + chronic renal insufficiency + IV contrast = lactic acidosis.
Management Categories:
- Category 1: Normal renal function, no comorbidities, no need to discontinue metformin.
- Category 2: Normal renal function with comorbidities, suspend metformin for 48 hours; restart if patient is stable, has no intercurrent renal risk factors, and had normal baseline renal function.
- Category 3: Renal dysfunction, suspend metformin for 48 hours, restart only if repeat kidney function tests (KFT) are normal.
Barium Studies
Barium sulfate is used for gastrointestinal tract (GIT) evaluation due to its inert nature and lack of absorption, with mucosal coating aiding in assessing mucosal details.
Types:
- Barium Swallow: For dysphagia to rule out stricture or mass, largely replaced by endoscopy; now used for motility disorders.
- Barium Meal: For abdominal pain to rule out gastric/duodenal ulcers, replaced by endoscopy.
- Small Bowel Follow-Through: For diarrhea or constipation to rule out Crohn's disease or other small bowel pathologies, replaced by barium enteroclysis (small bowel enema) or CT enterography.
- Barium Enema: For rectal bleeding to rule out polyps or masses, sometimes replaced by CT virtual colonoscopy.
Contraindications:
- Small bowel obstruction (ionic contrasts preferred).
- Intestinal perforation (ionic contrasts preferred).
- Tracheoesophageal fistula (non-ionic contrasts preferred due to risk of pulmonary edema with ionic contrasts).
Myelography:
- Involves injecting contrast into the subarachnoid space via lumbar puncture.
- Non-ionic contrast agents are preferred to reduce the risk of arachnoiditis.
- Most Common Side Effect: Headache.
- Most Severe Side Effect: Arachnoiditis.
FAQs on Chapter Notes: General Radiology
| 1. What are the two types of anodes used in radiography, and what are their characteristics? |  |
| 2. What is the 10-Day Rule in radiation protection for patients, particularly in mammography? |  |
| 3. What are the harmful effects of radiation that healthcare professionals should be aware of? |  |
| 4. What is elastography, and how is it used in advanced applications of ultrasound (USG)? |  |
| 5. How does the screen film system in conventional radiography work, and what are its advantages? |  |





