Cheat Sheet: Acids, Bases and Salts
1. Introduction to Acids and Bases
In everyday life, many substances show acidic or basic properties. For example:
- Acids usually taste sour (e.g., lemon, vinegar).
- Bases usually taste bitter and feel soapy (e.g., soap solution).
These substances can be identified using indicators, which change colour depending on whether a substance is acidic or basic.
Common indicators
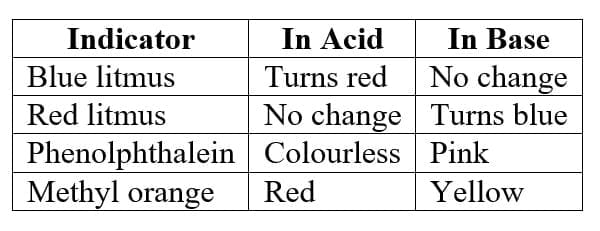
Litmus is a natural indicator extracted from lichens, while indicators like phenolphthalein and methyl orange are synthetic indicators.
2. Olfactory Indicators
Some substances change their smell in acidic or basic solutions. These are called olfactory indicators.
Examples include:
- Onion
- Vanilla
- Clove oil
Their smell disappears in a basic solution but remains in an acidic solution.
3. Chemical Properties of Acids
(a) Reaction of Acids with Metals
When acids react with metals, hydrogen gas is produced and a salt is formed.
General reaction:
Acid + Metal → Salt + Hydrogen gas
Example:
Zn + H₂SO₄ → ZnSO₄ + H₂
Hydrogen gas can be tested by bringing a burning matchstick, which produces a pop sound.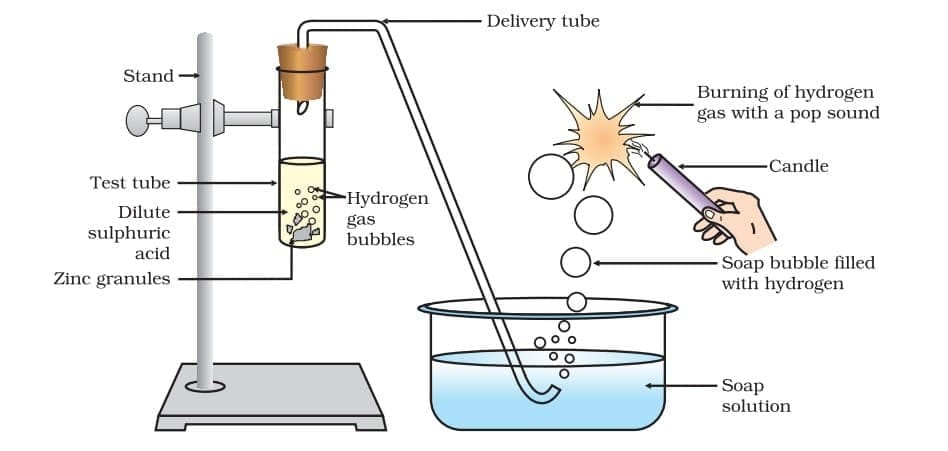 Reaction of zinc granules with dilute sulphuric acid and testing hydrogen gas by burning
Reaction of zinc granules with dilute sulphuric acid and testing hydrogen gas by burning
(b) Reaction of Acids with Metal Carbonates
Acids react with metal carbonates to produce:
- Salt
- Carbon dioxide gas
- Water
General reaction:
Metal carbonate + Acid → Salt + CO₂ + Water
Example:
Na₂CO₃ + 2HCl → 2NaCl + H₂O + CO₂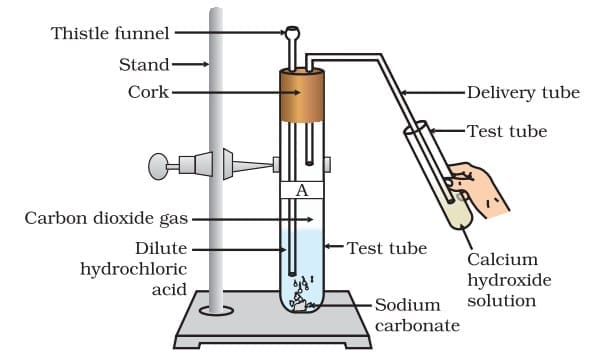 Passing carbon dioxide gas through calcium hydroxide solution
Passing carbon dioxide gas through calcium hydroxide solution
The CO₂ gas turns limewater milky due to formation of calcium carbonate.
(c) Reaction of Acids with Metal Hydrogencarbonates
Metal hydrogencarbonates also react with acids to produce salt, carbon dioxide, and water.
Example:
NaHCO₃ + HCl → NaCl + H₂O + CO₂
4. Neutralisation Reaction
When an acid reacts with a base, they cancel each other's effect and form salt and water.
General reaction: Acid + Base → Salt + Water
Example: NaOH + HCl → NaCl + H₂O
This reaction is called a neutralisation reaction.
5. Reaction of Bases
Reaction of Bases with Metals
Some bases react with metals to produce hydrogen gas and a salt.
Example:
2NaOH + Zn → Na₂ZnO₂ + H₂
Reaction of Metal Oxides with Acids
Metal oxides react with acids to produce salt and water.
Example:
CuO + 2HCl → CuCl₂ + H₂O
Because they behave like bases, metal oxides are called basic oxides.
Reaction of Non-Metal Oxides with Bases
Non-metal oxides react with bases to form salt and water, showing that they are acidic in nature.
Example:
CO₂ + Ca(OH)₂ → CaCO₃ + H₂O
6. Acids and Bases in Aqueous Solutions
Acids and bases show their properties only in aqueous solutions (water).
Acids in water
Acids produce hydrogen ions (H⁺) or hydronium ions (H₃O⁺) in water.
Example: HCl + H₂O → H₃O⁺ + Cl⁻
Bases in water
Bases produce hydroxide ions (OH⁻) in water.
Example: NaOH → Na⁺ + OH⁻
Bases that dissolve in water are called alkalis.
7. Dilution of Acids and Bases
Dissolving acids or bases in water is an exothermic reaction (heat is released).
Important rule:
Always add acid to water, never water to acid.
This prevents dangerous splashing caused by sudden heating.
8. Strength of Acids and Bases (pH Scale)
The pH scale measures how acidic or basic a solution is.
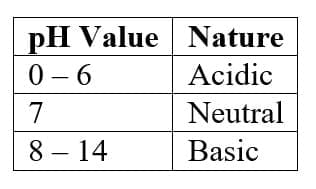
Important points:
- Lower pH → stronger acid
- Higher pH → stronger base
- Neutral solutions have pH = 7
The pH scale ranges from 0 to 14.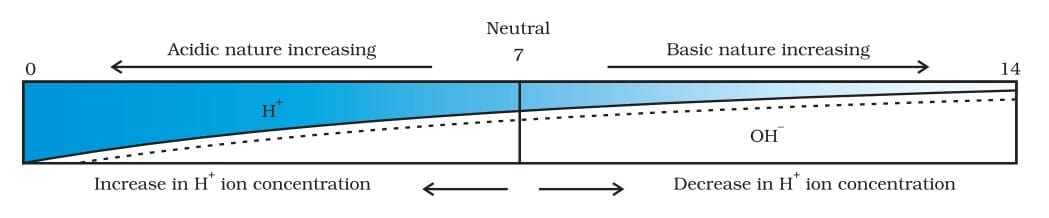 Variation of pH with the change in concentration of H+ (aq) and OH- (aq) ions
Variation of pH with the change in concentration of H+ (aq) and OH- (aq) ions
9. Importance of pH in Everyday Life
(a) pH in our digestive system
Our stomach produces hydrochloric acid for digestion.
Excess acid causes indigestion.
Antacids such as magnesium hydroxide neutralise excess acid.
(b) pH and tooth decay
When mouth pH falls below 5.5, tooth enamel begins to dissolve.
Toothpaste is basic and helps neutralise acids.
(c) pH of soil
Plants grow well only within a specific pH range.
If soil becomes too acidic, farmers treat it with quicklime or slaked lime.
(d) pH in acid rain
Rainwater with pH less than 5.6 is called acid rain.
It harms aquatic life and damages ecosystems.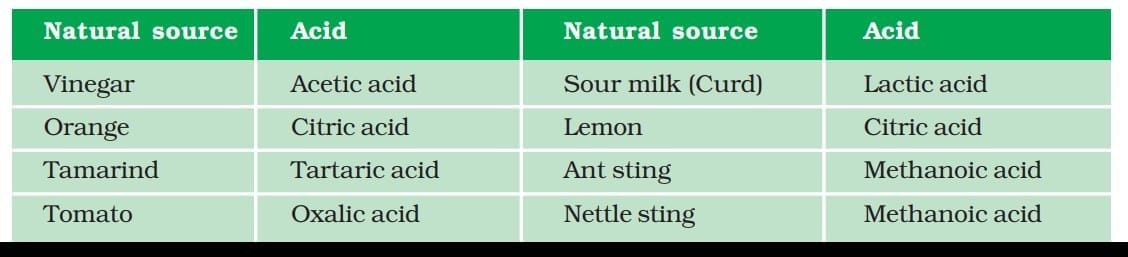 Some naturally occurring acids
Some naturally occurring acids
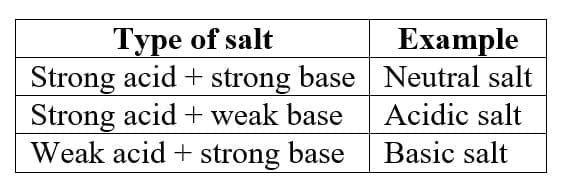
10. Salts
A salt is formed when an acid reacts with a base.
Example: HCl + NaOH → NaCl + H₂O
Salts may be:
- Neutral
- Acidic
- Basic
11. Chemicals from Common Salt
Common salt (NaCl) is an important raw material for many chemicals.
(a) Sodium Hydroxide (NaOH)
Produced by electrolysis of brine in the chlor-alkali process.
Reaction: 2NaCl + 2H₂O → 2NaOH + Cl₂ + H₂
Products obtained:
- Sodium hydroxide
- Chlorine gas
- Hydrogen gas
(b) Bleaching Powder
Prepared by passing chlorine gas over dry slaked lime.
Reaction: Ca(OH)₂ + Cl₂ → CaOCl₂ + H₂O
Uses:
- Bleaching cotton and linen
- Disinfecting drinking water
- Oxidising agent
(c) Baking Soda
Chemical name: Sodium hydrogencarbonate (NaHCO₃)
Prepared from common salt.
Uses:
- Baking powder
- Antacids
- Soda-acid fire extinguishers
On heating: 2NaHCO₃ → Na₂CO₃ + H₂O + CO₂
The CO₂ gas makes cakes soft and spongy.
(d) Washing Soda
Chemical formula: Na₂CO₃ · 10H₂O
Uses:
- Cleaning agent
- Softening hard water
- Manufacturing glass, soap and paper
12. Water of Crystallisation
Some salts contain a fixed number of water molecules in their crystals.
This is called water of crystallisation.
Example: CuSO₄ · 5H₂O (Copper sulphate)
When heated:
- Water molecules are removed
- Blue copper sulphate becomes white
Adding water restores the blue colour.
13. Plaster of Paris (POP)
Plaster of Paris is produced by heating gypsum.
Reaction:
CaSO₄ · 2H₂O → CaSO₄ · ½H₂O + 1½H₂O
When mixed with water:
CaSO₄ · ½H₂O + H₂O → CaSO₄ · 2H₂O
Uses:
- Plaster for fractured bones
- Making statues and toys
- Decorative materials
FAQs on Cheat Sheet: Acids, Bases and Salts
| 1. What are the common properties of acids and bases? |  |
| 2. How is the strength of acids and bases determined? |  |
| 3. Why is pH important in everyday life? |  |
| 4. What are salts and how are they formed? |  |
| 5. What is the significance of the pH scale? |  |







