Cheat Sheet: Carbon and its Compounds
1. Introduction to Carbon
Carbon is a very important element because it forms the basis of all living organisms and many materials used in daily life such as fuels, plastics, medicines, and fabrics.
Carbon compounds have low melting and boiling points compared to ionic compounds because they form covalent bonds rather than ionic bonds.
2. Bonding in Carbon - Covalent Bond
Carbon has 4 electrons in its outermost shell (valency = 4). Instead of gaining or losing four electrons, carbon shares electrons with other atoms.
This sharing of electrons forms a covalent bond.
Example:
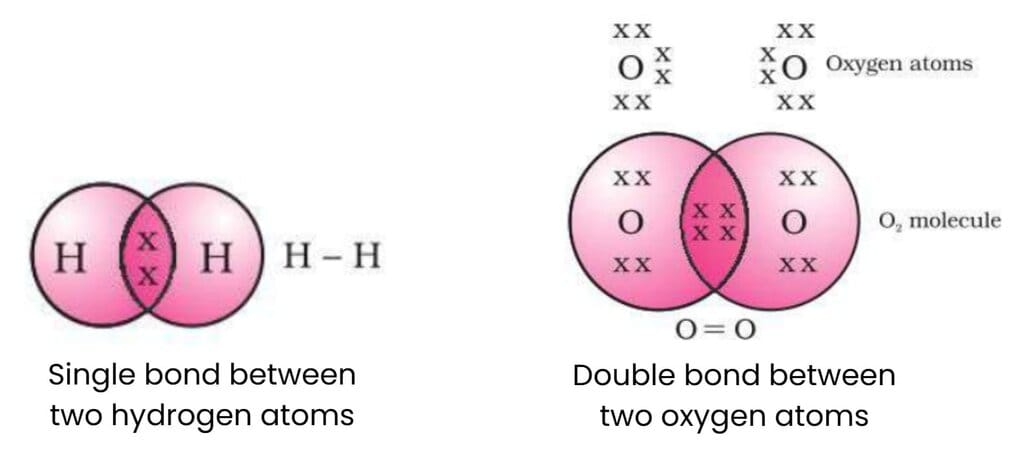
For example: In methane (CH₄), carbon shares electrons with four hydrogen atoms to complete its octet.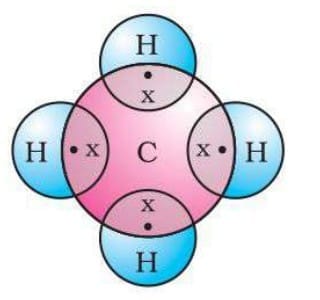 Electron dot structure for methane
Electron dot structure for methane
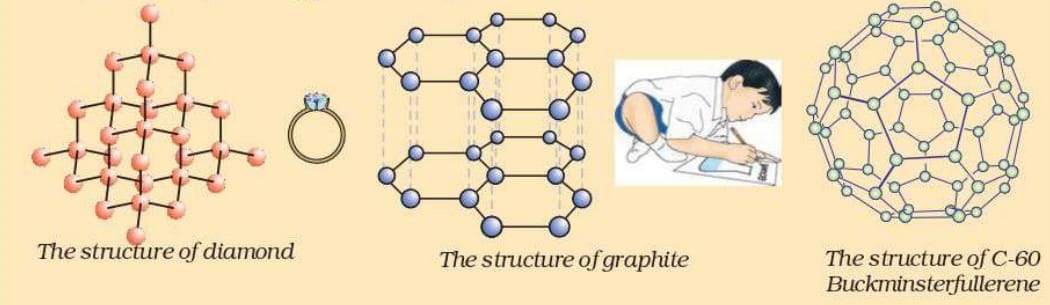
3. Allotropes of Carbon
Carbon exists in different forms called allotropes, which have different physical properties but the same chemical element.
Diamond
- Hardest natural substance
- Each carbon atom bonded to four other carbon atoms
- Very high melting point
- Used for cutting and drilling tools
Graphite
- Soft and slippery
- Carbon atoms arranged in layers
- Conducts electricity
- Used in electrodes and lubricants
Fullerenes
- Carbon molecules shaped like hollow spheres or tubes
- Example: Buckminsterfullerene (C₆₀).
4. Versatile Nature of Carbon
Carbon forms a large number of compounds because of two important properties.
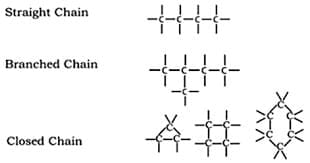
(a) Catenation
Catenation is the ability of carbon atoms to bond with each other to form long chains or rings.
Example:
- Straight chains
- Branched chains
- Ring structures
(b) Tetravalency
Carbon has four valence electrons, so it can form four covalent bonds with other atoms.
Example atoms bonded with carbon:
- Hydrogen
- Oxygen
- Nitrogen
- Sulphur
- Chlorine
Because of these properties, carbon forms millions of compounds.
5. Saturated and Unsaturated Compounds
Carbon compounds can be classified depending on the type of bond between carbon atoms.

Hydrocarbons with:
- Double bonds → Alkenes
- Triple bonds → Alkynes.
6. Chains, Branches and Rings
Carbon atoms can form different structures.
Straight Chain
Example: Butane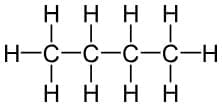
Branched Chain
Example: Isobutane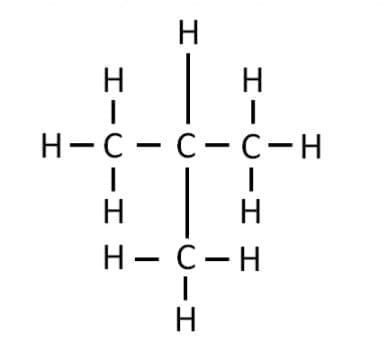
Ring Structure
Example: Cyclohexane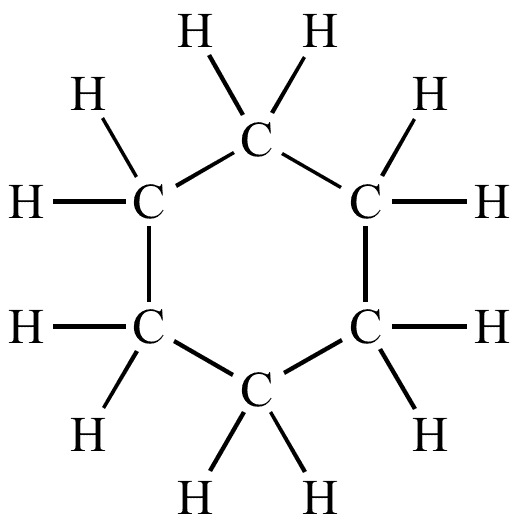
Different structures having the same molecular formula but different arrangements are called isomers.
7. Functional Groups
A functional group is an atom or group of atoms that determines the chemical properties of an organic compound.
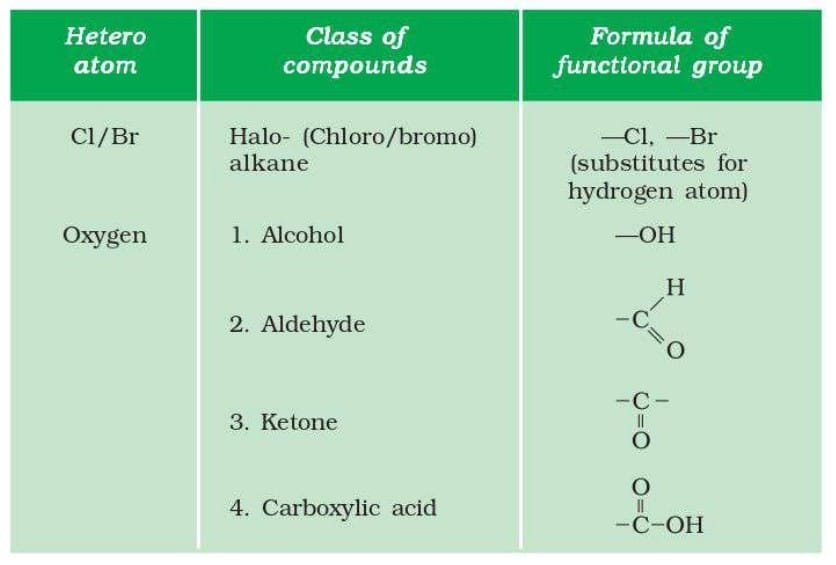
Functional groups give similar chemical properties to compounds within the same group.
8. Homologous Series
A homologous series is a group of organic compounds that:
- Have the same functional group
- Have similar chemical properties
- Differ by a -CH₂ unit
Example series of alkanes: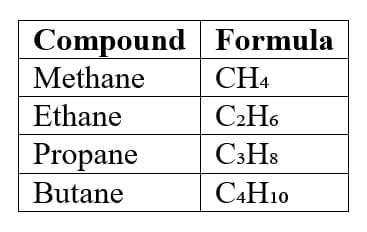
Physical properties change gradually as molecular mass increases.
9. Nomenclature of Carbon Compounds
Naming organic compounds depends on:
- Number of carbon atoms in the main chain
- Functional group present
- Type of bond (single, double, triple)
Examples: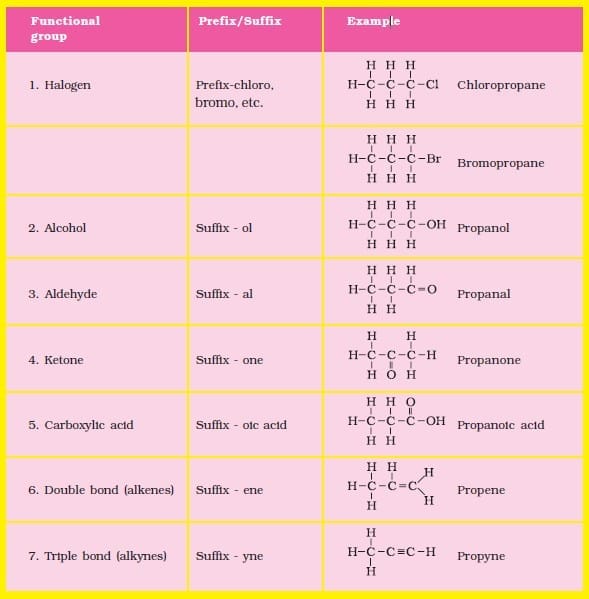
10. Chemical Properties of Carbon Compounds
(a) Combustion
Carbon compounds burn in oxygen to produce carbon dioxide, water, heat, and light.
Example: CH₄ + O₂ → CO₂ + H₂O + heat
Incomplete combustion produces carbon monoxide and soot.
(b) Oxidation
Oxidation means adding oxygen or removing hydrogen.
Example: CH₃CH₂OH → CH₃COOH
Alcohol can be oxidised to form carboxylic acids.
(c) Addition Reaction
Unsaturated hydrocarbons add hydrogen to become saturated hydrocarbons.
Example: C₂H₄ + H₂ → C₂H₆
This reaction occurs in the presence of nickel or palladium catalyst.
This process is used in hydrogenation of vegetable oils.
(d) Substitution Reaction
In saturated hydrocarbons, hydrogen atoms can be replaced by other atoms.
Example: CH₄ + Cl₂ → CH₃Cl + HCl
This occurs in the presence of sunlight.
11. Important Compounds of Carbon
Ethanol (Alcohol)
Properties:
- Colourless liquid
- Used in medicines, perfumes, and alcoholic drinks
Reaction with Sodium
2Na + 2C₂H₅OH → 2C₂H₅ONa + H₂
Dehydration
C₂H₅OH → C₂H₄ + H₂O (with conc. H₂SO₄)
Ethanol can be converted to ethene by dehydration.
Ethanoic Acid (Acetic Acid)
Properties:
- Gives vinegar its sour taste
- Freezes at 16.6°C (called glacial acetic acid)
Esterification Reaction
Alcohol + Carboxylic acid → Ester + Water
Example: CH₃COOH + C₂H₅OH → CH₃COOC₂H₅ + H₂O
Esters have pleasant fruity smells and are used in perfumes and flavourings.
12. Soaps and Detergents
Soap
Soap molecules have two ends:

In water, soap forms micelles, which trap dirt and grease so they can be washed away.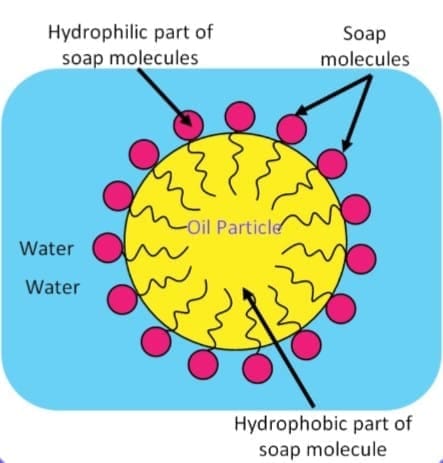 Structure of micelle
Structure of micelle
Detergents
Detergents are synthetic cleaning agents that work even in hard water, unlike soaps which form scum.
Examples:
- Washing powders
- Dishwashing liquids
- Shampoos.
FAQs on Cheat Sheet: Carbon and its Compounds
| 1. Why is carbon known as the backbone of organic chemistry? |  |
| 2. What are the different types of covalent bonds that carbon can form? |  |
| 3. How does carbon bonding affect the physical properties of its compounds? |  |
| 4. What is the significance of hybridization in carbon compounds? |  |
| 5. Why are carbon compounds essential for life? |  |







