CBSE Class 9 > Class 9 Notes > Science > Cheatsheet: Is Matter Around Us Pure?
Cheatsheet: Is Matter Around Us Pure?
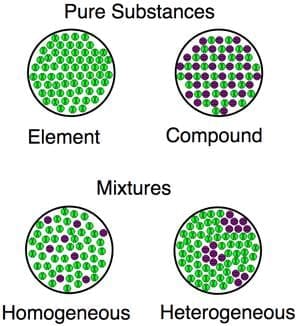
Mixtures
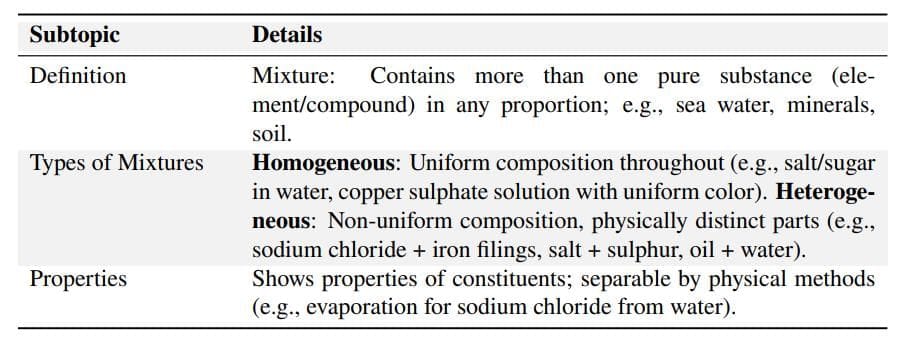
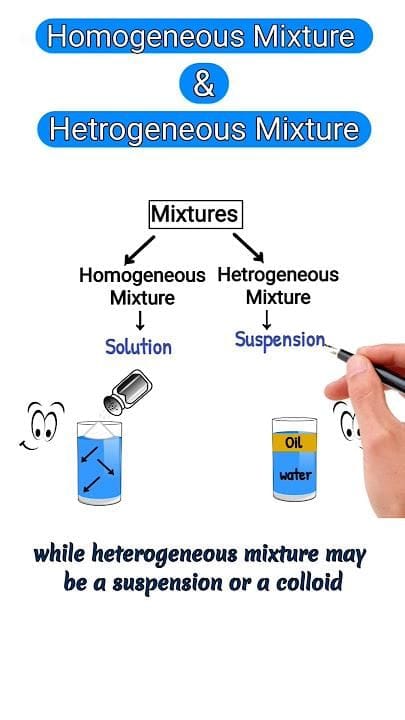
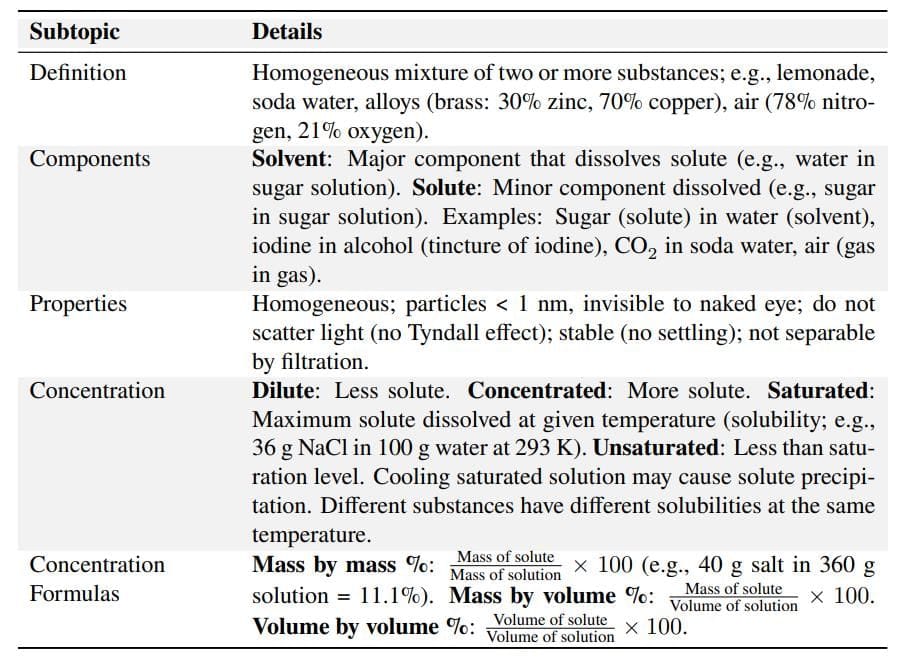
Solutions
Suspensions

Colloids

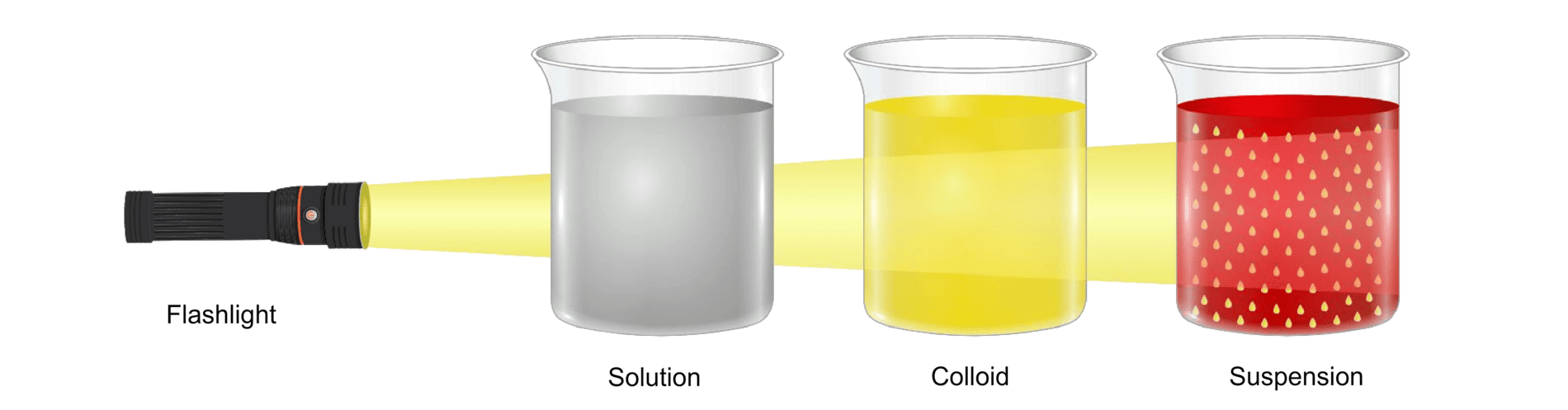 Tyndall Effect
Tyndall Effect
Physical and Chemical Changes

Types of Pure Substances
1. Elements

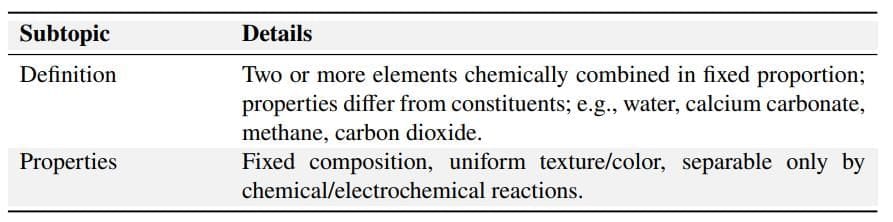
2. Compounds
Mixtures vs Compounds

The document Cheatsheet: Is Matter Around Us Pure? is a part of the Class 9 Course Science Class 9.
All you need of Class 9 at this link: Class 9
FAQs on Cheatsheet: Is Matter Around Us Pure?
| 1. What is the difference between a solution and a suspension? |  |
Ans. A solution is a homogeneous mixture where the solute is completely dissolved in the solvent, such as salt in water (NaCl in H₂O). In a suspension, the particles do not dissolve but remain suspended in the liquid, like sand in water. Over time, the particles in a suspension will settle to the bottom, while in a solution, the mixture remains uniform.
| 2. What are colloids and how do they differ from solutions and suspensions? |  |
Ans. Colloids are mixtures where very small particles are dispersed throughout a medium, but they do not settle out like in a suspension. An example of a colloid is milk, where fat globules are dispersed in water. Unlike solutions, colloids exhibit the Tyndall effect, scattering light due to the size of the dispersed particles, which is not observed in true solutions.
| 3. What are physical changes and how do they differ from chemical changes? |  |
Ans. Physical changes are changes that affect one or more physical properties of a substance without altering its chemical composition, such as melting ice into water. Chemical changes, on the other hand, involve a transformation that alters the chemical composition of the substance, resulting in new substances, like rust forming on iron (Fe) when it reacts with oxygen (O₂).
| 4. What are the types of pure substances? |  |
Ans. Pure substances can be classified into two main types: elements and compounds. Elements consist of only one type of atom, such as oxygen (O) or gold (Au), while compounds are made up of two or more different types of atoms chemically bonded together, such as water (H₂O) or carbon dioxide (CO₂).
| 5. How can we distinguish between mixtures and compounds? |  |
Ans. Mixtures are physical combinations of two or more substances that retain their individual properties and can be separated by physical means, such as filtering or distillation. Compounds are substances formed when two or more elements chemically combine in fixed ratios, resulting in new properties distinct from the original elements, like sodium chloride (NaCl) formed from sodium (Na) and chlorine (Cl).
Related Searches
practice quizzes, shortcuts and tricks, Summary, Extra Questions, Cheatsheet: Is Matter Around Us Pure?, video lectures, Free, Exam, pdf , Objective type Questions, Important questions, ppt, past year papers, Cheatsheet: Is Matter Around Us Pure?, Semester Notes, Sample Paper, Cheatsheet: Is Matter Around Us Pure?, study material, MCQs, mock tests for examination, Previous Year Questions with Solutions, Viva Questions;







