NCERT Solutions: Particulate Nature of Matter
Probe and Ponder (Page 98)
Q1: Why is it possible to pile up stones or sand, but not a liquid like water?
Ans: Stones and sand are solids. In solids, particles are tightly packed and held together by relatively strong interparticle attractions. This fixed arrangement gives solids a definite shape and allows them to rest on one another, so they can be piled up. Water is a liquid; its particles have weaker attractions and can move past one another, so liquid flows and cannot keep a free-standing pile of its own shape.
Q2: Why does water take the shape of folded hands but lose that shape when released?
Ans: Water is a liquid. Its particles can move around and rearrange themselves to fit the shape of the container (in this case, folded hands). When the hands are opened, gravity and the ability of the particles to move cause the water to flow and change shape again, because liquids have a definite volume but no fixed shape.
Q3: We cannot see air, so how does it add weight to an inflated balloon?
Ans: Air is a mixture of gases made of tiny particles (molecules) that cannot be seen individually. When a balloon is inflated, these gas particles occupy space inside the balloon and add mass to it. Because mass is present, the balloon becomes heavier - that is, air inside the balloon contributes to its weight.

Q4: Is the air we breathe today the same that existed thousands of years ago?
Ans: Yes. Matter, including air, is continuously recycled in nature through processes such as respiration, photosynthesis and weather cycles. The atoms and molecules in the air today have existed for a very long time and keep circulating through the environment.
Q5: Share your questions?
Ans:
- How small are the tiniest particles of matter, and can we ever see them?
- Why do some solids melt easily while others need very high temperatures?
- If gases have no fixed volume, how do they stay contained in the atmosphere?
- What happens to the particles when a substance changes from solid to liquid?
- Why don't all solids dissolve in water like sugar does?
InText (Page 99 - 110)
Q1: Is every speck of this fine chalk powder still composed of the same substance, or has it changed into something else on breaking or grinding? (Page 99)
Ans: Yes. Even after breaking or grinding, each speck of chalk powder is chemically the same as the original chalk. Grinding is a physical change that reduces particle size but does not alter the substance's chemical composition.
Q2: Are the units of chalk obtained in this manner considered the smallest units of chalk? (Page 100)
Ans: No. The particles obtained by grinding chalk are smaller pieces but not the smallest possible units. Each of these pieces is still made of many smaller constituent particles (atoms or molecules), which are the true basic units of the substance.
Q3: Chalk and sugar can both be broken down into their constituent particles. But how are the constituent particles held together to form the solid pieces we see? (Page 101)
Ans: The constituent particles in solids are held together by interparticle forces of attraction. These forces keep the particles closely packed and fixed in position, giving solids a definite shape and volume.
Q4: In the solid state, is there any way to move these particles apart? (Page 102)
Ans: In a solid, particles can only vibrate about fixed positions because they are very close together and strongly attracted to one another. To move them further apart, we must supply energy (for example, by heating) so that the solid melts into a liquid, where particles can move more freely.
 Solid Particles
Solid Particles
Q5: Solids have a definite volume; what about liquids and gases? (Page 103)
Ans:
- Liquids: Liquids have a definite volume but no definite shape; they take the shape of the container that holds them.
- Gases: Gases have neither definite shape nor definite volume; they expand to fill the entire container or space available to them.
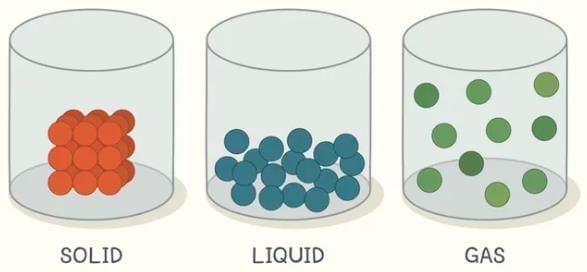
Q6: Do gases also have a fixed volume? (Page 105)
Ans: No. Gases do not have a fixed volume because their particles are far apart and move freely. They spread out to fill the space available, so the volume of a gas depends on the container it is in.
Q7: Sugar and sand are both solids. Why does sugar dissolve in water, but sand does not? (Page 108)
Ans: Sugar dissolves in water because water molecules can surround and separate the sugar particles (molecules), breaking the forces that hold sugar crystals together so the sugar disperses at the molecular level. Sand (silicon dioxide) has a rigid crystal structure and is not broken apart by water molecules, so sand does not dissolve and instead sinks or remains suspended.
Q8: How can we demonstrate the movement of gas particles that cannot be seen with the naked eye? (Page 110)
Ans: We can use visible tracers such as smoke or coloured vapours to show gas motion. For example, smoke from incense spreads through the air and its movement shows that gas particles move randomly and can carry other particles with them.
Keep the Curiosity Alive (Page 113 - 114)
Q1: Choose the correct option.
The primary difference between solids and liquids is that the constituent particles are:
(i) closely packed in solids, while they are stationary in liquids.
(ii) far apart in solids and have fixed position in liquids.
(iii) always moving in solids and have fixed position in liquids.
(iv) closely packed in solids and move past each other in liquids.
Ans: (iv)
Explanation: In solids, particles are closely packed and fixed in position due to strong interparticle attractions. In liquids, particles are still close but can move or slide past each other, allowing the liquid to flow and take the shape of its container.
Q2: Which of the following statements are true? Correct the false statements.
(i) Melting ice into water is an example of the transformation of a solid into a liquid.
Ans: True
Explanation: Melting is the process where a solid becomes a liquid on heating; ice turning into water is a direct example.
(ii) Melting process involves a decrease in interparticle attractions during the transformation.
Ans: True
Explanation: As a solid melts, particles gain energy and move farther apart, which effectively reduces the strength of interparticle attractions so particles can move past one another.
(iii) Solids have a fixed shape and a fixed volume.
Ans: True
Explanation: Solids keep their own shape and volume because their particles are closely packed and held in fixed positions.
(iv) The interparticle interactions in solids are very strong, and the interparticle spaces are very small.
Ans: True
Explanation: Strong attractions hold particles close together in solids, leaving very small spaces between them.
(v) When we heat camphor in one corner of a room, the fragrance reaches all corners of the room.
Ans: True
Explanation: Camphor sublimes on heating: it changes from solid to gas and its vapour spreads through the room by the random motion of gas particles, carrying the smell everywhere.
(vi) On heating, we are adding energy to the camphor, and the energy is released as a smell.
Ans: False
Explanation: Heating adds energy to camphor, causing sublimation (solid → gas). The smell is due to camphor molecules entering the air as vapour, not because the energy itself is released as a smell.
Q3: Choose the correct answer with justification. If we could remove all the constituent particles from a chair, what would happen?
(i) Nothing will change.
(ii) The chair will weigh less due to lost particles.
(iii) Nothing of the chair will remain.
Ans: (iii) Nothing of the chair will remain.
Explanation: A chair is made up of its constituent particles (atoms and molecules). If all these particles were removed, there would be no material left to give shape, mass or structure - the chair would cease to exist.
Q4: Why do gases mix easily, while solids do not?
Ans: Gas particles are far apart and move rapidly in all directions. Because of these large separations and high speeds, gases spread out and mix quickly with other gases. Solids have particles very close together and fixed in position, so they do not mix unless they are melted or broken into powder.
Q5: When spilled on the table, milk in a glass tumbler flows and spreads out, but the glass tumbler stays in the same shape. Justify this statement.
Ans: Milk is a liquid; its particles can move past each other, so milk flows and spreads on the table to take the shape of the surface. The glass tumbler is a solid; its particles are tightly fixed, giving the tumbler a definite shape that does not change when milk is spilled.
Q6: Represent diagrammatically the changes in the arrangement of particles as ice melts and transforms into water vapour.
Ans: As ice melts and then vaporises, the arrangement and motion of particles change as follows:
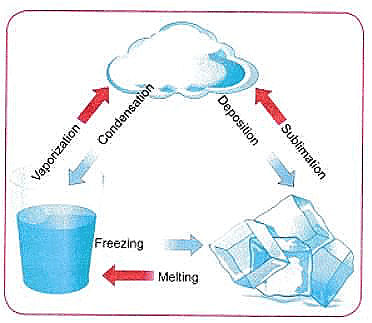
Ice (Solid)
- Arrangement: Water molecules are held in a regular, crystalline structure and are closely packed.
- Movement: Molecules vibrate about fixed positions.
Liquid Water
- Arrangement: Molecules are close but not in a fixed pattern; there is more space compared with a solid.
- Movement: Molecules can move around and slide past each other.
Water Vapour (Gas)
- Arrangement: Molecules are far apart and randomly distributed.
- Movement: Molecules move rapidly and randomly, filling the available space.
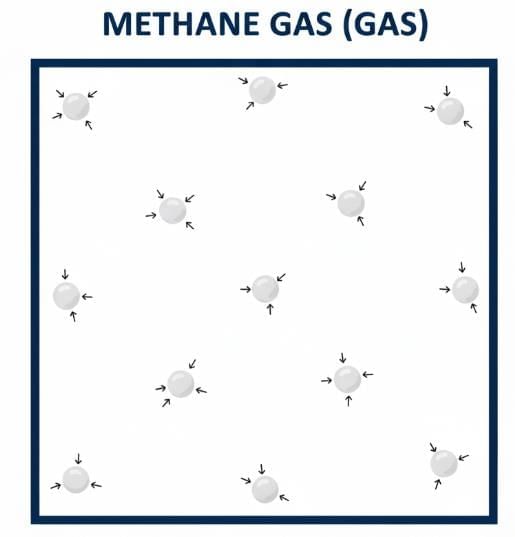
Q7: Draw a picture representing particles present in the following:
(i) Aluminium foil
(ii) Glycerin
(iii) Methane gas
Ans: Pictorial representation of particles of aluminium foil, glycerin and methane gas is shown below.


Q8: Observe Fig. 7.16a which shows the image of a candle that was just extinguished after burning for some time. Identify the different states of wax in the figure and match them with Fig. 7.16b showing the arrangement of particles.
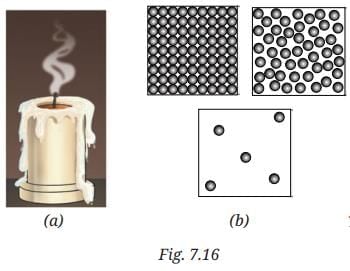
Ans: Different states of wax:
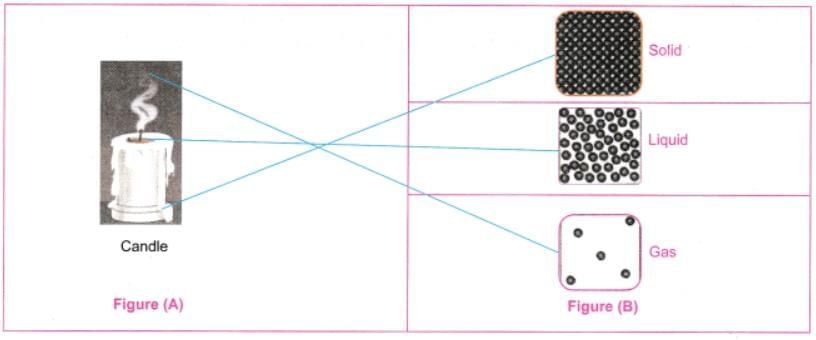
- Solid wax (at base): Rigid, unmelted portion - matches tightly packed particles in a fixed arrangement.
- Liquid wax (melted pool): Flowing around the wick - matches particles that are close but can move past one another.
- Gaseous wax (vapour/smoke): Rising as fumes - matches widely spaced, freely moving particles.
The figure illustrates melting (solid → liquid) and evaporation (liquid → gas), with particle arrangement changing from fixed to mobile.
Q9: Why does the water in the ocean taste salty, even though the salt is not visible? Explain.
Ans: Ocean water contains salts (mainly sodium chloride) dissolved as ions at the molecular level. These dissolved particles are too small to see, so the water looks clear; however, their presence gives the ocean its salty taste.
Q10: Grains of rice and rice flour take the shape of the container when placed in different jars. Are they solids or liquids? Explain.
Ans: Both grains of rice and rice flour are solids. Each grain or particle keeps its own shape and volume. They appear to take the shape of the container because many small solid particles can move and settle, allowing the collection to flow like a liquid, but the individual particles remain solid.
Discover, Design, and Debate (Page 115)
Q1: Fix a balloon over the neck of a bottle and put the bottle in hot water. Explore what will happen?
Ans: The balloon inflates. Heating the bottle warms the air inside; the air particles gain kinetic energy, move faster and occupy more space. The increased pressure inside the bottle pushes air into the balloon, causing it to expand. This shows how gas particles respond to heat by moving more and spreading apart.
Q2: Design and create simple models to represent particles of solids, liquids, and gases showing interparticle spacing using clay balls, beads, etc.
Ans:
- Solid: Arrange clay balls in a tight grid or box so they touch one another; this shows minimal spacing and fixed positions.
- Liquid: Place beads in a shallow tray so they are close but can move when the tray is tilted; this shows slight spacing and ability to flow.
- Gas: Scatter beads loosely in a large container so they are far apart and move freely when shaken; this shows large spacing and random motion.
Q3: Pretend to be particles of solids, liquids, and gases, at different temperatures-create and perform a role-play/dance showing particles in motion.
Ans:
- Solid (low temperature): Students stand close together and gently vibrate on the spot to show particles fixed in position.
- Liquid (medium temperature): Students stand close but move slowly around each other, showing sliding motion and flow.
- Gas (high temperature): Students move freely and rapidly in all directions, showing random motion and large separations.
- Increase the speed to represent a rise in temperature and show phase changes such as "melting" when solid dancers start moving like liquid dancers.
Q4: Debate in the class - 'Gases can spread and fill all the available space'. Is this property of gases beneficial or harmful?
Ans:
- Beneficial side: Gases spreading helps in breathing (oxygen reaching cells), dispersal of perfume and heating in kitchens (gas stoves), and many industrial processes. It also allows gases to mix in the atmosphere, which is essential for life and weather.
- Harmful side: The same property can spread pollutants and toxic gases quickly (air pollution, dangerous leaks), causing health hazards and environmental damage such as greenhouse gas effects.
Overall, gas diffusion is useful when controlled and harmful when uncontrolled; safety measures like proper ventilation and leak detection help reduce the risks.
FAQs on NCERT Solutions: Particulate Nature of Matter
| 1. What exactly are particles and why does matter have a particulate nature? |  |
| 2. How do particles in solids, liquids, and gases differ in arrangement and movement? |  |
| 3. Can particles really be seen under a microscope, or are they too small? |  |
| 4. Why do substances expand when heated if particles themselves don't change size? |  |
| 5. How does the particulate nature of matter explain why we can smell perfume from across a room? |  |






