Chapter Notes: Corrosive Poisons
Introduction
Corrosive poisons refer to substances that possess the ability to corrode or 'eat away' at and damage tissues through their chemical actions. These poisons primarily have local effects with minimal impact on the entire body. Corrosive poisons can be categorized into two main groups:
1. Strong Acids
- Inorganic Acids (Mineral Acids): These include:
- Sulfuric Acid
- Nitric Acid
- Hydrochloric Acid
- Hydrofluoric Acid
- Organic Acids: These include:
- Carbolic Acid
- Oxalic Acid
- Salicylic Acid
It is important to note that organic acids are generally weaker than inorganic acids. However, they have the potential to be absorbed into the bloodstream, resulting in both local and systemic effects.
2. Strong Alkalis
- Anhydrous ammonia
- Potassium hydroxide
- Sodium hydroxide
- Ammonium carbonate
- Potassium carbonate
- Sodium carbonate
Inorganic Acids: Sulphuric Acid, Nitric Acid, Hydrochloric Acid
Physical Properties
Comparison of Specific Characteristics of the Three Acids
Action
Only strong acids and alkalis act as corrosives, while their diluted forms function as irritants. Strong acids cause coagulation necrosis, forming a coagulum (eschar) due to their desiccating effect on proteins in surface tissues. This coagulum restricts the acid's ability to penetrate deeper. Conversely, strong alkalis induce liquefaction necrosis, leading to extensive tissue penetration. The squamous epithelium of the esophagus is more resistant to acids than the columnar epithelium of the stomach, but it is more vulnerable to alkalis. As a result, esophageal strictures are more frequent in alkali poisoning, while gastric and pyloric strictures are more common in acid poisoning.
Fig. 32.1: Process of coagulation necrosis caused by mineral acids
Comparison of specific features of sulphuric, nitric, and hydrochloric acids
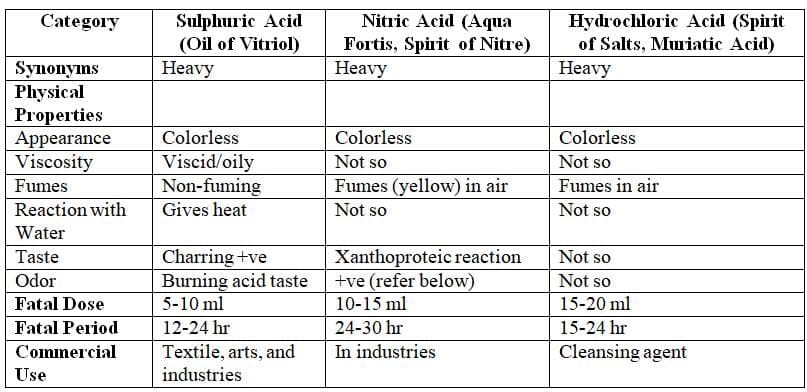
Signs and Symptoms of Corrosive Ingestion
- When corrosive substances are ingested orally, they trigger a range of common gastrointestinal and respiratory signs and symptoms.
- The severity of gastrointestinal symptoms varies depending on the type and concentration of the acid involved.
- Respiratory symptoms are present in all cases of inorganic acid poisoning, with the exception of hydrofluoric acid.
- In the oral cavity, specific signs may include:
- Chalky white teeth
- Swollen blackish tongue
- Swollen lips
- Acid burns, also known as eschar, are often visible as the vomited acid can flow from the mouth down to the neck and chest.
- The abdomen may become distended and tender.
- While consciousness may remain clear in some cases, it can be affected depending on the severity of the poisoning.
Treatment
- In cases of corrosive poisoning, certain common management measures are recommended.
Avoid
- Gastric lavage to prevent gastric perforation (except for organic acids).
- Administering carbonates, as they can produce carbon dioxide, leading to distention and perforation.
- Inducing vomiting is not advised due to the risk of stomach rupture.
- Examples of demulcents include:
- Canned condensed milk
- Beaten egg white
- Vegetable oils
- Starch solution
- Barley water
- Thin gruel
- Immediate dilution with milk or water is recommended within 30 minutes of ingestion, despite rapid tissue injury.
- Do not attempt to neutralise the acid with weak bases or dilute alkalizers like milk of magnesia or lime water, as exothermic reactions may worsen the injury.
- Never give strong alkalis or substances like sodium bicarbonate, as they can produce carbon dioxide gas, increasing the risk of stomach perforation.
- Maintain the airway and provide artificial respiration if there is any respiratory distress.
- Administer morphine or pethidine for pain relief.
- Provide intravenous fluids and electrolytes for dehydration.
- No oral feeding until an endoscopy confirms the extent of injury.
- Use antibiotics to control infections.
- While corticosteroids may help delay or prevent stricture formation in animals, their use in humans is still controversial.
- For skin lesions, use copious saline irrigation and treat with non-adherent gauze and wrapping. Deep second-degree burns may benefit from topical silver sulfadiazine.
- For acid injuries to the eyes, irrigate copiously while retracting the eyelids for 20-30 minutes. Antibiotic eye drops can help prevent infections, but it is wise to refer to an ophthalmologist for further treatment.
Causes of Death
- Immediate causes include:
- Suffocation and asphyxia due to edema of the glottis
- Exhaustion
- Shock from circulatory collapse or perforation of the stomach
- Delayed causes include:
- Starvation
- Dehydration due to stricture of the esophagus or pylorus
- The victim may also die from peritonitis or secondary infections.
Postmortem Findings
- External findings: Corrosion on areas that contacted the substance, including:
- Lips
- Mouth
- Throat
- Chin
- Angle of mouth
- Hands
- Clothing and furniture if spilled on them
- Internal findings: Restricted to the upper gastrointestinal and respiratory tracts, including:
- Pharynx and esophagus: Inflammatory changes with edema and bleeding.
- Stomach: Becomes soft and boggy, appearing black (sulfuric acid), yellowish (nitric acid), or brownish (hydrochloric acid). Contents are starry black or brown.
- Surrounding viscera: Sloughing may occur if the stomach wall is perforated, most commonly seen with sulfuric acid.
- Larynx and trachea: Show corrosive damage and inflammation. Congestion can happen due to inhaling nitric acid fumes.
Chemical Tests
- Sulfuric acid: When sulfuric acid is mixed with barium chloride, it produces a white solid called barium sulfate.
- Nitric acid: Adding nitric acid to a mixture of ferrous sulfate and sulfuric acid in a test tube creates a brown ring at the junction of the two liquids.
- Hydrochloric acid: Mixing hydrochloric acid with silver nitrate results in a thick white solid known as silver chloride.
Autopsy Findings in Cases of Accidental Corrosive Acid Spillage and Burns
- Face and trunk: The autopsy findings in cases of accidental corrosive acid spillage and burns may include severe damage to the face and trunk, with signs of corrosion, burns, and tissue destruction.
- Entire body: In more extensive cases, the entire body may exhibit similar signs of corrosive damage and burns, with affected areas showing significant injury and degradation of tissues.
Medicolegal Significance of Acid Poisoning Cases
- Accidental Ingestion: Most instances of acid poisoning occur accidentally. For instance, sulphuric acid is sometimes mistaken for safe substances like glycerine or castor oil, leading to accidental ingestion.
- Rare for Suicide: Acid poisoning is infrequently used as a method of suicide.
- Uncommon Murder Method: While acid attacks are not common in murder cases, they do occur.
- Abortion Method: In some cases, acids are misused as a method for abortion.
- Vitriolage: This refers to the act of throwing concentrated sulphuric acid on a person's face with the intent to cause harm, disfigurement, or death. The clinical presentation includes severe chemical burns on the face and other skin areas where the acid comes into contact.
- Treatment for Vitriolage: The initial treatment involves:
- Thoroughly washing the affected area with water and mild soap or alkali.
- Providing analgesics for pain relief.
- Administering antibiotics to prevent secondary infections.
- Legal Implications: Vitriolage can result in permanent disfigurement, which is classified as grievous hurt under Section 320 of the Indian Penal Code (IPC). If the act is motivated by jealousy or enmity, there may be additional injuries involved.
- Xanthoproteic Reaction: This is a local reaction that occurs when concentrated nitric acid comes into contact with organic matter, leading to yellowish discoloration of tissues, such as the skin on fingers. The mechanism involves the oxidation of organic matter by nitric acid, resulting in the formation of picric acid, which deeply stains tissues with its yellow-orange color. The presence of a xanthoproteic reaction is indicative of nitric acid poisoning.
- Historical Cases: There have been instances of criminal disposal of dead bodies using acids, such as the acid bath murder case involving John George Haigh, who killed a woman and attempted to dissolve her body in sulphuric acid. Police later discovered remains, including a foot and bone fragments, leading to Haigh's confession.
Hydrofluoric Acid: Overview
Hydrofluoric acid is a clear gas that becomes a fuming liquid when mixed with water. It is commonly used for etching glass and making electric bulbs cloudy.
Fatal Dose
- The amount of hydrofluoric acid that can be fatal varies, but it is usually around 15 ml. This can depend on an individual's sensitivity.
Fatal Period
- Death can occur within a few minutes to 2 hours after exposure, with some cases reported as late as 7 hours.
Hydrofluoric acid affects the body differently than other inorganic acids. The fluoride ion in the acid causes severe damage by binding to calcium and magnesium in the tissues, leading to a condition called hypocalcemia, which worsens the poisoning.
Symptoms after ingestion may include:
- Vomiting blood (haematemesis)
- Low blood volume shock (hypovolemic shock)
- Severe low calcium levels (hypocalcemia)
- Acidosis
- Convulsions
- Coma
Heart issues such as irregular heartbeats (arrhythmias) can occur due to the binding of potassium, magnesium, and calcium ions. Skin contact with hydrofluoric acid can cause very painful and deep burns that heal slowly.
Recommended treatments include:
- Rinse acid burns with plenty of water, apply calcium gluconate gel, and remove damaged tissue if necessary.
- Intra-arterial infusion of 20 percent calcium gluconate or calcium chloride is effective.
- Provide oxygen if exposed to fumes and perform a tracheotomy if necessary.
Injuries to the lips, tongue, and mouth may present as white patches or charring. The esophagus may show torn tissue with bleeding, inflammation, and sores, along with blackening of the stomach. The liver and kidneys may exhibit fatty degeneration.
Most incidents involving hydrofluoric acid are accidental.
Carbolic Acid (Phenol)
Carbolic acid, commonly known as phenol, is a toxic substance recognizable by its strong chemical odor. In its pure state, it appears as colorless, short, prismatic, needle-like crystals. When exposed to air, phenol turns pink and melts. It is fat-soluble, which means it can penetrate fatty tissues and potentially harm the nervous system. Phenol also dissolves in substances like glycerine, ether, alcohol, and to a lesser extent in water. It is especially renowned for its antiseptic and disinfectant qualities.
Other Members of Phenol Group
- Phenol has several derivatives, including cresol, creosote, Lysol, and Dettol. These compounds have similar toxicological effects to phenol but are generally less harmful.
- Cresol is a methyl phenol with meta, ortho, and para isomers, used as a disinfectant and antiseptic.
- Creosote is a blend of phenols, mainly consisting of cresol and guaiacol, often used as a home remedy for coughs and found in various proprietary products.
- Resorcinol is a colourless crystalline substance used to treat several skin conditions such as ringworm, psoriasis, and eczema.
- Lysol is a 50 per cent solution of cresol (3-methyl phenol) mixed with saponified vegetable oil.
- Dettol contains chlorinated phenol, specifically parachlorometaxylenol (PCMX), in a 4.8% solution in isopropyl alcohol, and is mostly non-toxic to adults, although some cases of fatality have been reported in children.
- Thymol is an alkyl derivative of phenol, derived from the oils of Thymus vulgaris, Monarda punctata, or Trachyspermum ammi. It appears as colourless crystals with a strong smell and taste and was previously used as an antihelminthic, antifungal, and antiseptic.
Routes of Absorption
- Through Intact Skin: Phenol can be absorbed through intact skin when applied locally or in case of spillage.
- Gastrointestinal Tract: It can be ingested orally, leading to absorption through the gastrointestinal tract.
- Respiratory Tract: Inhalation of phenol allows absorption through the respiratory tract.
- Per Rectum: Phenol can be absorbed when administered per rectum.
- Per Vaginum: Absorption can also occur when phenol is administered per vaginum.
Mechanism of Action and Effects
- Protoplasmic Poison: Phenol acts as a protoplasmic poison, forming a loose combination with proteins and penetrating deeply into tissues.
- Necrosis and Gangrene: When applied to the skin or mucosa, phenol causes necrosis and gangrene due to its destructive effect on cells.
- Initial Stimulation and Paralysis: Phenol initially stimulates local nerve endings, leading to a subsequent paralysis of these nerves, resulting in anaesthesia.
- Capillary Damage and Clotting: After absorption, phenol causes widespread damage to capillaries and clotting in superficial blood vessels.
- Effects on Central Nervous System, Heart, and Kidneys: Phenol affects the cells of the central nervous system, heart, and kidneys, leading to potential systemic effects.
Metabolism and Excretion
- Phenol is primarily metabolized by the kidneys into hydroquinone and pyrocatechol before being excreted in urine.
- When exposed to air, these metabolites can cause urine to change to an olive green or brown colour, a phenomenon known as carboluria.
- The body typically eliminates phenol and its by-products within 36 hours.
- Despite this rapid excretion, phenol is classified as a nephrotoxic substance.
- Other known nephrotoxins include heavy metals, methanol, oxalic acid, salicylates, phenacetin, EDTA, and penicillamine.
- Carbolism is a condition resulting from poisoning by carbolic acid.
- Symptoms of carbolism can include:
- Headache, dizziness, and ringing in the ears (tinnitus)
- Nausea, vomiting, diarrhea, and stomach pain
- Muscle spasms and seizures
- Loss of consciousness and coma
- Cold, clammy skin and excessive sweating
- Difficulty breathing with a bluish tint to the skin (cyanosis)
- Dilated pupils
- If a person survives the initial 48 hours after exposure, they may develop carboluria, followed by a lack of urine production (anuria).
- The urine may appear dark and smoky at first, changing to olive green when left to stand.
- Severe cases of carbolism may lead to methemoglobinemia, a condition where hemoglobin is altered and cannot effectively carry oxygen.
- In critical instances of carbolic acid poisoning, death can result from failure of the respiratory and circulatory systems.
Diagnosis
- Corrosions on the face, around, and inside the mouth (greyish white if phenol or brownish if Lysol).
- Phenolic odour (breath/vomitus).
- Carboluria
- Dilated pupils
- Stertorous breathing
- 5-15 gm
- Fatal period. 2-12 hours (rapid death if injected intrauterine).
- Toxicity rating -4
- It depends on the route of administration.
1. Poisoning through Skin Absorption:
- Remove contaminated garments and cleanse the affected area by mopping with a wet cloth and washing with soap and water.
- Apply olive oil, methylated spirits, or 10% ethyl alcohol to the area to prevent further absorption.
- Move the victim to fresh air and encourage deep breathing.
- Administer normal saline with sodium bicarbonate via intravenous drip.
2. Poisoning through Oral Route:
- Perform gastric lavage, as phenol, despite damaging the stomach wall, also hardens it. Use plenty of lukewarm water with animal charcoal, olive oil, magnesium or sodium sulfate, or saccharated lime, soap solution, or 10% glycerin. After lavage, leave 30 gm of magnesium sulfate or medicinal liquid paraffin in the stomach.
- Administer egg white and Epsom salt or demulcents orally.
- Provide symptomatic treatment.
- Administer artificial respiration if necessary.
- Perform tracheal aspiration of froth or secretions.
- Give glucose saline to induce diuresis.
External Manifestations:
- Greyish or brownish corrosions may be observed at the angle of the mouth, chin, front of the body, arms, and hands due to splashes, accompanied by a characteristic phenolic odour.
Internal Manifestations:
- Gastrointestinal Mucosa Corrosion" All cases of oral poisoning exhibit corrosion of the gastrointestinal mucosa, laryngeal, and pulmonary edema.
- Stomach Changes: Opening the stomach will release a phenolic odour. The stomach wall appears hardened, resembling a leather bottle, with marked corrosion and swelling of mucosal folds. The gastric mucosa shows coagulated greyish or brownish silvery mucus, and normal mucosal folds appear dark red.
- Kidney Changes: Haemorrhagic nephritis may be observed in the kidneys if the victim survives for some time after poisoning.
- Vomitus and Gastric Lavage Collection: These may show partially detached gastric mucosa.
Preservative for Viscera:
- Use a supersaturated solution of sodium chloride for sending viscera for chemical examination.
Common Uses of Phenol:
- Phenol is commonly used as a disinfectant and preservative for vaccines and sera.
Historical Context of Fatalities:
- Fatalities due to phenol were more common in the past than they are now.
- Accidental poisoning remains a concern, particularly in residential settings, often due to accidental consumption or body spillage.
Rare Uses in Suicidal and Homicidal Cases:
- Phenol is rarely used in suicidal and homicidal cases due to its strong odour.
Illegal Use as Abortifacients:
- Phenolic solutions are illegally used by quacks as abortifacients, syringing them into the uterine cavity to induce criminal abortion.
Ochronosis and Chronic Phenol Poisoning:
- Ochronosis is a condition associated with alkaptonuria, where homogentisic acid deposits in cartilages, ligaments, and fibrous tissues.
- In chronic phenol poisoning (now infrequent), phenolic metabolites pyrocatechol and hydroquinone may deposit in cartilages and ligaments, causing dark pigmentation.
Oxalic Acid: Overview
Physical Properties
It is a colorless, prismatic crystalline compound resembling magnesium sulfate (MgSO₄) and zinc sulfate (ZnSO₄).
Fatal Dose and Period
- Fatal Dose: 15-20 mg
- Fatal Period: 1-2 hours
- Toxicity Rating: 4
Mechanism of Action
- Local Effects: Acts as a corrosive, severely damaging skin and mucous membranes.
- Systemic Effects(upon absorption into the bloodstream):
- Electrolyte Disruption: Depletes tissue calcium, causing hypocalcemia.
- Cardiovascular System: Induces shock, potentially leading to death.
- Renal System: Causes tubular necrosis, resulting in uremia and possible death.
Signs and Symptoms
The clinical presentation varies based on the dose and timing:
Fulminating (High Dose, ≥15 g orally):
- Immediate sour, acidic taste and throat constriction.
- Burning pain from mouth to epigastrium, radiating across the abdomen.
- Symptoms include epigastric tenderness, nausea, coffee-ground vomiting, intense thirst, diarrhea, electrolyte imbalances, and potentially death.
Acute: Primarily driven by hypocalcemia, presenting with:
- Muscle irritability, tenderness, tetany, and convulsions.
- Tingling in extremities, coma, cardiovascular collapse, and death.
Delayed: Characterized by uremia, with symptoms including:
- Reduced urine output, presence of albumin, blood, and envelope-shaped calcium oxalate crystals (visible under a microscope).
Differences between oxalic acid, and magnesium and zinc sulphates
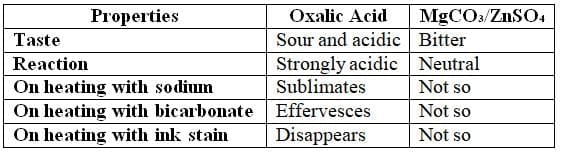
Treatment
- Gastric Lavage: Use calcium lactate (2 teaspoons per lavage).
- Antidotes: Administer lime water, calcium lactate, calcium gluconate, calcium chloride, or chalk suspended in water or milk orally. These form insoluble calcium oxalate, which is easily excreted.
- Intravenous Calcium: 10 ml calcium gluconate IV, administered frequently.
- Parathyroid Extracts: 100 units intramuscularly.
- Supportive Measures:
- Demulcent drinks to soothe mucosa.
- Bowel wash with enema and purgatives (e.g., castor oil).
- Symptomatic treatment as needed.
Postmortem Findings
- External: Typically no specific findings; burns on the face or skin are rare.
- Internal:
- Mouth and Upper GI: Mucosa of the mouth, tongue, pharynx, and esophagus may appear bleached, whitened, scalded, or red if a strong solution was ingested.
- Stomach:
- Mucosa is reddened with punctate erosions, giving a "velvety red" or blackish appearance.
- Stomach wall is softened but not perforated.
- Contents may appear gelatinous brown due to acid hematin formation.
- Kidneys: Swollen and congested; histopathological examination shows tubules filled with oxalate crystals.
- Other Viscera: Generally congested.
Medicolegal Importance
- Accidental Ingestion: Commonly mistaken for magnesium sulfate.
- Suicidal/Homicidal Use: Rare due to the substance's distinct taste.
- Abortifacient: Occasionally used to induce illegal abortions.
- Other Uses:
- Illegally used to erase signatures.
- Found naturally as oxalates in vegetables like beets, spinach, rhubarb, and cabbage, though poisoning from these sources is rare.
- Commercial Applications:
- Cleaning or bleaching leather.
- Bookbinding.
- Calico printing.
- Removing iron stains from linen or ink stains.
Formic Acid (Methanoic Acid, Formylic Acid)
Formic acid is a clear liquid that has a strong odor and is completely soluble in water. It is widely used as a descaler or bath cleaner. Individuals working with airplane glue, cellulose formate, and in the tanning industry are often exposed to a 60 percent solution of formic acid.
When formic acid comes into contact with the skin, it can cause several adverse effects, including:
- Brownish discoloration of the skin
- Dermatitis
- Formation of pustules
- Development of vesicles
- In some cases, sloughing of the skin, depending on the concentration and duration of exposure
Health Risks
Formic acid poses serious health risks, including:
- Potentially fatal gastrointestinal damage
- Severe metabolic acidosis
- Intravascular hemolysis
- Disseminated intravascular coagulation
Accidental ingestion in children typically does not result in death due to the strong taste deterring them from consuming lethal amounts. However, it can be a concern in suicide attempts. Symptoms of formic acid exposure include:
- Acute tracheobronchitis
- Cough
- Sore throat
- Chest pain
- Light-headedness
Skin burns caused by formic acid can lead to systemic toxicity. If absorbed into the body, it may cause:
- Systemic acidosis
- Hematuria
- Renal damage
The metabolism of methanol can produce toxic formic acid metabolites. Treatment for formic acid exposure includes:
- Correcting acidosis with intravenous sodium bicarbonate
- Treating renal failure with hemodialysis
Formic acid is utilized in various applications such as:
- Dyeing colorfast wool
- Electroplating
- Coagulating and regenerating latex rubber
- Dehairing and tanning leather
- Manufacturing acetic acid, airplane dope, allyl alcohol, cellulose formate, phenolic resins, and laundry oxalates
- Industries such as textiles, insecticides, refrigeration, and paper
In South India, formic acid is commonly reported as a method of self-harm, particularly among low-income families and laborers. In Europe, although relatively rare, it is also known as a method for suicide.
Salicylic Acid
Salicylic acid, a compound known as acetylsalicylic acid and commonly called Aspirin.
Strong Alkalis
Alkalis, like acids, can be harmful when concentrated, but they are less dangerous when diluted. These substances are found in various household products such as:
- Drain cleaners
- Oven cleaners
- Dishwasher products
- Some paint strippers
Alkalis are also used in various industries due to their properties.
Mechanism of Action
Alkalis typically contain hydroxyl groups. When dissolved in water, they release hydroxide ions. Alkali agents harm the gastrointestinal tract through:
- Liquefaction necrosis, which involves the breakdown of fats and proteins
- Deep tissue penetration
Unlike acids, alkalis cause significant damage as the injury can worsen rapidly and may continue to progress for weeks after it begins.
Alkalis tend to cause more serious harm to the esophagus compared to the stomach, which is the opposite of acids. Severe damage to the esophagus can occur if the pH is above 11. However, if a large amount of a corrosive substance is swallowed, damage can occur from the mouth to the small intestine.
Anhydrous Ammonia
Physical Properties
Ammonia is an irritant gas, highly soluble in water. When dissolved, it forms a strong ammonia solution (Liquor Ammoniae fortis), known as Spirits of Hartshorn, containing 32.5% ammonia. This colorless, pungent liquid is used in agriculture, mining, plastics, and explosives industries.
Mechanism of Action
Ammonia is absorbed via the respiratory tract, causing pulmonary edema and direct mucosal injury through alkaline burns.
Signs and Symptoms
Inhalation:
Runny nose, increased salivation, asthma, severe upper respiratory irritation, pneumonia, pulmonary edema, bronchitis, and obstructive lung disease.
Death typically occurs due to bronchopneumonia.
Ingestion:
Intense pain, dysphagia, and potential esophageal stenosis.
Eye Exposure:
Watery eyes, corneal damage, conjunctivitis, palpebral edema, and possible blindness in severe cases.
Fatal Dose
Liquid Form: 10-30 ml.
Gaseous Form: 0.5% in air.
Treatment
Remove from exposure and ensure rest.
Administer symptomatic treatment.
For eye exposure, irrigate with water and apply topical antibiotics.
Postmortem Findings
- Characteristic ammonia odor.
- Brownish staining of affected skin.
- Grayish sloughing of impacted mucosa.
- Congestion of gastrointestinal walls (rarely perforated), respiratory tract, and pulmonary edema.
Other Alkalies
Potassium Hydroxide and Sodium Hydroxide
These compounds are commonly used as drain and oven cleaners.
Fatal Dose: 5 grams.
Ammonium Carbonate
This appears as a hard, translucent crystalline mass with a strong ammoniacal taste and pungent smell.
Potassium and Sodium Carbonate
These are white crystalline powders, highly soluble in water but insoluble in alcohol, used for cleaning and washing purposes.
Fatal Dose: 15 grams.
Signs and Symptoms
Ingestion causes an acrid, soapy taste and a burning sensation. Vomiting and stools may contain altered blood. Prolonged exposure may lead to liver damage.
Tests
- Carbonate alkalis form a white precipitate with inorganic acids.
- Hydroxide alkalis produce a yellow precipitate with silver nitrate.
Fatal Period
Death typically occurs within 24 hours.
Treatment
- Avoid inducing vomiting.
- Perform stomach wash cautiously using diluted acetic or tartaric acid.
- Administer morphine for pain relief.
- Use demulcents like raw eggs or milk.
- Treat esophageal strictures with bougie dilation, and consider gastrostomy or esophagostomy if needed.
- Administer corticosteroids early to prevent strictures.
Postmortem Findings
The gastrointestinal tract shows corrosion, less severe than with mineral acids, with necrosed, inflamed, or blood-stained areas in the mucous membrane, often appearing chocolate or black. Stenosis is common at the lower esophagus.
Medicolegal Importance
Poisoning is usually accidental, often due to ammonia vapor leaks in industrial or storage settings.
FAQs on Chapter Notes: Corrosive Poisons
| 1. What are the common physical properties of corrosive poisons? |  |
| 2. What are the primary signs and symptoms of exposure to corrosive poisons? |  |
| 3. What treatments are available for someone poisoned by a corrosive substance? |  |
| 4. What are the common causes of death associated with corrosive poisonings? |  |
| 5. What are the key postmortem findings in cases of corrosive poisoning? |  |






